Contaminated land dealing with hydrocarbon contamination Petroleum hydrocarbons

Contaminated land: dealing with hydrocarbon contamination Petroleum hydrocarbons – occurrence, composition and significance

Contents of presentation Sources of hydrocarbon contamination n Composition of oils n Non-hydrocarbon components of oil n Hydrocarbon behaviour in the subsurface n The most important components? n Environmental Simulations International

Potential hydrocarboncontaminated sites – 1 n Stating the obvious – Filling stations, distribution depots – Oil production, refineries and associated – Garages/automotive industry – Haulage yards – Scrap metal industry – Airports, aerospace industry – Waste processing & disposal Environmental Simulations International

Potential hydrocarboncontaminated sites – 2 n And also… – Gasworks – Metalworking industry – Paints/inks/coatings industry – Anybody who uses solvents! – Agricultural facilities – Anywhere with a boilerhouse/ furnace n Including domestic oil storage Environmental Simulations International

Composition of oils Crude oil and petroleum products are complex mixtures n They are characterised by differing boiling point ranges and components n – Carbon numbers/simulated distillation Environmental Simulations International
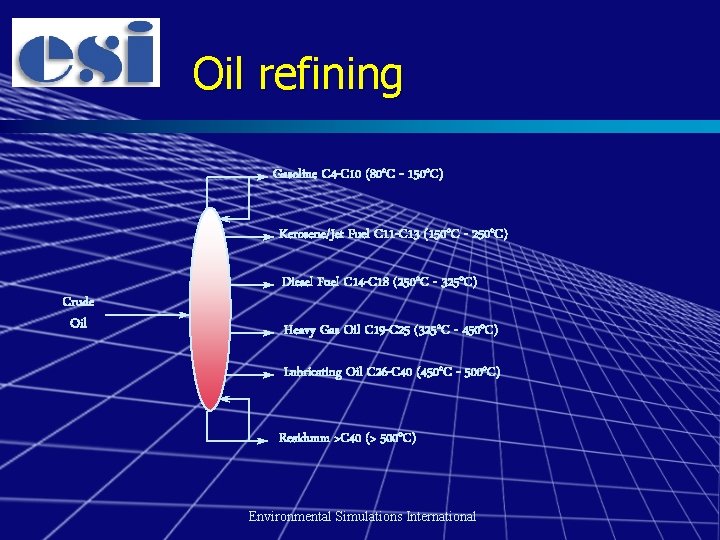
Oil refining Gasoline C 4 -C 10 (80 o. C - 150 o. C) Kerosene/Jet Fuel C 11 -C 13 (150 o. C - 250 o. C) Crude Oil Diesel Fuel C 14 -C 18 (250 o. C - 325 o. C) Heavy Gas Oil C 19 -C 25 (325 o. C - 450 o. C) Lubricating Oil C 26 -C 40 (450 o. C - 500 o. C) Residuum >C 40 (> 500 o. C) Environmental Simulations International

Main component groups Aliphatic hydrocarbons n Aromatic hydrocarbons n NSO components n – Nitrogen, sulphur and oxygen-containing compounds – “Polar components” – “Asphaltenes” Environmental Simulations International
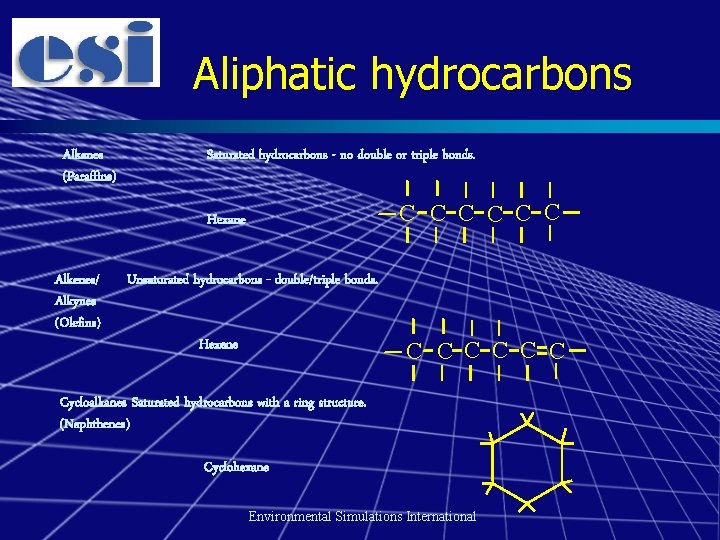
Aliphatic hydrocarbons Alkanes (Paraffins) Saturated hydrocarbons - no double or triple bonds. C C C Hexane Alkenes/ Alkynes (Olefins) Unsaturated hydrocarbons - double/triple bonds. Hexene C C C Cycloalkanes Saturated hydrocarbons with a ring structure. (Naphthenes) Cyclohexane Environmental Simulations International

Aromatic hydrocarbons BTEX CH 3 Benzene C 6 H 6 o-Xylene CH 3 C 8 H 11 CH 3 Toluene C 7 H 8 m-Xylene C 8 H 11 CH 3 CH 2 CH 3 Ethylbenzene C 8 H 10 p-Xylene CH 3 C 8 H 11 Environmental Simulations International CH 3
![Aromatic hydrocarbons – PAH’s Polycyclic aromatic hydrocarbons (PAH) [polynuclear aromatic hydrocarbons (PNA)] Naphthalene C Aromatic hydrocarbons – PAH’s Polycyclic aromatic hydrocarbons (PAH) [polynuclear aromatic hydrocarbons (PNA)] Naphthalene C](http://slidetodoc.com/presentation_image/4fcf8e4b3ea3f1d3dc55b056f3726df7/image-10.jpg)
Aromatic hydrocarbons – PAH’s Polycyclic aromatic hydrocarbons (PAH) [polynuclear aromatic hydrocarbons (PNA)] Naphthalene C 10 H 8 Benzo(a)Pyrene C 20 H 12 Phenanthrene C 14 H 10 Dibenzo(a, h)Anthracene C 22 H 14 Pyrene C 16 H 10 Fluoranthene Environmental Simulations International
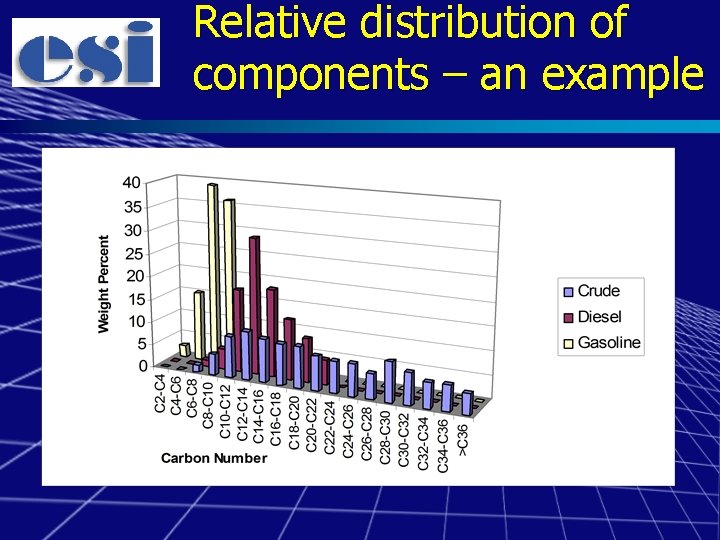
Relative distribution of components – an example

Hydrocarbon behaviour in the subsurface n Oil is a light non-aqueous phase liquid (LNAPL) – – n Residual oil will be held in pore spaces in soil Free oil “floats” on groundwater The more soluble components can dissolve in groundwater – MTBE & TAME are highly soluble n n The more volatile components can partition into soil gas Sorption of components into/onto soil can be an important process Environmental Simulations International

How oil components might be distributed Petrol Spill soil surface Residual Hydrocarbons (~1% HC by weight) in Soil Pores (soil contamination) vadose zone Volatilised (~50 mg HC/kg soil) Constituents (vapour plume) Mobile & Residual Hydrocarbons(~9% by weight) (free-product) Before Oil Flow Through Soil During Oil Flow Through Soil capillary fringe After Oil Flow Through Soil water table saturated zone Dissolved Constituents (plume) (~130 mg HC/L water) groundwater flow
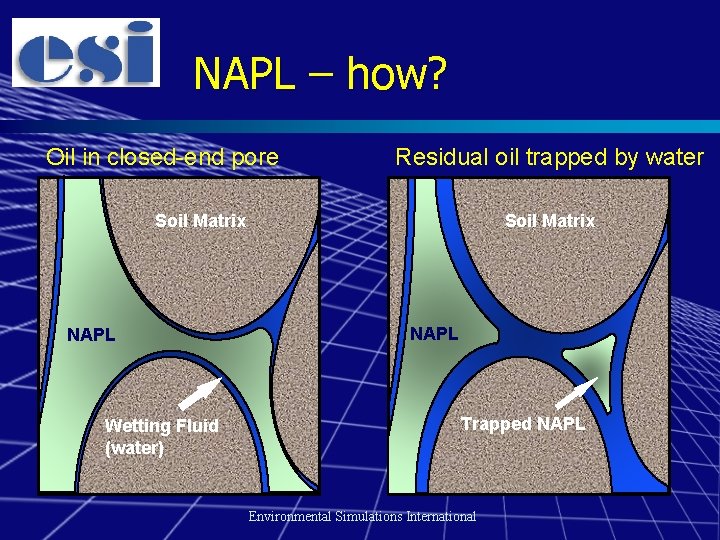
NAPL – how? Oil in closed-end pore Residual oil trapped by water Soil Matrix NAPL Wetting Fluid (water) NAPL Trapped NAPL Environmental Simulations International

Why is NAPL important? n Direct effects of oil at receptor – Also potential effect on buried materials n Residual NAPL can also be a source of contamination for long periods of time – Water soluble components can cause longterm groundwater plumes – Volatile components are a potential source of vapours Environmental Simulations International

Petroleum contamination – what to look for Liquid phase (free-product NAPL) n Dissolved phase (groundwater plume) n Solid phase (hydrocarbon attached to soil) n Vapour phase n Combination of several phases n
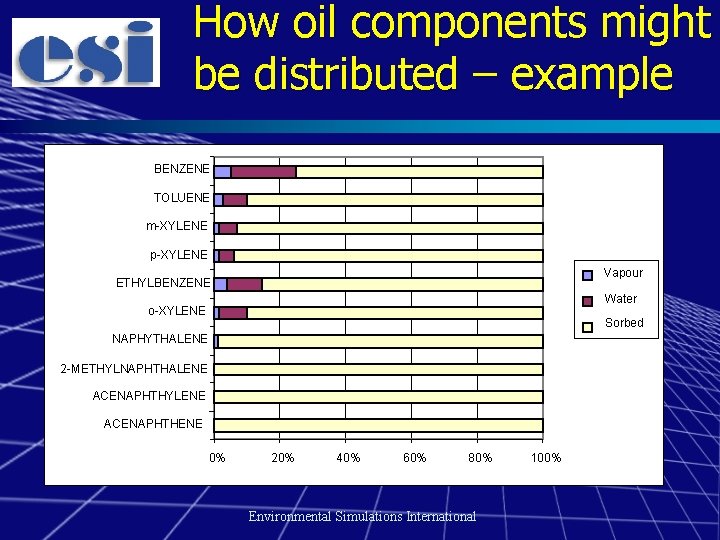
How oil components might be distributed – example BENZENE TOLUENE m-XYLENE p-XYLENE Vapour ETHYLBENZENE Water o-XYLENE Sorbed NAPHYTHALENE 2 -METHYLNAPHTHALENE ACENAPHTHYLENE ACENAPHTHENE 0% 20% 40% 60% 80% Environmental Simulations International 100%

Non-hydrocarbon components n Organic additives – Petrol (gasoline): MTBE, TAME – Fuels: proprietary performance additives – Luboils: proprietary performance additives n Metals – Naturally occurring components of crude n e. g. , vanadium, nickel – Significant contaminants in waste luboils – Leaded petrol (TEL) n Max. 0. 013 g/l in 1980’s leaded petrol but earlier or special use petrol could be higher – Proprietary performance additives Environmental Simulations International

Additives in unleaded petrol MTBE Methyl tertiary butyl ether TAME Tertiary methyl amyl ether C 2 H 5 CH 3 O CH 3 C CH 3 • MTBE most common in UK • Typically 1 -5% in petrol • Highly soluble (26000 mg/l) • Very low taste/odour threshold ETBE Ethyl tertiary butyl ether CH 3 CH 2 CH 3 O Environmental Simulations International C CH 3

Weathering n Preferential reduction in the concentration of some components relative to others – Biodegradation tends to favour removal of nalkanes (straight carbon chain alkanes), low molecular weight cycloalkanes and light aromatics – Volatilisation and dissolution tends to remove low molecular weight aromatics (especially BTEX) and aliphatics – Dissolution is very important for MTBE and TAME n Weathering of organic lead additives Environmental Simulations International
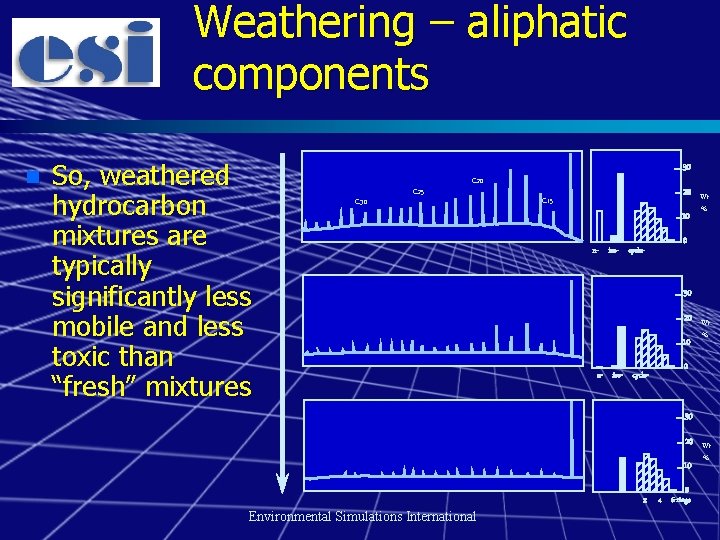
Weathering – aliphatic components n So, weathered hydrocarbon mixtures are typically significantly less mobile and less toxic than “fresh” mixtures C 30 C 25 30 C 20 20 Wt C 15 10 n- iso- cyclo- % 0 30 20 Wt 10 Environmental Simulations International 2 0 4 6 -rings %

Summary n n Hydrocarbon contamination may arise at a wide variety of sites Crude oils and petroleum products are complex mixtures of components – We will discuss the implications further in more detail n n Multiphase behaviour must be considered Weathering may be important Environmental Simulations International

The most important components? n Certain groups of components often merit particular (but not sole) consideration, e. g. – BTEX n Toxicity, vapours – PAH’s n n Potential carcinogenicity, relatively persistent BUT this depends on the source-pathwayreceptor relationships that you are considering – Is the conceptual model sound?
- Slides: 23