Continuing versus Suspending AngiotensinConverting Enzyme Inhibitors and Angiotensin

Continuing versus Suspending Angiotensin-Converting Enzyme Inhibitors and Angiotensin Receptor Blockers: Impact on Adverse Outcomes in Hospitalized Patients with Severe Acute Respiratory Distress Syndrome Coronavirus 2 (SARS-Co. V-2) THE BRACE CORONA TRIAL Renato D. Lopes, MD, Ph. D on behalf of the BRACE CORONA Investigators

Declaration of Interest • Research grants or contracts from Amgen, Bristol-Myers Squibb, Glaxo. Smith. Kline, Medtronic, Pfizer, Sanofi-Aventis. • Funding for educational activities or lectures from Pfizer. • Funding for consulting or other services from Bayer, Boehringer Ingelheim, Bristol-Myers Squibb, Daiichi Sankyo, Portola. • Details at: https: //dcri. org/about-us/conflict-of-interest/ • The BRACE CORONA trial was sponsored by D'Or Institute for Research and Education (IDOR) and the Brazilian Clinical Research Institute (BCRI).

Equipoise on the Effects of RAAS Inhibition in COVID-19 HYPOTHESIS 1: RAAS inhibition is harmful. HYPOTHESIS 2: RAAS inhibition is protective. ACEI and ARB could increase ACE 2 receptor expression and thus enhance viral binding and viral entry leading to worse outcomes in patients with COVID-19. Diminishing production of angiotensin II with an ACEI or ARB enhances the generation of angiotensin (1– 7), which attenuates inflammation and fibrosis and therefore could attenuate lung injury. Lopes RD et al. Am Heart J 2020; 226: 1 -10 Jarcho JA et al. N Engl J Med 2020; 382: 2462 -2464
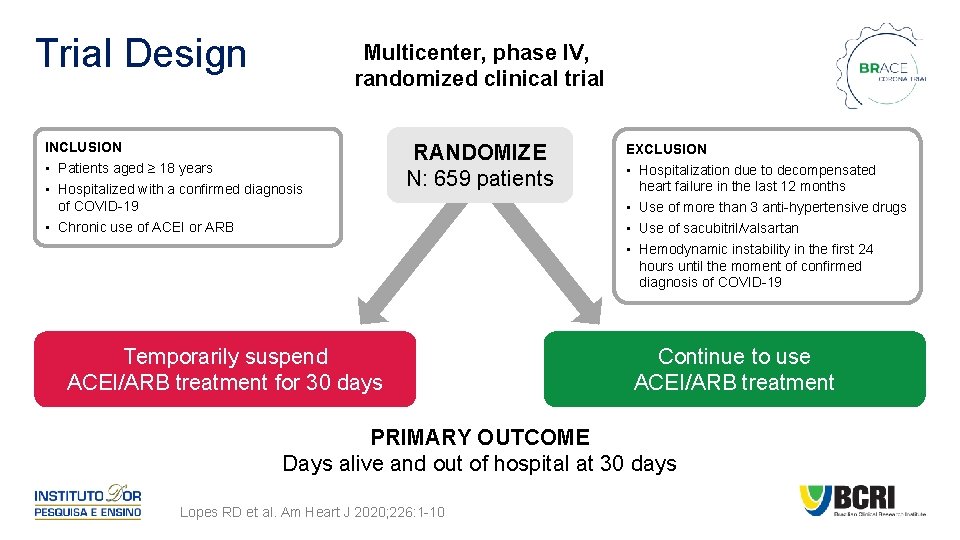
Trial Design Multicenter, phase IV, randomized clinical trial INCLUSION • Patients aged ≥ 18 years • Hospitalized with a confirmed diagnosis of COVID-19 • Chronic use of ACEI or ARB RANDOMIZE N: 659 patients Temporarily suspend ACEI/ARB treatment for 30 days EXCLUSION • Hospitalization due to decompensated heart failure in the last 12 months • Use of more than 3 anti-hypertensive drugs • Use of sacubitril/valsartan • Hemodynamic instability in the first 24 hours until the moment of confirmed diagnosis of COVID-19 Continue to use ACEI/ARB treatment PRIMARY OUTCOME Days alive and out of hospital at 30 days Lopes RD et al. Am Heart J 2020; 226: 1 -10
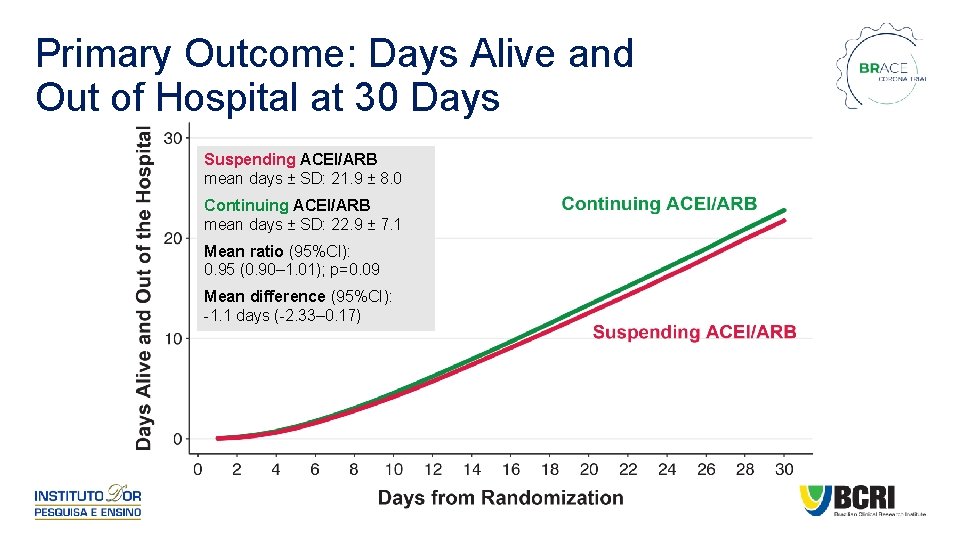
Primary Outcome: Days Alive and Out of Hospital at 30 Days Suspending ACEI/ARB mean days ± SD: 21. 9 ± 8. 0 Continuing ACEI/ARB mean days ± SD: 22. 9 ± 7. 1 Mean ratio (95%CI): 0. 95 (0. 90– 1. 01); p=0. 09 Mean difference (95%CI): -1. 1 days (-2. 33– 0. 17)
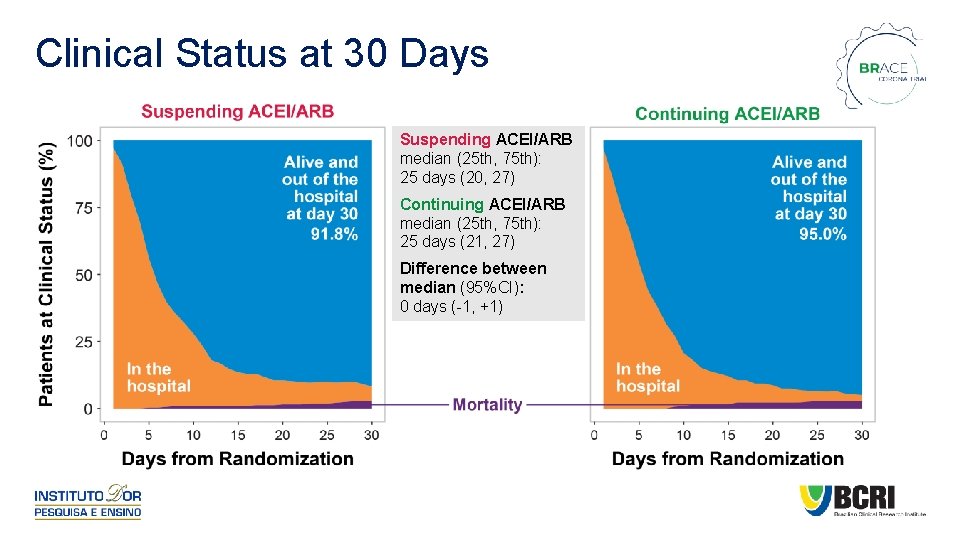
Clinical Status at 30 Days Suspending ACEI/ARB median (25 th, 75 th): 25 days (20, 27) Continuing ACEI/ARB median (25 th, 75 th): 25 days (21, 27) Difference between median (95%CI): 0 days (-1, +1)

Conclusion and Clinical Implication • The BRACE CORONA provides randomized trial evidence showing that suspending ACEI/ARB therapy for 30 days did not impact the number of days alive and out of hospital. • Because these data indicate that there is no clinical benefit from routinely suspending these medications in hospitalized patients with mild to moderate COVID-19, they should be generally continued for those with an indication. • Our findings constitute contemporary and high-quality randomized evidence to guide the care of patients with COVID-19.
- Slides: 7