Continuing versus Suspending AngiotensinConverting Enzyme Inhibitors and Angiotensin

Continuing versus Suspending Angiotensin-Converting Enzyme Inhibitors and Angiotensin Receptor Blockers: Impact on Adverse Outcomes in Hospitalized Patients with Severe Acute Respiratory Distress Syndrome Coronavirus 2 (SARS-Co. V-2) THE BRACE CORONA TRIAL Renato D. Lopes, MD, Ph. D on behalf of the BRACE CORONA Investigators

Declaration of Interest • Research grants or contracts from Amgen, Bristol-Myers Squibb, Glaxo. Smith. Kline, Medtronic, Pfizer, Sanofi-Aventis. • Funding for educational activities or lectures from Pfizer. • Funding for consulting or other services from Bayer, Boehringer Ingelheim, Bristol-Myers Squibb, Daiichi Sankyo, Portola. • Details at: https: //dcri. org/about-us/conflict-of-interest/ • The BRACE CORONA trial was sponsored by the D'Or Institute for Research and Education (IDOR) and the Brazilian Clinical Research Institute (BCRI).

Background • Membrane-bound angiotensin-converting enzyme 2 (ACE 2) is the functional receptor for SARS-Co. V-2, the virus responsible for the coronavirus disease 2019 (COVID-19). • ACE 2 expression may increase due to upregulation in patients using angiotensinconverting enzyme inhibitors (ACEI) and angiotensin receptor blockers (ARBs). • There is conflicting observational evidence about the potential impact of ACEIs/ARBs on patients with COVID-19. – Select preclinical investigations have raised concerns about their safety in patients with COVID-19. – Preliminary data hypothesize that renin-angiotensin-aldosterone system (RAAS) inhibitors could benefit patients with COVID-19 by decreasing acute lung damage and preventing angiotensin-II-mediated pulmonary inflammation. • Given the frequent use of these agents worldwide, randomized clinical trial evidence is urgently needed to guide the management of patients with COVID-19. Gheblawi M et al. Circulation Res 2020; 126: 1456 -74. Soler MJ et al. Curr Hypertens Rep 2008; 10: 410. Patel AB et al. JAMA 2020; 323: 1769 -1770.
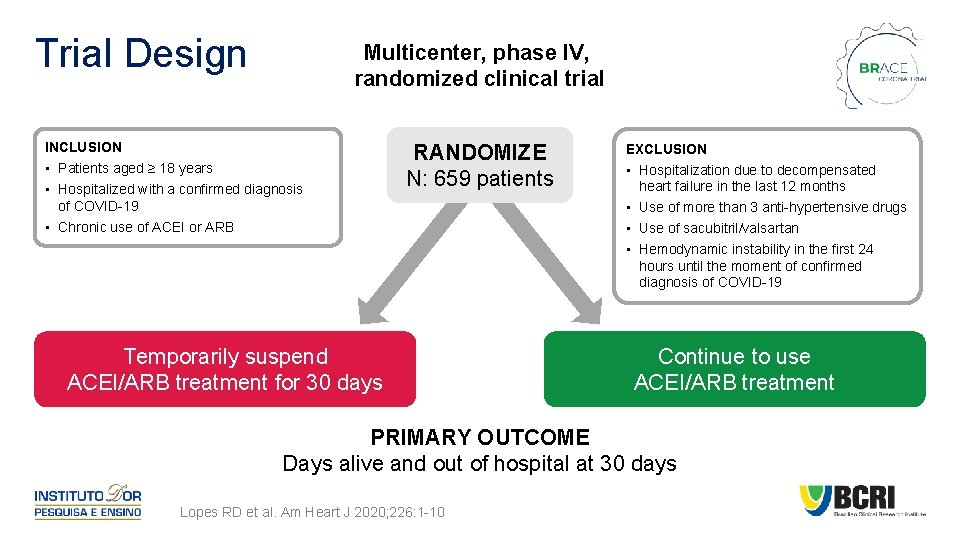
Trial Design Multicenter, phase IV, randomized clinical trial INCLUSION • Patients aged ≥ 18 years • Hospitalized with a confirmed diagnosis of COVID-19 • Chronic use of ACEI or ARB RANDOMIZE N: 659 patients Temporarily suspend ACEI/ARB treatment for 30 days EXCLUSION • Hospitalization due to decompensated heart failure in the last 12 months • Use of more than 3 anti-hypertensive drugs • Use of sacubitril/valsartan • Hemodynamic instability in the first 24 hours until the moment of confirmed diagnosis of COVID-19 Continue to use ACEI/ARB treatment PRIMARY OUTCOME Days alive and out of hospital at 30 days Lopes RD et al. Am Heart J 2020; 226: 1 -10

Trial Organization EXECUTIVE COMMITTEE DATA SAFETY MONITORING BOARD Renato D. Lopes (Study Chair) Alexandre Biasi Cavalcanti (Chair) André Feldman (Brazil) Luciano Drager Andréa Silvestre de Sousa (Brazil) Lucas Petri Damiani Ariane Vieira Scarlatelli Macedo (Brazil) C. Michael Gibson (USA) Christopher B. Granger (USA) CLINICAL EVENTS CLASSIFICATION (CEC) COMMITTEE Denilson Campos de Albuquerque (Brazil) Brazilian Clinical Research Institute (BCRI) Guilherme D`Andréa Saba Arruda (Brazil) John H. Alexander (USA) Lilian Mazza Barbosa (Brazil) ACADEMIC COORDINATING CENTERS Mayara Fraga Santos (Brazil) D'Or Institute for Research and Education (IDOR) Natalia Zerbinatti Salvador (Brazil) Brazilian Clinical Research Institute (BCRI) Olga Ferreira de Souza (Brazil) Pedro Gabriel Melo de Barros e Silva (Brazil) Renata Moll Bernardes (Brazil)

Primary Outcome Number of days alive and out of hospital through 30 days This endpoint represents the follow-up time (30 days or the day of death) minus the hospitalization days. Lopes RD et al. Am Heart J 2020; 226: 1 -10 Fanaroff AC et al. Circ-cardiovasc Qual 2018; 11: e 004755.

Statistical Analysis • The primary analysis followed the intention-to-treat principle. • Continuous variables were described as mean ± standard deviation or medians (25 th, 75 th percentiles) according to the distribution. • As the primary outcome was measured as days alive and out of hospital at 30 days, the analyses were based on the mean and standard deviation as well as median (25 th, 75 th) of this outcome. – The comparison between the groups were made using a generalized additive model for location scale and shape with zero inflated beta-binomial distribution. Lopes RD et al. Am Heart J 2020; 226: 1 -10
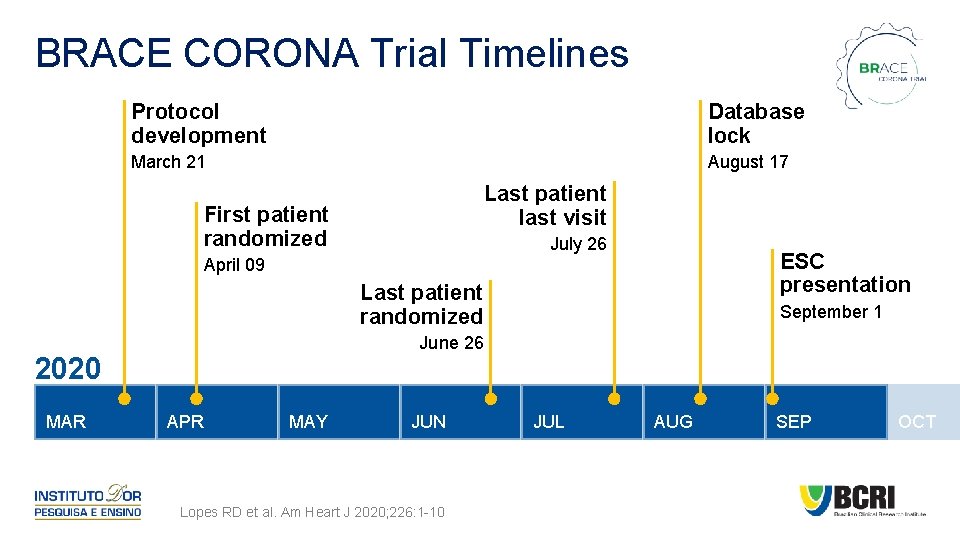
BRACE CORONA Trial Timelines Protocol development Database lock March 21 August 17 Last patient last visit First patient randomized July 26 ESC presentation April 09 Last patient randomized June 26 2020 MAR September 1 APR MAY JUN Lopes RD et al. Am Heart J 2020; 226: 1 -10 JUL AUG SEP OCT
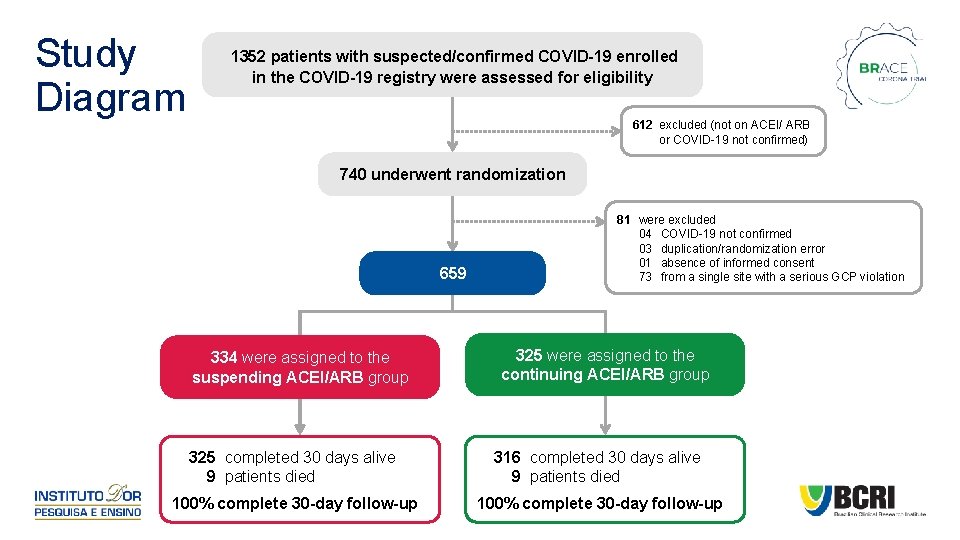
Study Diagram 1352 patients with suspected/confirmed COVID-19 enrolled in the COVID-19 registry were assessed for eligibility 612 excluded (not on ACEI/ ARB or COVID-19 not confirmed) 740 underwent randomization 659 334 were assigned to the suspending ACEI/ARB group 81 were excluded 04 COVID-19 not confirmed 03 duplication/randomization error 01 absence of informed consent 73 from a single site with a serious GCP violation 325 were assigned to the continuing ACEI/ARB group 325 completed 30 days alive 9 patients died 316 completed 30 days alive 9 patients died 100% complete 30 -day follow-up
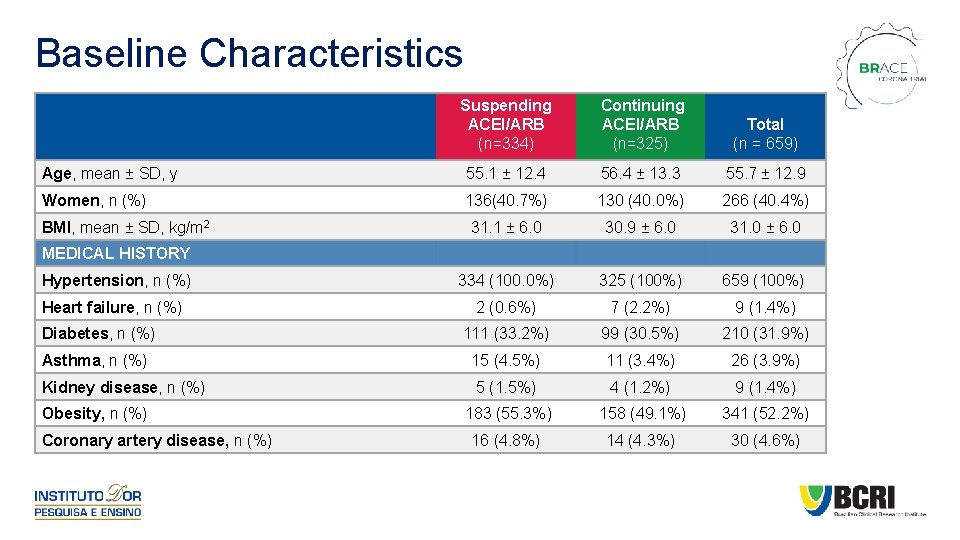
Baseline Characteristics Suspending ACEI/ARB (n=334) Continuing ACEI/ARB (n=325) Total (n = 659) Age, mean ± SD, y 55. 1 ± 12. 4 56. 4 ± 13. 3 55. 7 ± 12. 9 Women, n (%) 136(40. 7%) 130 (40. 0%) 266 (40. 4%) 31. 1 ± 6. 0 30. 9 ± 6. 0 31. 0 ± 6. 0 Hypertension, n (%) 334 (100. 0%) 325 (100%) 659 (100%) Heart failure, n (%) 2 (0. 6%) 7 (2. 2%) 9 (1. 4%) Diabetes, n (%) 111 (33. 2%) 99 (30. 5%) 210 (31. 9%) Asthma, n (%) 15 (4. 5%) 11 (3. 4%) 26 (3. 9%) Kidney disease, n (%) 5 (1. 5%) 4 (1. 2%) 9 (1. 4%) 183 (55. 3%) 158 (49. 1%) 341 (52. 2%) 16 (4. 8%) 14 (4. 3%) 30 (4. 6%) BMI, mean ± SD, kg/m 2 MEDICAL HISTORY Obesity, n (%) Coronary artery disease, n (%)
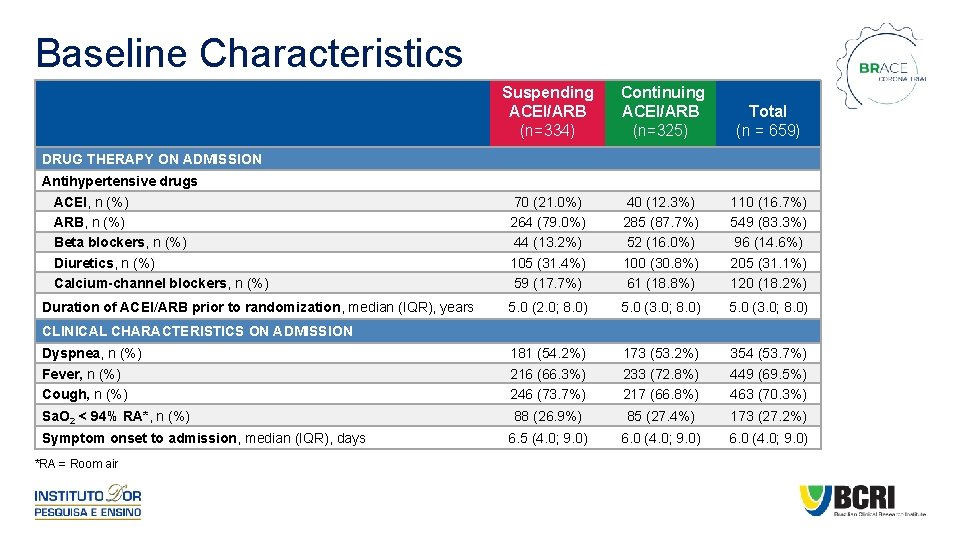
Baseline Characteristics Suspending ACEI/ARB (n=334) Continuing ACEI/ARB (n=325) Total (n = 659) Antihypertensive drugs ACEI, n (%) ARB, n (%) Beta blockers, n (%) Diuretics, n (%) Calcium-channel blockers, n (%) 70 (21. 0%) 264 (79. 0%) 44 (13. 2%) 105 (31. 4%) 59 (17. 7%) 40 (12. 3%) 285 (87. 7%) 52 (16. 0%) 100 (30. 8%) 61 (18. 8%) 110 (16. 7%) 549 (83. 3%) 96 (14. 6%) 205 (31. 1%) 120 (18. 2%) Duration of ACEI/ARB prior to randomization, median (IQR), years 5. 0 (2. 0; 8. 0) 5. 0 (3. 0; 8. 0) Dyspnea, n (%) Fever, n (%) Cough, n (%) 181 (54. 2%) 216 (66. 3%) 246 (73. 7%) 173 (53. 2%) 233 (72. 8%) 217 (66. 8%) 354 (53. 7%) 449 (69. 5%) 463 (70. 3%) Sa. O 2 < 94% RA*, n (%) 88 (26. 9%) 85 (27. 4%) 173 (27. 2%) 6. 5 (4. 0; 9. 0) 6. 0 (4. 0; 9. 0) DRUG THERAPY ON ADMISSION CLINICAL CHARACTERISTICS ON ADMISSION Symptom onset to admission, median (IQR), days *RA = Room air
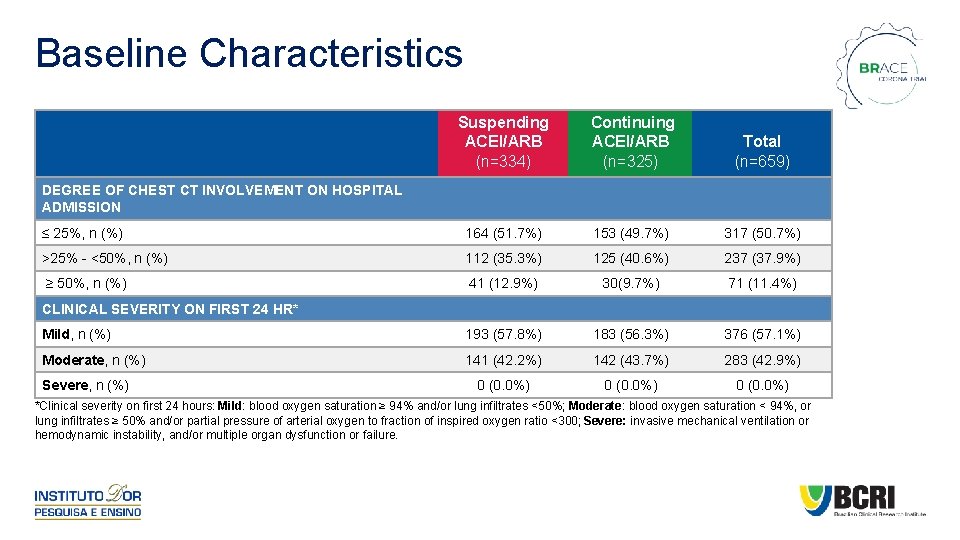
Baseline Characteristics Suspending ACEI/ARB (n=334) Continuing ACEI/ARB (n=325) Total (n=659) ≤ 25%, n (%) 164 (51. 7%) 153 (49. 7%) 317 (50. 7%) >25% - <50%, n (%) 112 (35. 3%) 125 (40. 6%) 237 (37. 9%) ≥ 50%, n (%) 41 (12. 9%) 30(9. 7%) 71 (11. 4%) Mild, n (%) 193 (57. 8%) 183 (56. 3%) 376 (57. 1%) Moderate, n (%) 141 (42. 2%) 142 (43. 7%) 283 (42. 9%) 0 (0. 0%) DEGREE OF CHEST CT INVOLVEMENT ON HOSPITAL ADMISSION CLINICAL SEVERITY ON FIRST 24 HR* Severe, n (%) *Clinical severity on first 24 hours: Mild: blood oxygen saturation ≥ 94% and/or lung infiltrates <50%; Moderate: blood oxygen saturation < 94%, or lung infiltrates ≥ 50% and/or partial pressure of arterial oxygen to fraction of inspired oxygen ratio <300; Severe: invasive mechanical ventilation or hemodynamic instability, and/or multiple organ dysfunction or failure.
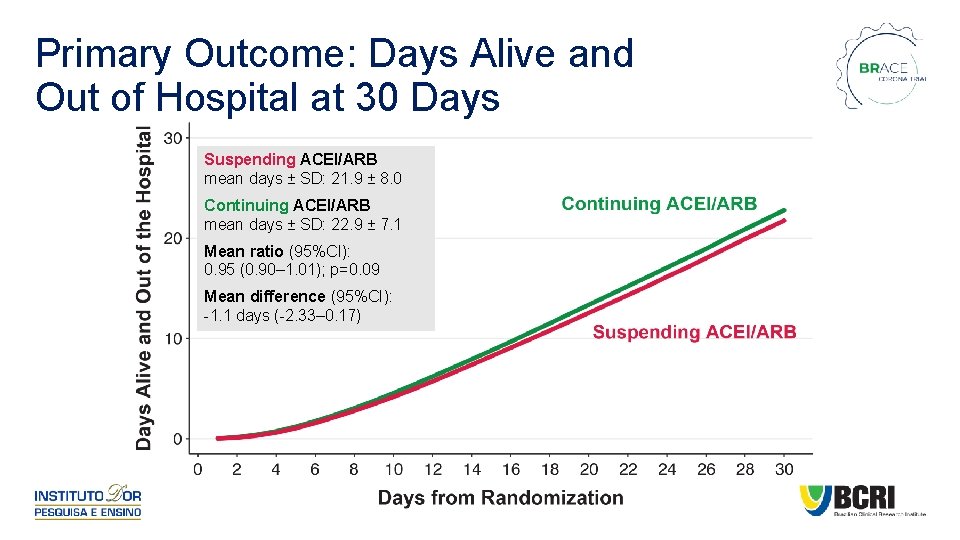
Primary Outcome: Days Alive and Out of Hospital at 30 Days Suspending ACEI/ARB mean days ± SD: 21. 9 ± 8. 0 Continuing ACEI/ARB mean days ± SD: 22. 9 ± 7. 1 Mean ratio (95%CI): 0. 95 (0. 90– 1. 01); p=0. 09 Mean difference (95%CI): -1. 1 days (-2. 33– 0. 17)
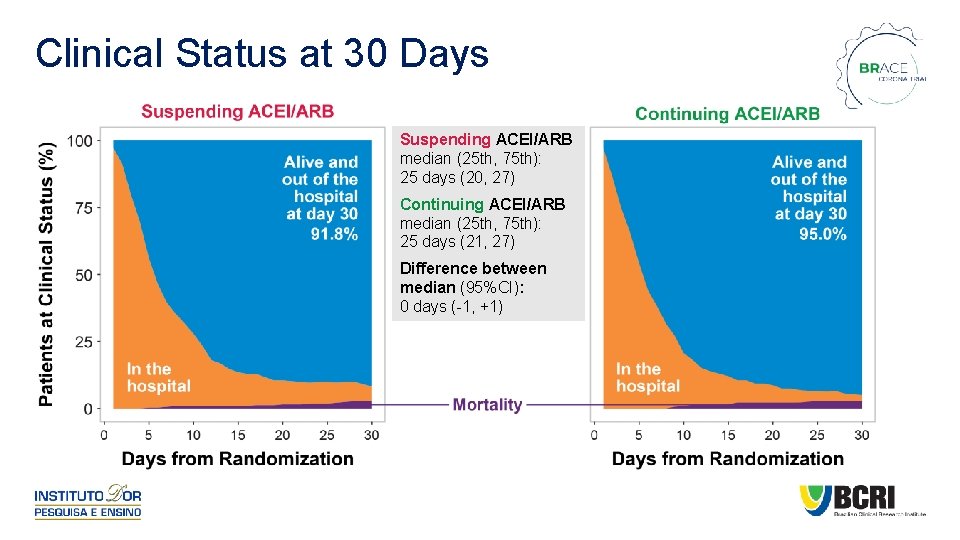
Clinical Status at 30 Days Suspending ACEI/ARB median (25 th, 75 th): 25 days (20, 27) Continuing ACEI/ARB median (25 th, 75 th): 25 days (21, 27) Difference between median (95%CI): 0 days (-1, +1)
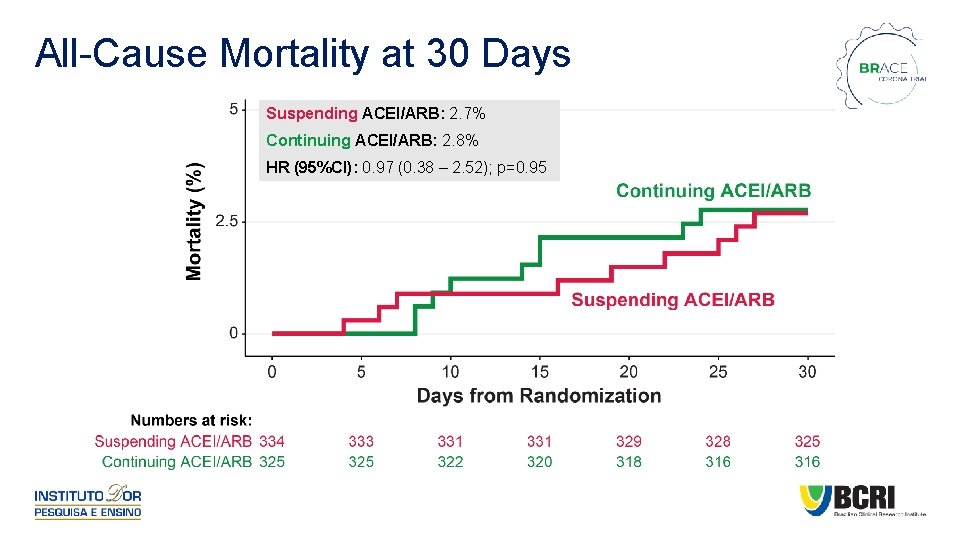
All-Cause Mortality at 30 Days Suspending ACEI/ARB: 2. 7% Continuing ACEI/ARB: 2. 8% HR (95%CI): 0. 97 (0. 38 – 2. 52); p=0. 95

Conclusion In patients hospitalized with COVID-19, suspending ACEI/ARB therapy for 30 days did not impact the number of days alive and out of hospital.

Clinical Implication • The BRACE CORONA provides randomized trial evidence showing that suspending ACEI/ARB therapy for 30 days did not impact the number of days alive and out of hospital. • Because these data indicate that there is no clinical benefit from routinely suspending these medications in hospitalized patients with mild to moderate COVID-19, they should be generally continued for those with an indication. • Our findings constitute contemporary and high-quality randomized evidence to guide the care of patients with COVID-19.

Acknowledgement Thank you to the IDOR and BCRI teams, investigators, study coordinators, and study participants who made BRACE CORONA possible.
- Slides: 18