Content Objective SWBAT Differentiate between atomic number and

Content Objective ● SWBAT ● Differentiate between atomic number and mass number and what they represent ● Relate atomic number and mass number with subatomic particles in an atom. Language Objective ●SWBAT ● Write nuclide symbols with the atomic number and the mass number in the appropriate place.

Subatomic Particles �There are 3 subatomic particles – �Protons �Neutrons �Electrons

Vocabulary �Atom- from the Greek atomos=indivisible. The atom is the smallest particle of an element that retains the properties of that element. �An atom is electrically neutral

What does it mean to be electrically neutral? �The atom has no charge �The number of protons = the number of electrons

Comparing Subatomic Particles Location Outside the nucleus Inside the nucleus

Atomic Number (Z) �Number of protons in the nucleus of an atom �This number is found on the Periodic Table �Atomic Number identifies an element �Always a positive number (b/c it is a counting #) �Tells number of electrons in a neutral atom

Mass Number (A) �Definition - the total number of protons and neutrons in the nucleus of an atom - mass number – atomic number = neutrons ex. N: mass number of 14 atomic number of 7 7 neutrons

Question �What observations can you make about atomic numbers on the periodic table? �Atomic Number increases as you go across the rows from left to right.

Questions �What is the atomic number of Chlorine? �What can you tell me about its protons? �What element has 20 protons? �What is the relationship between the # protons and the atomic number? �They’re equal.
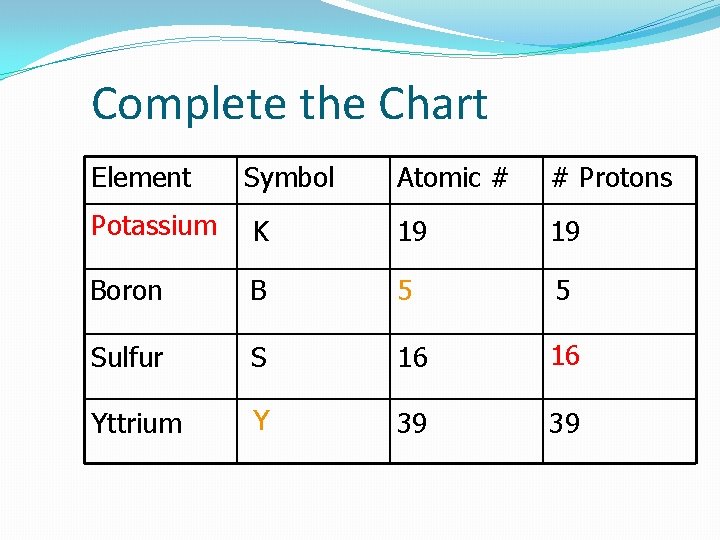
Complete the Chart Element Symbol Atomic # # Protons Potassium K 19 19 Boron B 5 5 Sulfur S 16 16 Yttrium Y 39 39

Question �What do the atomic number and the mass number have in common? �Both Positive integers �Both have the same # of protons

How to find # of Neutrons �Mass # - Atomic# = # Neutrons �Or �# protons + # neutrons= Mass # �(atomic number + # neutrons) = Mass #
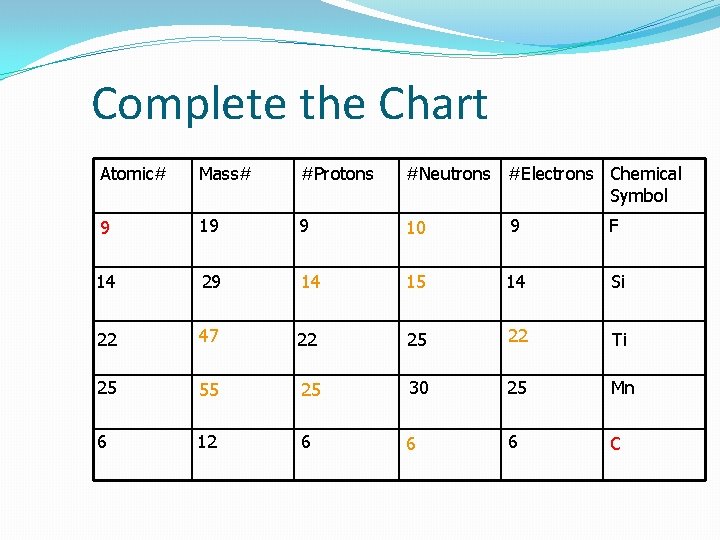
Complete the Chart Atomic# Mass# #Protons #Neutrons #Electrons Chemical Symbol 9 19 9 10 9 F 14 29 14 15 14 Si 22 47 22 25 22 Ti 25 55 25 30 25 Mn 6 12 6 6 6 C

Nuclide Symbol �A=Mass # �Z= Atomic #

Nuclide �A specific kind of atom �Specification of an element in terms of its nuclear composition/structure �Tells number of protons and number of neutrons Chemical Symbol Nuclide Symbol C # protons # neutrons # electrons 6 [6, 7, 8] 6 6 7 6
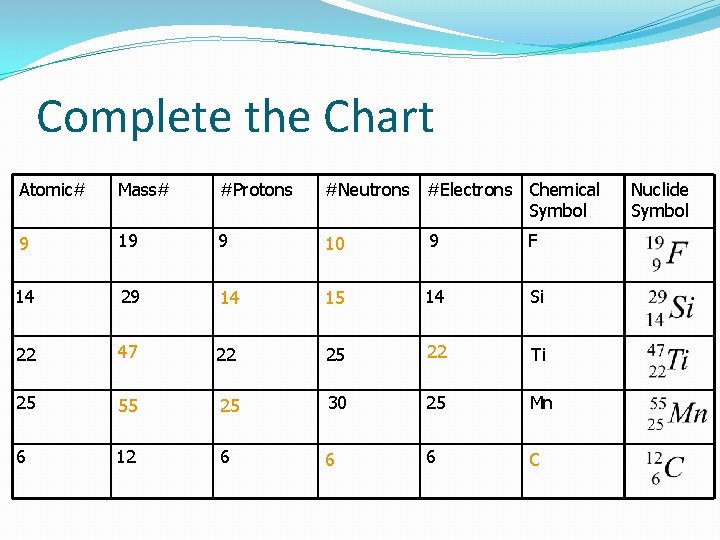
Complete the Chart Atomic# Mass# #Protons #Neutrons #Electrons Chemical Symbol 9 19 9 10 9 F 14 29 14 15 14 Si 22 47 22 25 22 Ti 25 55 25 30 25 Mn 6 12 6 6 6 C Nuclide Symbol

Exit Question �What accounts for the mass of the atom? �# protons & # neutrons in the nucleus
- Slides: 17