Conservation of Mass and Reactions Objective 4 TEK

Conservation of Mass and Reactions

Objective 4 TEK 8 The student knows tat changes in matter affect everyday life. (C ) The student is expected to investigate and identify the law of conservation of mass.
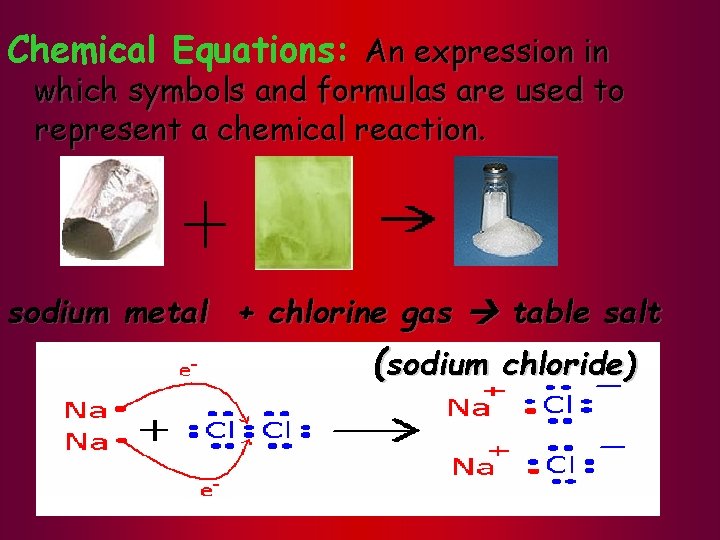
Chemical Equations: An expression in which symbols and formulas are used to represent a chemical reaction. sodium metal + chlorine gas table salt (sodium chloride)

The meaning of chemical equations A mathematical equation: x+2 x=3 x A chemical equation identifies the starting and finishing chemical as reactants and products: reactants products Example: Formation of water 2 H 2 + O 2 2 H 20 A chemical equation is balanced when it reflects the conservation of mass and charge.

Law of Conservation of Mass is neither created nor destroyed during chemical or physical reactions. Antoine Lavoisier Total mass of reactants = Total mass of products

The Law of conservation of mass states that matter cannot be created or destroyed in any chemical reaction The bonds between atoms in the reactants are rearranged to form new compounds, but none of the atoms disappear, and no new atoms are formed. So: Chemical equations must be balanced, meaning the numbers and kinds of atoms must be the same on both sides of the reaction arrow. The numbers placed in front of formulas to balance equations are called coefficients, and they multiply all the atoms in the chemical formula.

Balancing Chemical Equations The following five steps can be used as a guide to balance chemical equations. Balance this chemical reaction. Sulfuric Acid reacts with sodium hydroxide to yield sodium sulfate and water Step 1: Write an unbalanced equation, using correct formulas for all reactants and products. H 2 SO 4 + Na. OH Na 2 SO 4 + H 2 O
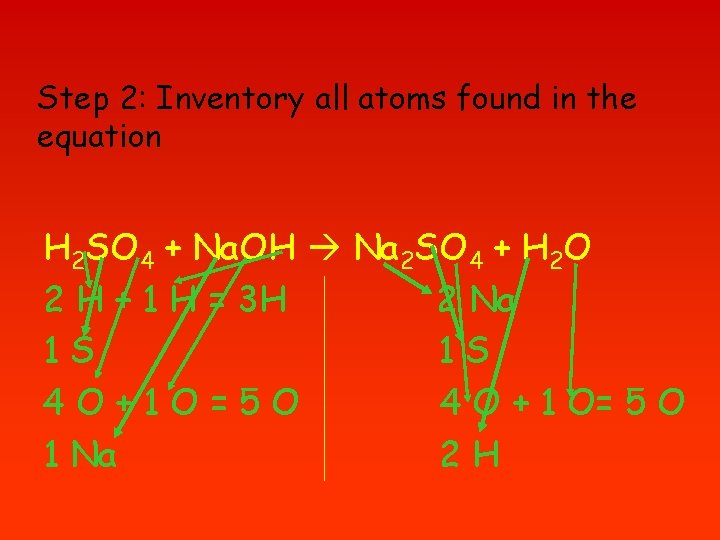
Step 2: Inventory all atoms found in the equation H 2 SO 4 + Na. OH Na 2 SO 4 + H 2 O 2 H + 1 H = 3 H 2 Na 1 S 1 S 4 O+1 O=5 O 4 O + 1 O= 5 O 1 Na 2 H

Step 3: Compare the number of each atom on each side of the equation. Add coefficients to balance the number of atoms. Remember that adding a coefficient affects all elements in the compound. H 2 SO 4 + 2 Na. OH Na 2 SO 4 + 2 H 2 O 2 H + 1 H = 3 H 4 H 2 Na 6 O 1 S 1 S Equal 4 O+1 O=5 O Equal 4 O + 1 O= 5 O 6 O 1 Na 2 H 4 H

Step 4: Check the equation to make sure the numbers and kinds of atoms on both sides of the equation are same. H 2 SO 4 + 2 Na. OH Na 2 SO 4 + 2 H 2 O 2 H + 2 H = 4 H 2 Na 1 S 1 S 4 O+2 O=6 O 4 O + 2 O= 6 O 2 Na 4 H

Step 5: Make sure the coefficients are reduced to their lowest wholenumber value (ok here). H 2 SO 4 + 2 Na. OH Na 2 SO 4 + 2 H 2 O 1: 2: 1: 2

Balance the following equations. 1. 2. 3. 4. 5. KCl. O 3 → KCl + O 2 P 4 + O 2 → P 2 O 5 Al 2 O 3 → Al + O 2 Al 2(SO 4)3 + Ca(OH)2 → Al(OH)3 + Ca. SO 4 Ca(OH)2 + H 3 PO 4 → Ca 3(PO 4)2 + H 2 O

Answers Balance the following equations. 1. 2. 3. 4. 5. 2 KCl. O 3 → 2 KCl + 3 O 2 P 4 + 5 O 2 → 2 P 2 O 5 2 Al 2 O 3 → 4 Al + 3 O 2 Al 2(SO 4)3 +3 Ca(OH)2→ 2 Al(OH)3 + 3 Ca. SO 4 3 Ca(OH)2 + 2 H 3 PO 4 → Ca 3(PO 4)2 + 6 H 2 O

What do coefficients mean? • 1. They indicate the number of particles of atoms, molecules, and formula units found in the reaction 2 H 2 + O 2 2 H 2 O Indicates that 2 molecules of hydrogen react with 1 molecule of oxygen to produce 2 molecules of water. • 2. They are used to determine the amount of reactants and products.
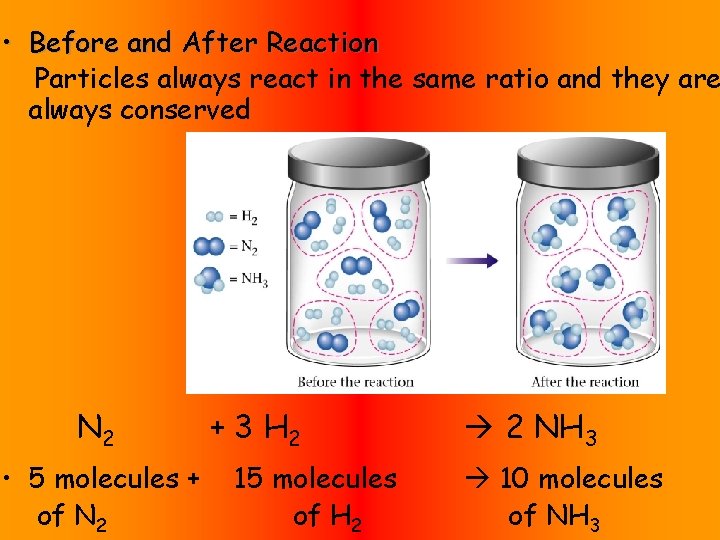
• Before and After Reaction Particles always react in the same ratio and they are always conserved N 2 • 5 molecules + of N 2 + 3 H 2 15 molecules of H 2 2 NH 3 10 molecules of NH 3

Water from a Camel fat Camels store the fat, tristearin (C 57 H 110 O 6), in the hump. As well as being a source of energy, the fat is a source of water, because when it is used the reaction takes place. Given the following information, what mass of water can be made from 1000 g of fat? 2 C 57 H 110 O 6(s) + 163 O 2(g) 114 CO 2(g) + 110 H 2 O(l) 1000 g C 57 H 110 O 6 + 2930 g O 2 2817 g CO 2 + ? H 2 O

Remember that mass is conserved. Therefore the mass of reactants = mass of products 1000 g C 57 H 110 O 6 + 2930 g O 2 2817 g CO 2 + ? H 2 O Mass of reactants 1000 g + 2930 g = 3930 g = Mass of products = 3930 g – 2817 g = 1113 g H 2 O

Water in Space In the space shuttle, the CO 2 that the crew exhales is removed from the air by a reaction within canisters of lithium hydroxide. On average, each astronaut exhales about 880 g of CO 2 daily. What mass of water will be produced when this amount of CO 2 reacts with 956 g of Li. OH according to the following equation? CO 2(g) + 2 Li. OH(s) Li 2 CO 3(aq) + H 2 O(l) 880 g. CO 2 + 956 g Li. OH 1476 g Li 2 CO 3 + ? H 2 O

Remember that mass is conserved. Therefore the mass of reactants = mass of products 880 g. CO 2 + 956 g Li. OH 1476 g Li 2 CO 3 + ? H 2 O Mass of reactants 880 g + 956 g = 1836 g = Mass of products = 1836 g - 1476 g = 360 g H 2 O

This demonstration is the combustion of diethyl ether in air. If 33. 8 g of diethyl ether is added to the balloon, how many grams of carbon dioxide are given off? C 2 H 5 OC 2 H 5 + 6 O 2 4 CO 2 + 5 H 2 O 33. 8 g C 2 H 5 OC 2 H 5 + 87. 7 g O 2 ? g CO 2 + 41. 2 g H 2 O = 80. 3 g CO 2

If this experiment was done in your classroom, why would it be difficult to prove the law of conservation of mass? C 2 H 5 OC 2 H 5 (l)+ 6 O 2 (g) 4 CO 2 (g) + 5 H 2 O (g) CO 2 and H 2 O are gases and they would move throughout the room.
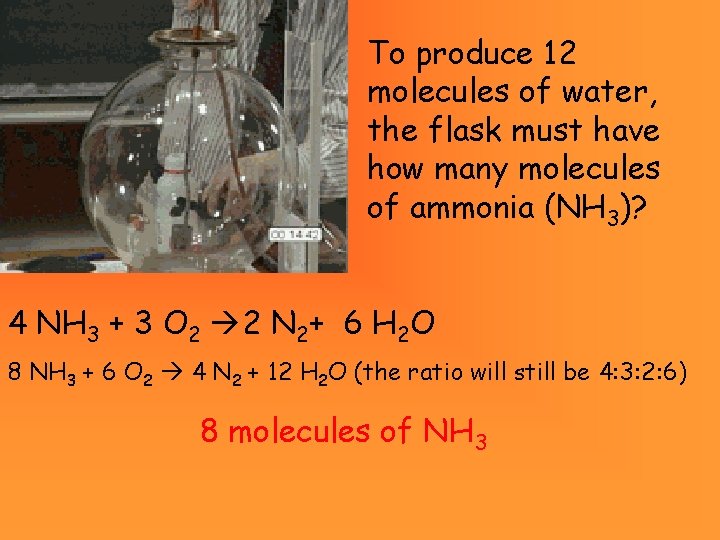
To produce 12 molecules of water, the flask must have how many molecules of ammonia (NH 3)? 4 NH 3 + 3 O 2 2 N 2+ 6 H 2 O 8 NH 3 + 6 O 2 4 N 2 + 12 H 2 O (the ratio will still be 4: 3: 2: 6) 8 molecules of NH 3
Methane gas is burned in excess oxygen to produce carbon dioxide & water. If 25. 0 grams of methane is burned in 100. g of oxygen (O 2) and 68. 8 g CO 2 are produced, how many grams of water is produced? CH 4 (g) + 2 O 2 (g) 2 H 2 O (l) + CO 2 (g) 25. 0 g + 100. 0 g = 68. 8 g + ? 56. 2 g H 2 O

Phosphorus reacts with oxygen to produce diphosphorus pentoxide according to the equation: P 4 (s) + 5 O 2 (g) 2 P 2 O 5 How many particles of phosphorus must be present to produce 30 molecules of P 2 O 5? 15 P 4 + 75 O 2 30 P 2 O 5 (ratio is still 1: 5: 2) 15 particles of P 4
- Slides: 24