Contaminant Hydrogeology VI Yoram Eckstein Ph D Fulbright

Contaminant Hydrogeology VI Гидрогеология Загрязнений и их Транспорт в Окружающей Среде Yoram Eckstein, Ph. D. Fulbright Professor 2013/2014 Tomsk Polytechnic University Tomsk, Russian Federation Fall Semester 2013

Selected Topics in Environmental Chemistry

Acid Base Properties • Water auto dissociates (self dissociation) – 2 H 2 O ↔ H 3 O+(aq) + OH–(aq) Kw = 1. 01 x 10‐ 14@ 25 °C • Neutral water has a p. H = 7. 0

Acid Base Properties • Amphiprotic (Brǿnsted acid or base ) - p. H < 7 can be due to organic acids from decaying organic matter – (COOH)2 + H 2 O ↔ H 3 O+(aq) + COOHCOO–(aq) Ka = 5. 6 × 10‐ 2 - p. H>7 can be due to soluble carbonates from rocks and/or other sources

Redox Chemistry in Natural Waters Oxidation and reduction (Redox) reactions play an important role in the geochemical processes that occur in surface- and ground-water. Redox reactions are defined as reactions in which electrons are transferred. The species receiving electrons is reduced, that donating electrons is oxidized. Redox reactions determine the mobility of many inorganic compounds as well as biologically important materials such as nitrogen and sulfur. In addition, redox conditions govern the particulars for the biological degradation of complex hydrocarbon contaminants

Redox Chemistry in Natural Waters • Most important oxidizing agent in natural waters is dissolved molecular oxygen Half‐reaction in Acidic Solutions O 2+4 H++4 e- 2 H 2 O Half‐reaction in Basic Solutions O 2+2 H 2 O+4 e- 4 OH-

Redox Chemistry in Natural Waters

REDOX Reactions in Aquatic Environments Many elements can exist in a number of oxidation states in near-surface geologic environments, including the macroelements C, N, O, S, Mn and Fe, and important contaminants including As, Se, Cr, Hg, U, Mo, V, Sb, W, Cu, Ag, and Pb. The oxidation state of these elements, in large part, determines the speciation and biogeochemical fate of these elements. Generally, p. H and Eh (p. E) are considered the master geochemical variables controlling the geochemical reactions of elements in geologic and aquatic environments.

REDOX Reactions in Aquatic Environments Redox potential is a an intensity parameter of overall redox reaction potential in the system (similar in concept to p. H), not the capacity of the system for specific oxidation or reduction reactions. The redox value of standard half reactions (Eo) and details of how to calculate redox capacity can be found in any elementary chemistry text. The following inorganic oxidation reactions consume dissolved oxygen in surface- or ground-water: Sulfide Oxidation 2 O 2 + HS- = SO 42 - + H+ Iron Oxidation O 2 + 4 Fe+2 + 4 H+ = 4 Fe 3+ + 2 H 2 O Nitrification 2 O 2 + NH 4+ = NO 3 - + 2 H+ + H 2 O Manganese (II) Oxidation O 2 + 2 Mn 2+ + 2 H 2 O = 2 Mn. O 2 + 4 H+ Iron Sulfide Oxidation 15 O 2 + 4 Fe. S 2 + 14 H 2 O = 4 Fe(OH)3 + 8 SO 42 - + 16 H+

REDOX Reactions in Aquatic Environments The following redox reactions consume organic matter in surfaceor ground-water: (1) Aerobic Degradation CH 2 O + O 2 = CO 2 + H 2 O (2) Denitrification 5 CH 2 O + 4 NO 3 - = 2 N 2 + 5 HCO 3 - + H+ + 2 H 2 O (3) Manganese (IV) Reduction (4) Ferric Iron Reduction CH 2 O + 2 Mn. O 2 + 3 H+ = 2 Mn 2+ + HCO 3 -+ 2 H 2 O CH 2 O + 4 Fe(OH)3 + 7 H+ = 4 Fe 2+ + HCO 3 - + 10 H 2 O (5) Sulfate Reduction 2 CH 2 O + SO 42 - = HS- + 2 HCO 3 - + H+ (6) Methane Fermentation 2 CH 2 O + H 2 O = CH 4 + HCO 3 - + H+

Redox Classification of Natural Waters Ø Oxic waters - waters that contain measurable dissolved oxygen. Ø Suboxic waters - waters that lack measurable oxygen or sulfide, but do contain significant dissolved iron (> ~0. 1 mg L-1). Ø Reducing waters (anoxic) - waters that contain both dissolved iron and sulfide.

The Redox ladder Oxic Post - oxic Sulfidic O 2 H 2 O Aerobes NO 3 - Dinitrofiers N 2 Mn. O 2 Maganese reducers Mn 2+ Fe(OH)3 Iron reducers Fe 2+ SO 42 - Sulfate reducers H 2 S Methanic CO 2 Methanogens CH 4 H O 2 H 2 The redox-couples are shown on each stair-step, where the most energy is gained at the top step and the least at the bottom step. (Gibb’s free energy becomes more positive going down the steps)

BOD and COD BOD and Aquatic Ecosystems

Biochemical Oxygen Demand (BOD) �The capacity ofthe organic and biological matter in a sample of natural water to consume oxygen via catalytic processing of microorganisms present or �The amount of oxygen required by aerobic microorganisms to decompose the organic matter in a sample of water �Easily determined by measuring O 2 before and after sealing a water sample seeded with bacteria

Chemical Oxygen Demand (COD) �Indirectly measures amount of oxygen needed to decompose all organic substances (artificial and natural) by using dichromate ion to oxidize biological and organic matter in a natural water sample �Since stable organics, & anything that can be oxidized are targeted too – so it has always larger values than BOD
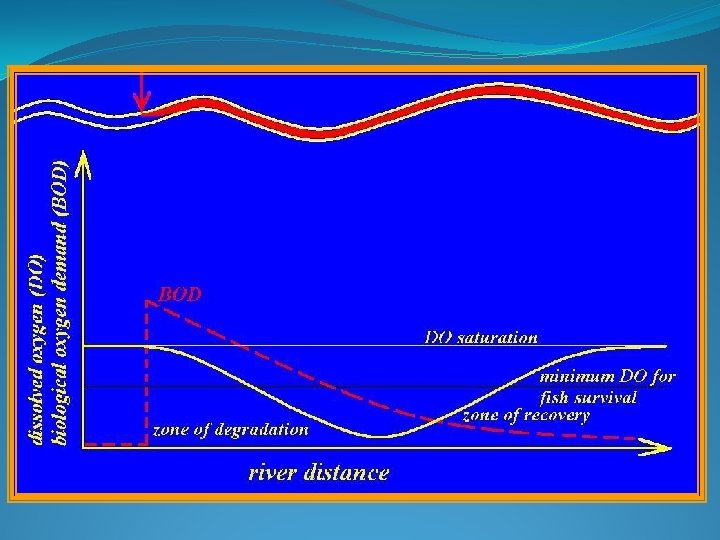
BOD and Aquatic Ecosystems

BOD and Aquatic Ecosystems Rate of the BOD decay in a stream (typically 0. 2/day)

Anaerobic Decomposition of Organic Matter Anaerobic (O 2 free) decomposition of organic matter by microorganisms (fermentation) can produce CH 4 and CO 2 2 CH 2 O Bacteria CH 4 + CO 2 organic matter – In swamps the methane bubbles up to the surface and may ignite – In some rural communities (India, China), ‘digestor units’ convert bio‐organic waste to methane – Process can also occur in landfills

Anaerobic Decomposition of Organic Matter In lakes the lack of oxygen creates a reducing environment at the bottom • Insoluble Fe 3+ (+e‐) Soluble Fe 2+ • Mixing does occur with seasons

Sulfur in Natural Waters Hydrogen sulfide: Sources include, volcanoes, hot springs, swamps (anaerobic bacteria), 10 – 15% from anthropogenic sources (oil refinery, natural gas wells, paper mills, ore smelting, etc)

Sulfur in Natural Waters H 2 S Solubility H 2 S + 2 O 2 H 2 SO 4 4370 ml/L at 0 °C; 1860 ml/L at 40 °C Some anaerobic bacteria can decompose various sulfur containing organic matter (amino acids, etc) and produce, amongst other things, hydrogen sulfide, CH 3 SH, CH 3 SSCH 3, etc. 2 SO 42 -+3 CH 2 O+4 H+ 2 S+3 CO 2+5 H 2 O Overall reaction showing how some anaerobic bacteria can use sulfate ion as the oxidizing agent to decompose organic matter (important in seawater , where sulfate concentration is much higher than fresh water

Acid Mine Drainage (AMD) “Outflow of acidic water (p. H<3. 0) from abandoned coal or metal/ore mines” – Typically occurs when certain geology is exposed (mining, construction etc) to water or air resulting in the oxidation of these minerals Fe. S 2 Pyrite Fe. S 2 Marcasite Fex. Sx Pyrrhotite Cu 2 S Chalcocite Cu. S Covellite Cu. Fe. S 2 Chalcopyrite Mo. S 2 Molybdenite Ni. S Millerite Pb. S Galena Zn. S Sphalerite Fe. As. S Arsenopyrite

Acid Mine Drainage (AMD) 2 Fe. S 2 + 7 O 2 + 2 H 2 O 2 Fe 2+ + 3 SO 42 - + 4 H+ (I) First step produces acidity 4 Fe 2+ + O 2+ 4 H+ 2 H 2 O + 4 Fe 3+ (II) Second step is usually slow, but can be catalyzed by acidic bacteria, & consumes some of the H produced in (I) 4 Fe 3+ + 12 H 2 O 4 Fe(OH)3 + 12 H+ (III) 3 rd step the Fe 3+ is soluble in high p. H water initially produced; however as AMD becomes more diluted the hydroxide precipitates, giving the yellowish brown color (kills!!)

Acid Mine Drainage (AMD) 4 Fe. S 2 + 14 Fe 3+ + 8 H 2 O 15 Fe 2+ + 2 SO 42 - + 16 H+ Fe 3+ then catalyses further production of acidity, without the need for O 2

Consequences and Some Solution to AMD Consequences • High acidity leads to leaching of various metals – Heavy metals (Fe, Cu, Pb, Zn, Cd, Co, Cr, Ni, and Hg) – Metalloids (As Sb) – Other metals & elements Al, Mn, Si, Ca, Na, K, Mg and Ba – Whatever maybe present • Can get p. H < 0! • Discoloration of waterways, choking & killing of aquatic organisms (fish, plant, microorganisms, etc)

Consequences and Some Solution to AMD Solutions • Some mines near natural limestone deposits which can neutralize the AMD • Limestone chips can be added • Addition of calcium oxide or hydroxide • Anaerobic bacteria • Sealing the mines

The p. E Scale Used to characterize the reducing nature of natural water • Low p. E ~ lots of electrons available thus water is very reducing • High p. E ~ few electrons available so dominant species are oxidizing in nature • It is defined as – log 10 of the effective concentration (or activity of) electrons in water – Analogous to p. H scale, recall you really don’t have bare protons, so don’t really have bare electrons’, – Dimensionless numbers (i. e. no units) • Along with p. H it can be used to determine the dominant species in a body of water

Example Using p. E Scenario: Traditional leather tanning industries soak the hides in an aqueous solution of chromium (III). Suppose the waste water from the tannery contains 26 mg/L chromium (III). As it enters a river, the dissolved oxygen can oxidize the chromium (III) to dichromate. If the water in the river is well aerated and has a p. H of 7. 0, what’s the dominant species? Step I Large amounts of dissolved O 2 in H 2 O; dominant proces O 2 + 4 H+ + 4 e- 2 H 2 O is reduction of O 2 p. E = p. Eo – (1/n) log. Q

Example Using p. E O 2 + 4 H+ + 4 e- 2 H 2 O Step I p. E = p. Eo – (1/n) log. Q p. Eo = Eo/(0. 0591) = 1. 229/0. 0591 = = 20. 795 p. E = 20. 795 – (1/4)log{1/(PO 2 [H+]4)} = = 14. 63 • Can use this step for any well aerated water system, as long as you know the p. H • Assumption O 2 in equilibrium with the water • Assumption is no dissolved CO 2

Example Using p. E Cr 2 O 72 -(aq) + 14 H+(aq) + 6 e- 2 Cr 3+(aq) + 7 H 2 O Step 2: Calculate p. Eo = Eo/0. 0591 = 1. 33/0. 0591 = 22. 504 Step 3: set up the correct expression for Q, & substitute in your known values 14. 63 = 22. 504 – - (1/6)log{[Cr 3+]/([Cr 2 O 72 -][10 -7]14)} The chromium and oxygen are in the same water system, so they are at equilibrium i. e. same p. E!!!

Example Using p. E Step 4: Simple rule of logs re‐arrange the expression 14. 63 = 22. 504 – (1/6)log(1/[10 -7]14) – - (1/6)log([Cr 3+]/([Cr 2 O 72 -][10 -7]14) Step 5: Simplify the expression 14. 63 = 8. 504 - (1/6)log([Cr 3+]/[Cr 2 O 72 -]) -36. 756 = log([Cr 3+]/[Cr 2 O 72 -]) and finally: 1. 75 × 10 -37 = [Cr 3+]/[Cr 2 O 72 -] This small number means the dichromate dominates! Which is very toxic, and carcinogenic

Acid Base Chemistry in Natural Waters Natural waters contain lots of CO 2 – Source mostly from air, but can be from decomposition of organics – Easily forms carbonic acid – Acid easily dissociates – Reason rain water slightly acidic – the p. H of CO 2 saturated water is 5. 6 @ 25 °C, given that the [CO 2] is 365 ppm
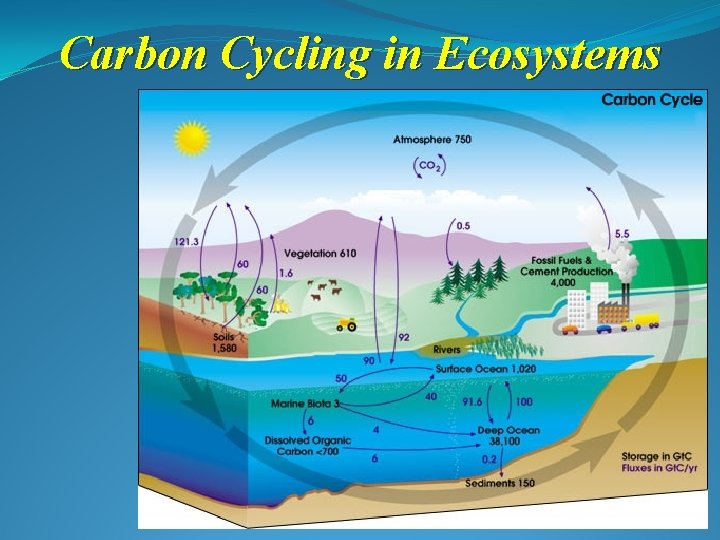
Carbon Cycling in Ecosystems

Carbon Dioxide and Water CO 2 + H 2 O ↔ H 2 CO 3 (KH= 10 -1. 5 mol/atm·L) H 2 CO 3 ↔ HCO 3 - + H+ (H 1 a≈ 10 -6. 3 mol/L) HCO 3 - ↔ CO 32 - + H+ (H 2 a≈ 10 -10. 3 mol/L)

Acid Base Chemistry in Natural Waters Oceans are a large sink for atmospheric CO 2 – Sequestration of CO 2 in the ocean would increase the acidity of surrounding waters – Increased acidity could be detrimental to some ocean life – Increase in atmospheric CO 2 has decreased ocean p. H ~ 0. 1

Biotransformation and Biodegradation Aerobic Anaerobic

Biotransformation Ø Biotransformation is the chemical modification (or modifications) made by an organism on chemical compounds such as (but not limited to) nutrients, amino acids, toxins, etc. Ø If this modification ends in mineral compounds like CO 2, NH 4+, or H 2 O, the biotransformation is called mineralisation.

Biotransformation Ø Biotransformation of various pollutants is a sustainable way to clean up contaminated environments. These bioremediation and biotransformation methods harness the naturally occurring, microbial catabolic diversity to degrade, transform or accumulate a huge range of compounds including hydrocarbons (e. g. oil), polychlorinated biphenyls (PCBs), polyaromatic hydrocarbons (PAHs), pharmaceutical substances, radionuclides and metals.

Biotransformation Ø Biological processes play a major role in the removal of contaminants and pollutants from the environment. Some microorganisms possess an astonishing catabolic versatility to degrade or transform such compounds. New methodological breakthroughs in sequencing, genomics, proteomics, bioinformatics and imaging are producing vast amounts of information.

Biotransformation Ø As we learn more through functional genomic analysis of various bacterial species, biological processes are gradually replacing some older physico-chemical methods; Ø Some technologies that have been used are: Ø high-temperature incineration Ø various types of chemical decomposition (e. g. , base-catalyzed dechlorination) Ø UV oxidation.
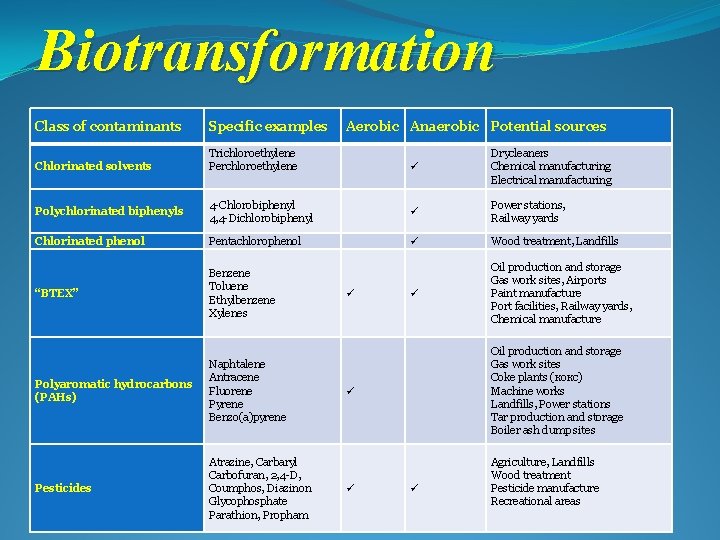
Biotransformation Class of contaminants Specific examples Aerobic Anaerobic Potential sources Chlorinated solvents Trichloroethylene Perchloroethylene Drycleaners Chemical manufacturing Electrical manufacturing Polychlorinated biphenyls 4 -Chlorobiphenyl 4, 4 -Dichlorobiphenyl Power stations, Railway yards Chlorinated phenol Pentachlorophenol Wood treatment, Landfills “BTEX” Benzene Toluene Ethylbenzene Xylenes Oil production and storage Gas work sites, Airports Paint manufacture Port facilities, Railway yards, Chemical manufacture Polyaromatic hydrocarbons (PAHs) Naphtalene Antracene Fluorene Pyrene Benzo(a)pyrene Pesticides Atrazine, Carbaryl Carbofuran, 2, 4 -D, Coumphos, Diazinon Glycophosphate Parathion, Propham Oil production and storage Gas work sites Coke plants (кокс) Machine works Landfills, Power stations Tar production and storage Boiler ash dump sites Agriculture, Landfills Wood treatment Pesticide manufacture Recreational areas

Biotransformation Ø Microbes will adapt and grow at subzero temperatures, as well as extreme heat, desert conditions, in water, with an excess of oxygen, and in anaerobic conditions, with the presence of hazardous compounds or on any waste stream. Ø The main requirements are an energy source and a carbon source. Ø Because of the adaptability of microbes and other biological systems, these can be used to degrade or remediate environmental hazards.

Biotransformation We can subdivide these microorganisms into the following four groups: Ø Aerobic Ø Anaerobic Ø Ligninolytic fungi Ø Methylotrophs

Biotransformation Ø Aerobic bacteria v In the presence of oxygen. v Examples of aerobic bacteria recognized for their degradative abilities are Pseudomonas, Alcaligenes, Sphingomonas, Rhodococcus, and Mycobacterium. These microbes have often been reported to degrade pesticides and hydrocarbons, both alkanes and polyaromatic compounds. v Many of these bacteria use the contaminant as the sole source of carbon and energy.

Biotransformation Ø Anaerobic bacteria v In the absence of oxygen. v Anaerobic bacteria are not as frequently used as aerobic bacteria. v There is an increasing interest in anaerobic bacteria used for bioremediation of polychlorinated biphenyls (PCBs) in river sediments, dechlorination of the solvent trichloroethylene (TCE), and chloroform.

Biotransformation Ø Ligninolytic fungi v Fungi such as the white rot fungus Phanaerochaete chrysosporium have the ability to degrade an extremely diverse range of persistent or toxic environmental pollutants. v Common substrates used include straw, saw dust, or corn cobs.

Biotransformation Ø Methylotrophs v Aerobic bacteria that grow utilizing methane for carbon and energy. v The initial enzyme in the pathway for aerobic degradation, methane monooxygenase, has a broad substrate range and v is active against a wide range of compounds, including the chlorinated aliphatics trichloroethylene and 1, 2 -dichloroethane.

Environmental Factors in Biotransformation Ø Nutrients v Although the microorganisms are present in contaminated soil, they cannot necessarily be there in the numbers required for ioremediation of the site. Their growth and activity must be stimulated. v Biostimulation usually involves the addition of nutrients and oxygen to help indigenous microorganisms.

Environmental Factors in Biotransformation v These nutrients are the basic building blocks of life and allow microbes to create the necessary enzymes to break down the contaminants. v All of them will need nitrogen, phosphorous, and carbon v Carbon is the most basic element of living forms and is needed in greater quantities than other v elements. In addition to hydrogen, oxygen, and nitrogen it constitutes about 95% of the weight of cells.
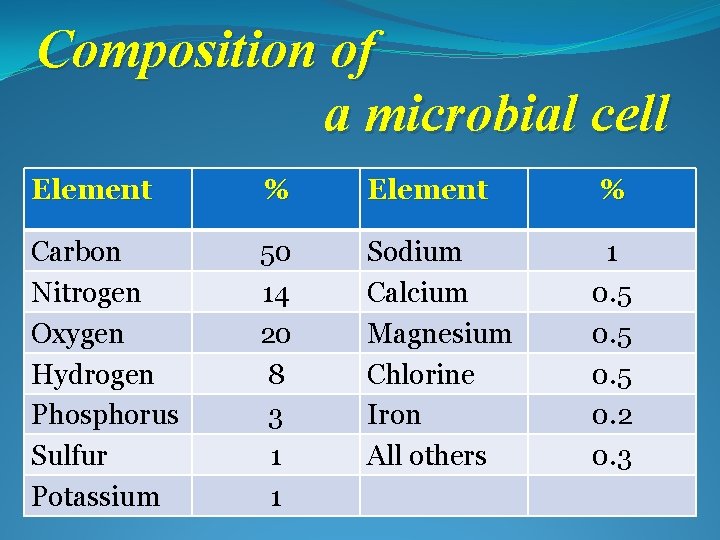
Composition of a microbial cell Element % Carbon Nitrogen Oxygen Hydrogen Phosphorus Sulfur Potassium 50 14 20 8 3 1 1 Sodium Calcium Magnesium Chlorine Iron All others 1 0. 5 0. 2 0. 3

Optimum environmental conditions for the degradation of contaminants Parameters Condition required for microbial activity Soil moisture Soil p. H 25– 28% of water 5. 5– 8. 8 Oxygen content Nutrient content T (°C) Aerobic, minimum airfilled pore space of 10% N & P Contaminants 15– 45 Not too toxic Heavy metals Type of soil Total content 2000 ppm Low clay or silt content Optimum value for an oil degradation 30– 90% 6. 5– 8. 0 10– 40% C: N: P = 100: 1 20 -30 5– 10% of dry weight of soil 700 ppm

Oil biodegradation Ø Petroleum oil is toxic for most life forms and episodic and chronic pollution of the environment by oil causes major ecological perturbations. Marine environments are especially vulnerable, since oil spills of coastal regions and the open sea are poorly containable and mitigation is difficult. Ø In addition to pollution through human activities, millions of tons of petroleum enter the marine environment every year from natural seepages.
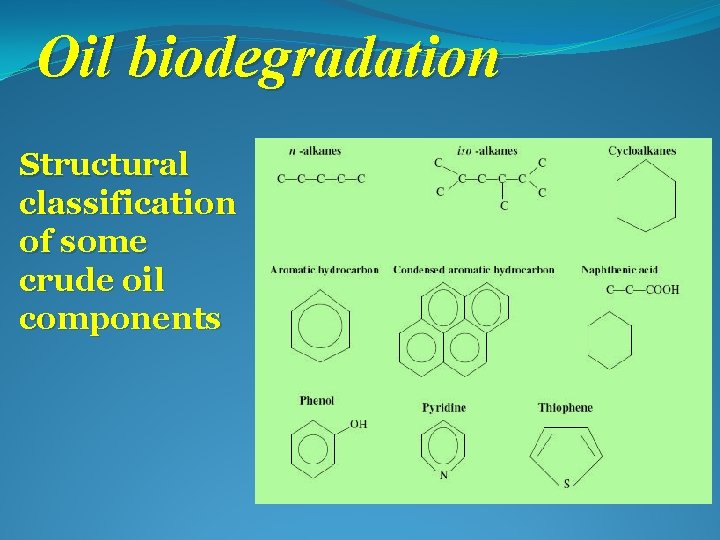
Oil biodegradation Structural classification of some crude oil components
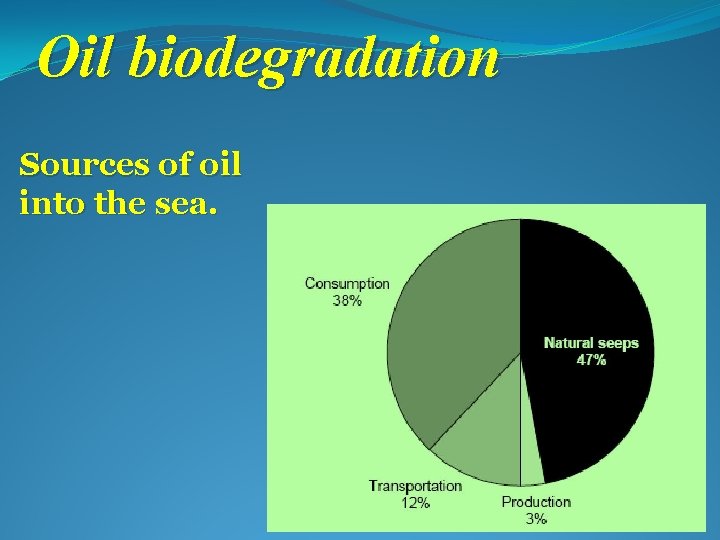
Oil biodegradation Sources of oil into the sea.
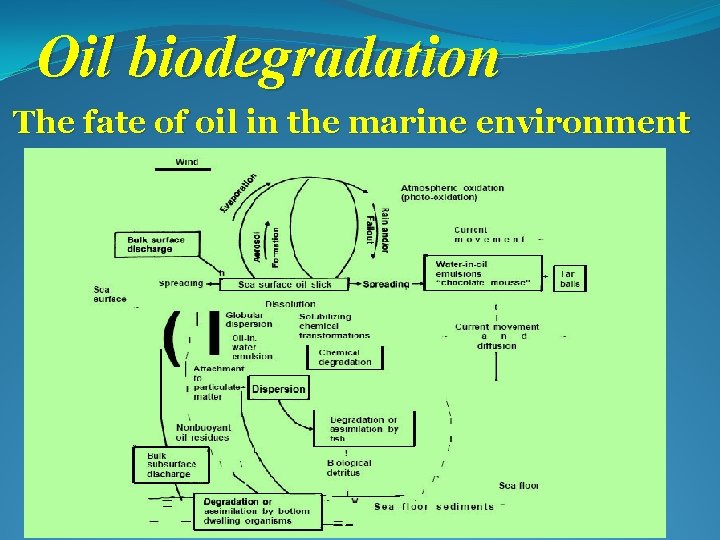
Oil biodegradation The fate of oil in the marine environment
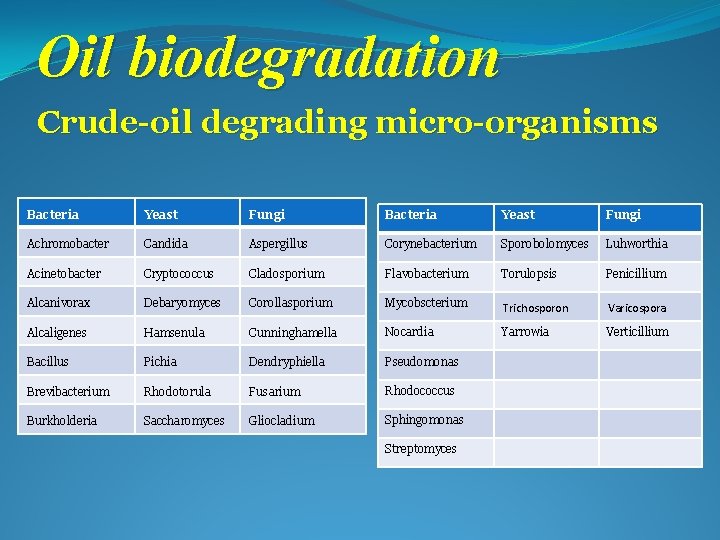
Oil biodegradation Crude-oil degrading micro-organisms Bacteria Yeast Fungi Achromobacter Candida Aspergillus Corynebacterium Sporobolomyces Luhworthia Acinetobacter Cryptococcus Cladosporium Flavobacterium Torulopsis Penicillium Alcanivorax Debaryomyces Corollasporium Mycobscterium Trichosporon Varicospora Alcaligenes Hamsenula Cunninghamella Nocardia Yarrowia Verticillium Bacillus Pichia Dendryphiella Pseudomonas Brevibacterium Rhodotorula Fusarium Rhodococcus Burkholderia Saccharomyces Gliocladium Sphingomonas Streptomyces
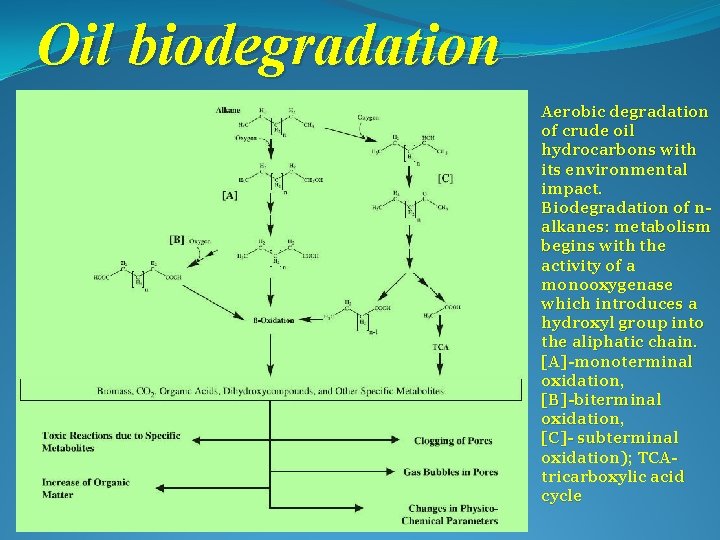
Oil biodegradation Aerobic degradation of crude oil hydrocarbons with its environmental impact. Biodegradation of nalkanes: metabolism begins with the activity of a monooxygenase which introduces a hydroxyl group into the aliphatic chain. [A]-monoterminal oxidation, [B]-biterminal oxidation, [C]- subterminal oxidation); TCAtricarboxylic acid cycle
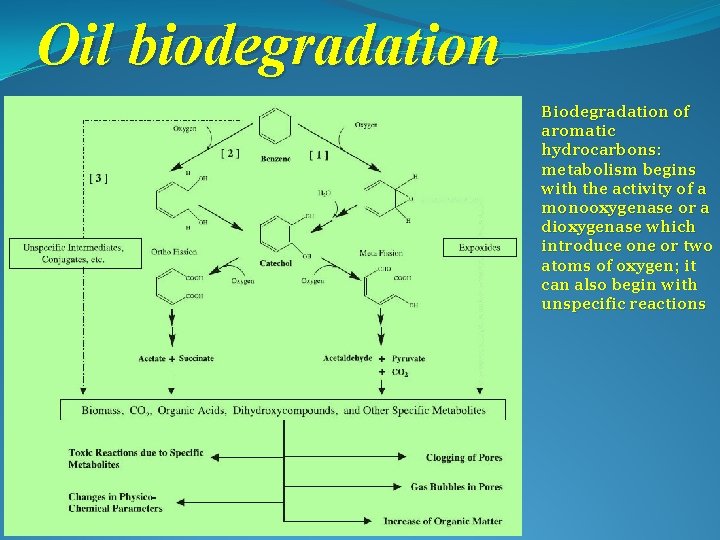
Oil biodegradation Biodegradation of aromatic hydrocarbons: metabolism begins with the activity of a monooxygenase or a dioxygenase which introduce one or two atoms of oxygen; it can also begin with unspecific reactions
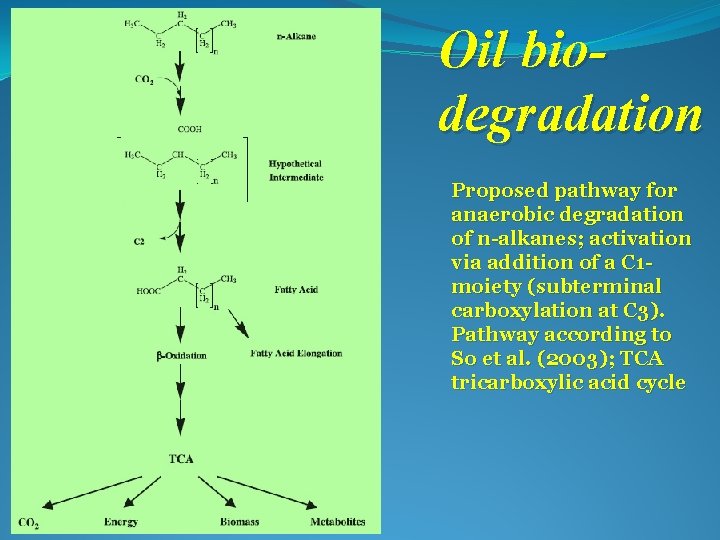
Oil biodegradation Proposed pathway for anaerobic degradation of n-alkanes; activation via addition of a C 1 moiety (subterminal carboxylation at C 3). Pathway according to So et al. (2003); TCA tricarboxylic acid cycle
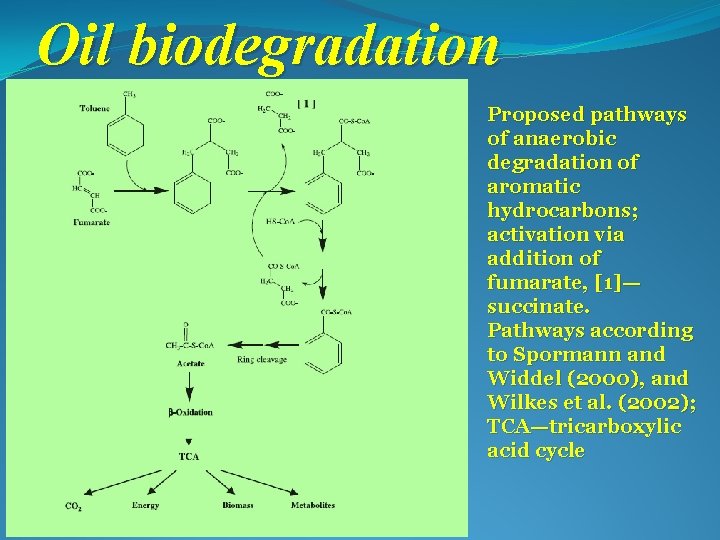
Oil biodegradation Proposed pathways of anaerobic degradation of aromatic hydrocarbons; activation via addition of fumarate, [1]— succinate. Pathways according to Spormann and Widdel (2000), and Wilkes et al. (2002); TCA—tricarboxylic acid cycle

Oil biodegradation Ø Despite its toxicity, a considerable fraction of petroleum oil entering marine systems is eliminated by the hydrocarbon-degrading activities of microbial communities, in particular by a remarkable recently discovered group of specialists, the so-called hydrocarbonoclastic bacteria (HCB). Ø Alcanivorax borkumensis, a paradigm of HCB and probably the most important global oil degrader, was the first to be subjected to a functional genomic analysis.

Oil biodegradation Ø The functional genomic analysis of Alcanivorax borkumensis has yielded important new insights into its capacity for (i) n-alkane degradation including metabolism, biosurfactant production and biofilm formation, (ii) scavenging of nutrients and cofactors in the oligotrophic marine environment, as well as (iii) coping with various habitat-specific stresses.

Oil biodegradation Compound Reaction Naphthalene (C 10 H 8) C 10 H 8 + 20 H 2 O → 10 CO 2 + 48 H+ + 48 e- 1 -Methylnaphthalene (C 11 H 10) C 11 H 10 + 22 H 2 O → 11 CO 2 + 54 H+ + 54 e- 2 -Methylnaphthalene (C 11 H 10) C 11 H 10 + 22 H 2 O → 11 CO 2 + 54 H+ + 54 e 2 -Ethylnaphtalene (C 12 H 12) C 12 H 12 + 24 H 2 O → 12 CO 2 + 60 H+ + 60 e- Phenanthrene (C 14 H 10) C 14 H 10 + 28 H 2 O → 14 CO 2 + 66 H+ + 66 e- Anthracene (C 14 H 10) Pyrene (C 16 H 10) C 14 H 10 + 28 H 2 O → 14 CO 2 + 66 H+ + 66 e. C-16 H 10 + 32 H 2 O → + 16 CO 2 + 74 H+ + 74 e Benzene (C 6 H 6) C 6 H 6 + 12 H 2 O → + 6 CO 2 + 30 H+ + 30 e- Toluene (C 7 H 8) C 7 H 8 + 14 H 2 O → + 7 CO 2 + 36 H+ + 36 e- Xylene (C 8 H 10) C 8 H 10 + 16 H 2 O → + 8 CO 2 + 42 H+ + 42 e-

Mycoremediation Ø Mycoremediation is a form of bioremediation, the process of using mushrooms to return an environment (usually soil) contaminated by pollutants to a less contaminated state. Ø In an experiment a plot of soil contaminated with diesel oil was inoculated with mycelia of oyster mushrooms; traditional bioremediation techniques (bacteria) were used on control plots. After four weeks, more than 95% of many of the PAH (polycyclic aromatic hydrocarbons) had been reduced to non-toxic components in the mycelial-inoculated plots.

Other biodegradation processes Ø The bacterium Deinococcus radiodurans (the most radioresistant organism known) has been modified to consume and digest toluene and ionic mercury from highly radioactive nuclear waste.

Energy and Mass Cycling in Ecosystems we all are eating solar energy In most ecosystems, sunlight is absorbed and converted into usable forms of energy via photosynthesis. These usable forms of energy are carbon-based.
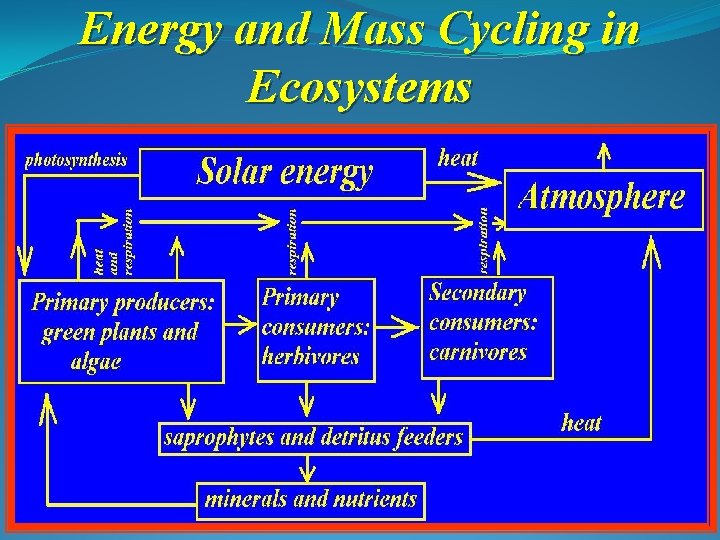
Energy and Mass Cycling in Ecosystems
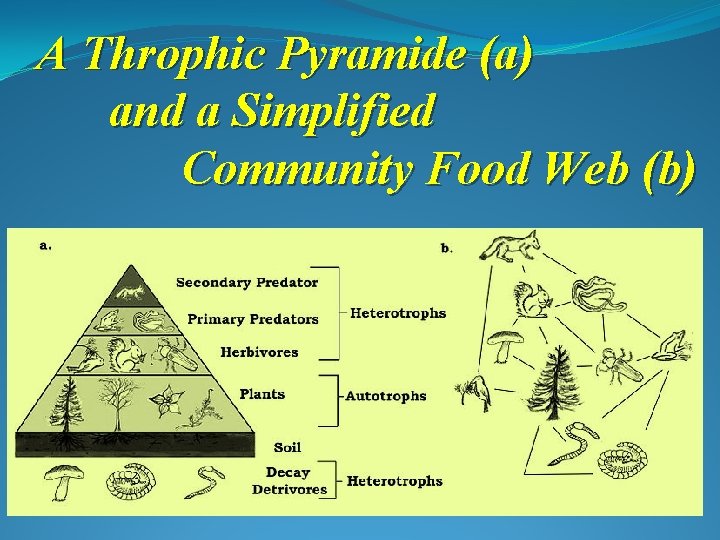
A Throphic Pyramide (a) and a Simplified Community Food Web (b)
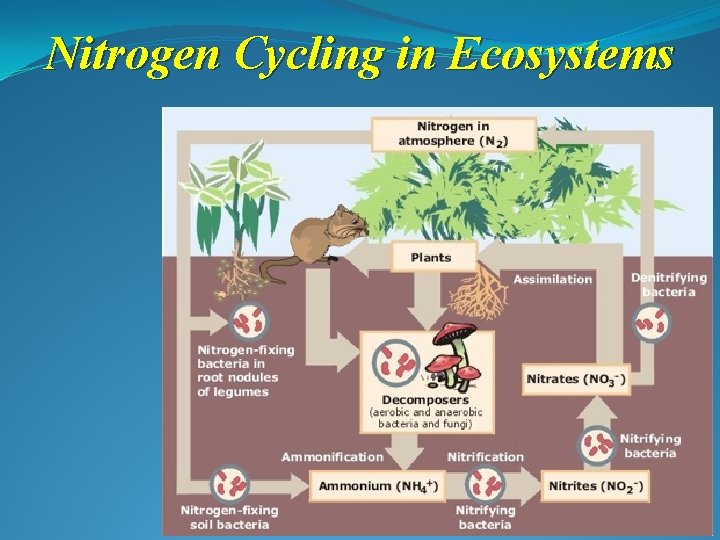
Nitrogen Cycling in Ecosystems
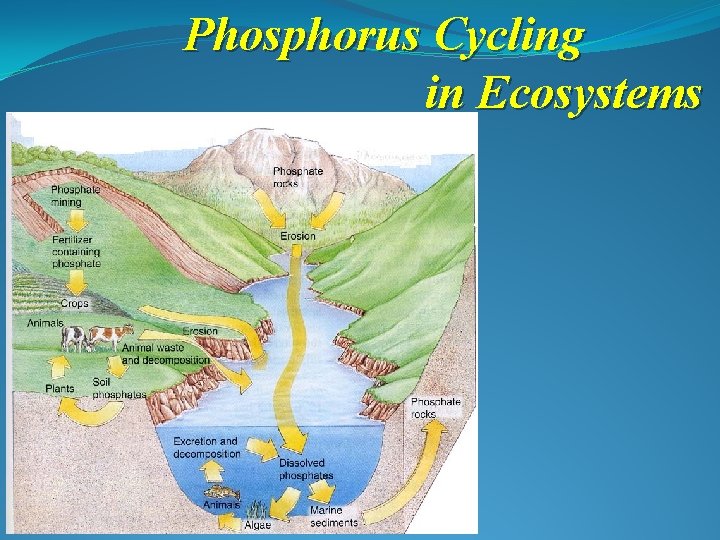
Phosphorus Cycling in Ecosystems

Bioaccumulation and Biomagnification Bioconcentration factor
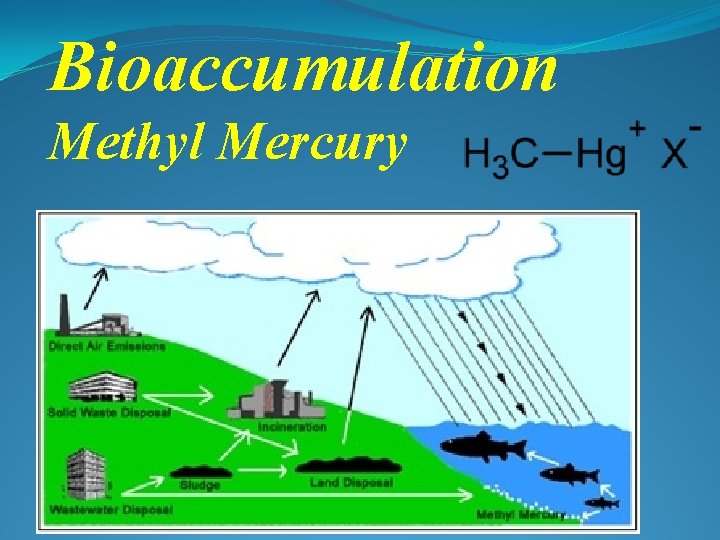
Bioaccumulation Methyl Mercury

Biomagnification DDT Hg

Bioaccumulation Bioconcentration factor BCF = C(in the organism) C(water)
![Regression Equations for Estimating BCF for Varieties of Fish log[BCF] = 0. 76 · Regression Equations for Estimating BCF for Varieties of Fish log[BCF] = 0. 76 ·](http://slidetodoc.com/presentation_image/57a2b978842b23efe1d30dde0bb04b73/image-76.jpg)
Regression Equations for Estimating BCF for Varieties of Fish log[BCF] = 0. 76 · log[Kow] - 0. 23 Rainbow Trout, Moskitofish, Bluegill sunfish log[BCF] = 2. 791 - log[S] S – water solubility (ppm)* Brook Trout, Carp, Rainbow Trout, Fathead Minnow log[BCF] = log[Kow] - 1. 32 log[BCF] = 1. 119 · log[Kow] - 1. 579 *ppm – parts per million Various

Kinetics of Biotransformation Ø A simple biodegradation model is one in which microorganisms are in contact with water containing a dissolved organic chemical that serves as the energy substrate. Ø Because chemical uptake of the organic chemical into a cell of the microorgamism is followed by enzymatic biotransformation, rates of biodegradation and uptake rates are equivalent (steady-state)

Kinetics of Biotransformation Michaelis-Menten enzyme kinetics where: V is the rate of chemical uptake per cell [M/(cell time)] Vmax is the maximum possible chemical uptake rate C is the concentration of dissolved chemical [M/L 3] Ks is the 0. 5 saturation constant [M/L 3] [C]
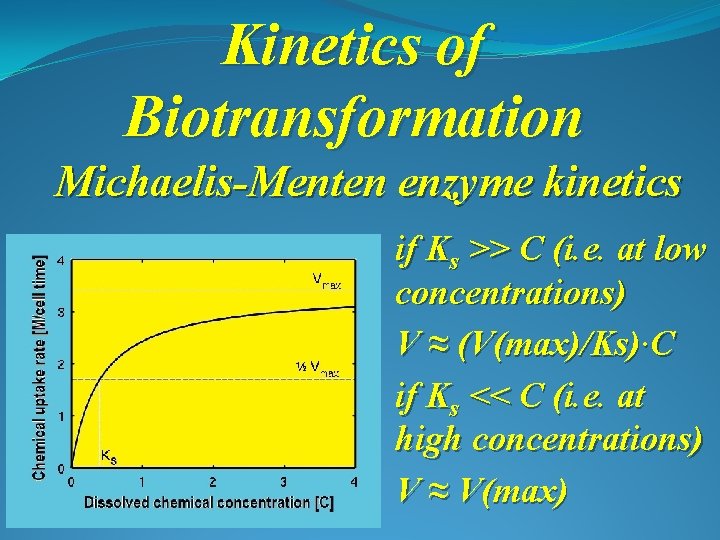
Kinetics of Biotransformation Michaelis-Menten enzyme kinetics if Ks >> C (i. e. at low concentrations) V ≈ (V(max)/Ks)∙C if Ks << C (i. e. at high concentrations) V ≈ V(max)
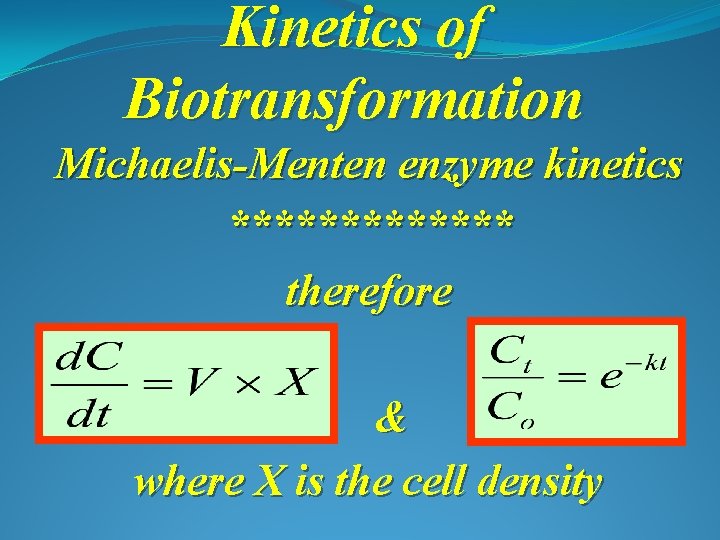
Kinetics of Biotransformation Michaelis-Menten enzyme kinetics ******* therefore & where X is the cell density

Kinetics of Biotransformation Michaelis-Menten enzyme kinetics and in the same way & where X is the cell density and μ is specific growth rate
![Kinetics of Biotransformation Examples of aerobic degradation rate constants COMPOUND ANTHRACENE RATE CONSTANT [day-1] Kinetics of Biotransformation Examples of aerobic degradation rate constants COMPOUND ANTHRACENE RATE CONSTANT [day-1]](http://slidetodoc.com/presentation_image/57a2b978842b23efe1d30dde0bb04b73/image-82.jpg)
Kinetics of Biotransformation Examples of aerobic degradation rate constants COMPOUND ANTHRACENE RATE CONSTANT [day-1] 0. 007 -0. 055 ATRAZINE (N-PHOSPHORYLATED) 0. 22 BENZENE 0. 11 CHLOROBENZENE PARATHION 0. 0045 <0. 00016 PHENOL 0. 079 2, 4, 5 -TETRACHLOROETHYLENE 0. 001 1, 4, 5 -TRICHLOROPHENOACETIC ACID 0. 0005
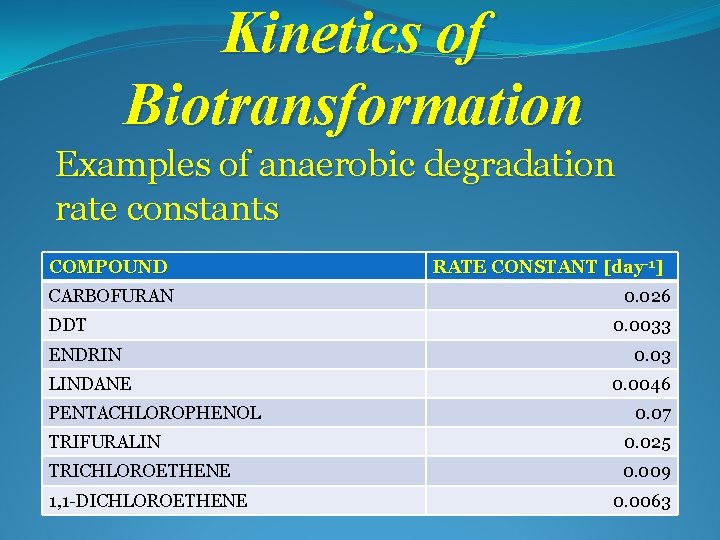
Kinetics of Biotransformation Examples of anaerobic degradation rate constants COMPOUND CARBOFURAN DDT ENDRIN LINDANE PENTACHLOROPHENOL RATE CONSTANT [day-1] 0. 026 0. 0033 0. 0046 0. 07 TRIFURALIN 0. 025 TRICHLOROETHENE 0. 009 1, 1 -DICHLOROETHENE 0. 0063

Abiotic chemical transformations Photodegradation – the nature of light *********** Light is a type of electromagnetic waves ν = c/λ where ν is wave frequency [sec-1] c is speed of all electromagnetic waves (light included) = 3. 0∙ 108 m/sec λ is the wave length [m]
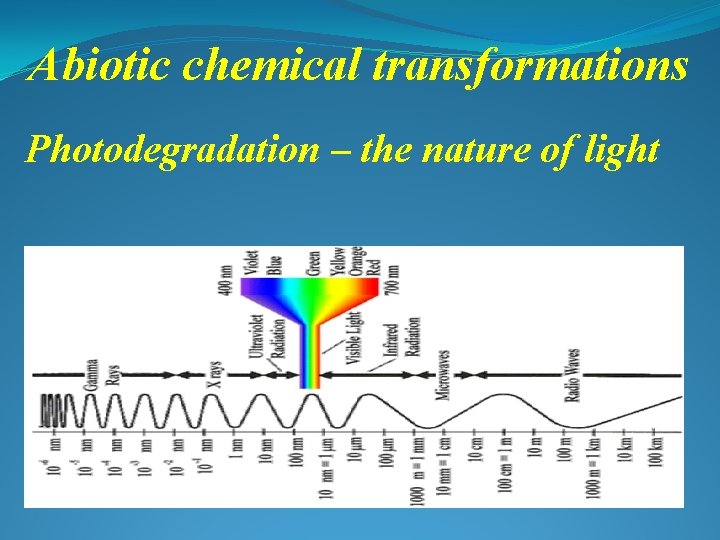
Abiotic chemical transformations Photodegradation – the nature of light
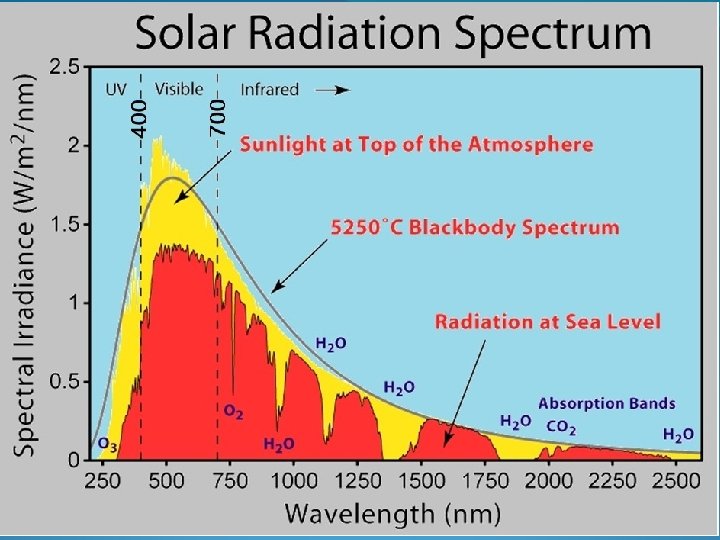

Abiotic chemical transformations Photodegradation – the nature of light Solar radiation spectrum 100 nm < λ < 3000 nm Visible light 400 nm < λ < 700 nm λ < 400 nm λ > 700 nm ultraviolet infrared ozone partial absorbtion water

Abiotic chemical transformations Photodegradation – the nature of light Light penetration into surface waters Iz = I o -ηz e where I is the light intensity [watts/m 2] η is the extinction coefficient [m-1]

Abiotic chemical transformations Photodegradation (photodissociation, photolysis, or photodecomposition) Ø Photodegradation is decomposition of a compound by radiant energy ØIt is a chemical reaction in which a chemical compound is broken down by photons. ØPhotodegradation includes photodissociation, the breakup of molecules into smaller pieces by photons

Abiotic chemical transformations Photolysis of water ØThe bulk of our Earth's oxygen does not come from photosynthesis, but from photodissociation - that is, the breaking down of water into its component parts by ultraviolet light. Ø H 2 O + 2 photons (light) → 2 e- + 2 H+ + ½O 2 ØThis happens to water vapor in the upper atmosphere. The lighter hydrogen escapes into space and is lost, the oxygen - being heavier settles to the Earth.

Abiotic chemical transformations Photodegradation – the nature of light Solar radiation energy E = h∙ν where E is the photon energy h is Plank’s constant = 6. 6∙ 10 -34 J∙sec and ν is wave frequency [sec-1]

Abiotic chemical transformations Photodegradation (photodissociation, photolysis, or photodecomposition) ØThe photochemical transformation of a molecule into lower molecular weight fragments, usually in an oxidation proces. This term is widely used in the destruction (oxidation) of pollutants by UV-based processes.

Abiotic chemical transformations Photodegradation (photodissociation, photolysis, or photodecomposition) ØIt is not limited to visible light. ØAny photon with sufficient energy can affect the chemical bonds of a chemical compound. ØSince a photon's energy is inversely proportional to its wavelength, electromagnetic waves with the energy of visible light or higher, such as ultraviolet light, x-rays and gamma rays are usually involved in such reactions.

Abiotic chemical transformations Direct and indirect photodissociation of organic compounds in water ØDirect photolysis is a process in which molecules get excited by the absorption of a photon, and that results in a chemical reaction, usually oxidation. ØThe direct effects of UV irradiation include transformation of organic compounds into other substances, breaking of chemical bonds, or even complete degradation of organic substances.

Abiotic chemical transformations Direct and indirect photodissociation of organic compounds in water ØAlso, UV radiation causes dissociation of oxidizing compounds and formation of highly reactive radicals that are capable of degrading organic pollutants. ØIndirect photolysis of substances occurs through a reaction with OH-radicals, ozone or NO 3 -; these three chemicals are considered the most important photo-oxidizing agents present in the environment.
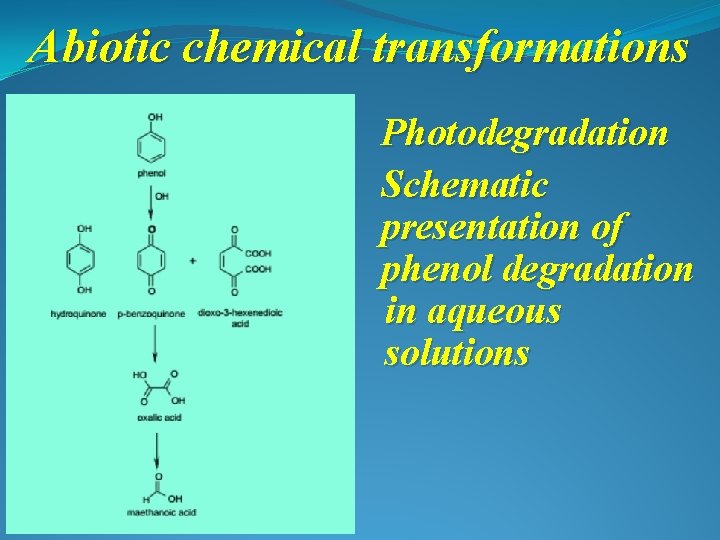
Abiotic chemical transformations Photodegradation Schematic presentation of phenol degradation in aqueous solutions
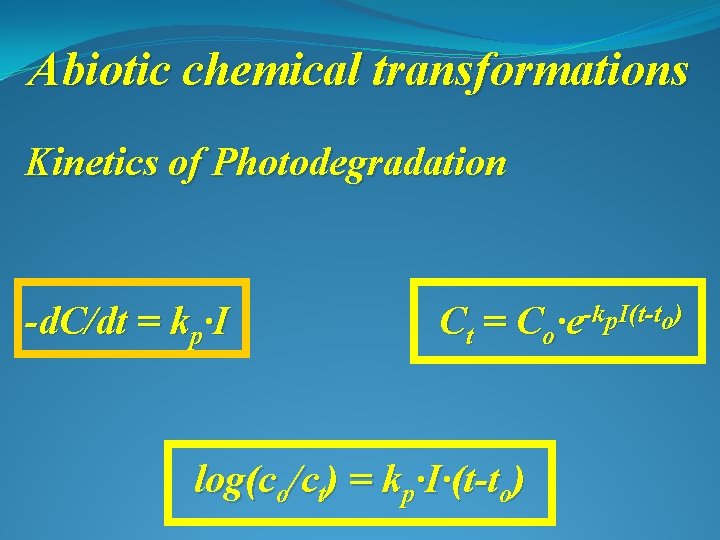
Abiotic chemical transformations Kinetics of Photodegradation -d. C/dt = kp∙I Ct = Co∙e-kp. I(t-to) log(co/ct) = kp∙I∙(t-to)

Abiotic chemical transformations Kinetics of Photodegradation Examples of Half-Lives for Degradation via Direct Photolysis Pesticides λ (nm) t½ PAHs λ (nm) t½ Parathion 5 10 days Anthracene 366 0. 73 hrs Mirex 5 1 year Benz[a]Anthracene 5 3. 3 hrs Methyl Parathion 5 30 days Benzo[a]Pyrene 5 1 hr Methoxychlor 5 29 days Chrysene 313 4. 4 hrs Malathion 5 15 hrs Fluoranthene 313 21 hrs DDE 5 22 hrs Naphtalene 313 70 hrs 2, 4 -D, Methyl ester 5 62 days Phenanthrene 313 8. 4 hrs 2, 4 -D, Butoxyethyl ester 5 12 days 313, 366 0. 68 hrs Carbaryl 5 50 hrs Pyrene
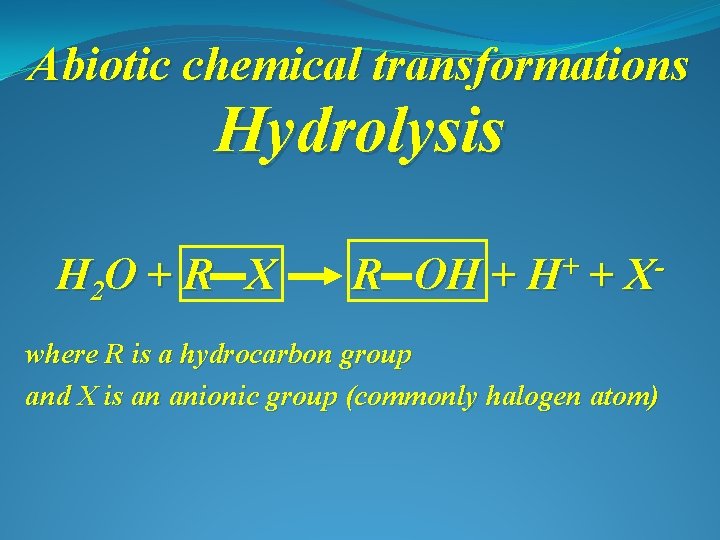
Abiotic chemical transformations Hydrolysis H 2 O + R X R OH + + H + X where R is a hydrocarbon group and X is an anionic group (commonly halogen atom)

Abiotic chemical transformations Examples of Hydrolysis

Rates of Hydrolysis d. C/dt = -k. H 2 O × C × [H 2 O] = -k’n × C Ct = Co × e-k’n t
- Slides: 101