Conservation of mass 91515 Brain Teaser Girls Like

Conservation of mass 9/15/15

Brain Teaser • Girls Like, Boys Use • What 8 -letter word describes something girls like, boys use and parents are afraid of yet remember fondly? • Hint: It has these letters: macroen
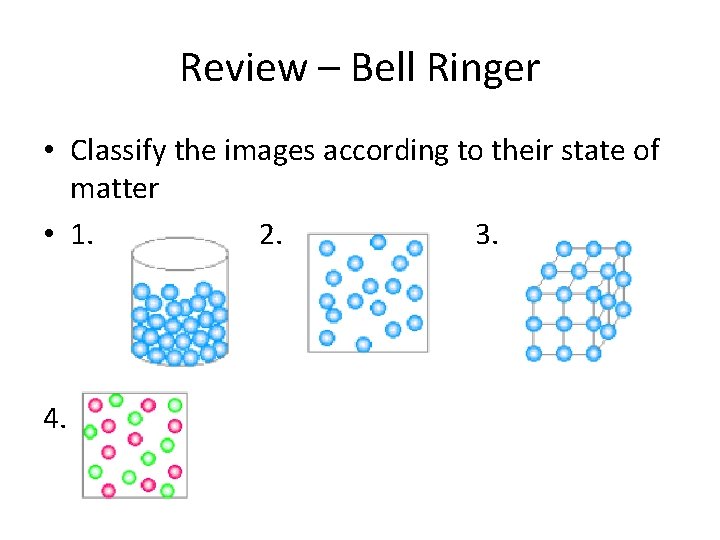
Review – Bell Ringer • Classify the images according to their state of matter • 1. 2. 3. 4.

Evidence of a physical change • A chemical reaction always produces a change in properties – Color change – Formation of a gas – Burning of wood (releases heat - energy) – Smell changes (odor) – Temperature changes

The Law of Conservation of mass or matter (don’t write this yet) • Why is it a law and not a theory? • After scientists started measuring the mass before and after chemical reactions, they noticed that the total mass involved in the reaction stayed the same (took hundreds of years of data to see that the observation was consistent every time!) • Is it an explanation? • No, its an observation! Therefore it is a law!

The Law • The law of conservation of mass states that mass is neither created or destroyed in any process – it is conserved. Mass of reactants = mass of products Mass. R = Mass. P
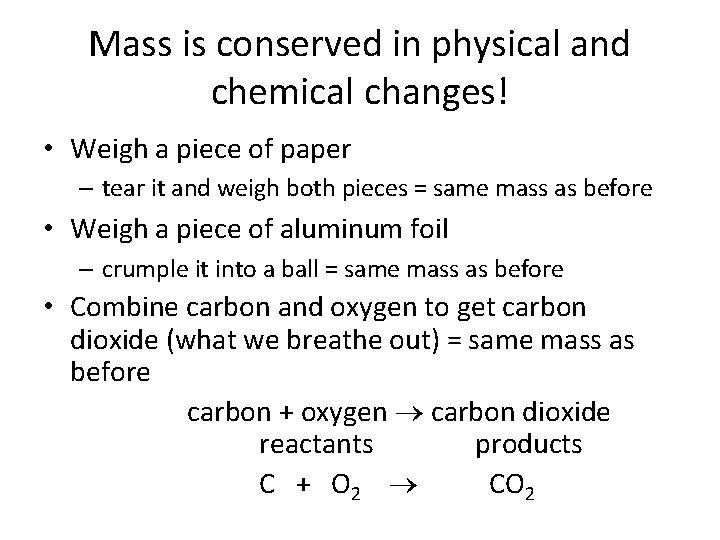
Mass is conserved in physical and chemical changes! • Weigh a piece of paper – tear it and weigh both pieces = same mass as before • Weigh a piece of aluminum foil – crumple it into a ball = same mass as before • Combine carbon and oxygen to get carbon dioxide (what we breathe out) = same mass as before carbon + oxygen carbon dioxide reactants products C + O 2 CO 2

Conservation of mass videos • Conservation videos – https: //www. youtube. com/watch? v=2 S 6 e 11 NBwi w (Block 4 –Todd Ramsey) – Chemical reaction conservation demo: https: //www. youtube. com/watch? v=Wwmsy 4 hu. Z Q 0 (B 1 & B 4 – Mr. King) – Phase change conservation demo (: https: //www. youtube. com/watch? v=x 9 b. Jzf 2 t. Nh. Y

Ø From your homework: Ø 178. 8 g H 2 O (liq) (I do) practice 20. 0 g H (gas) + ____ g O (gas) ØWhole is 178. 8 g Ø 2 parts make up the whole (Hydrogen and Oxygen) ØIf we know 1 part of the whole, we can calculate the other part of the whole! Ø 178. 8 = 20 – x Ø 178. 8 – 20 = x Ø 158. 8 = x = 159 g (3 SF) ØYou just did a basic conservation of mass problem!
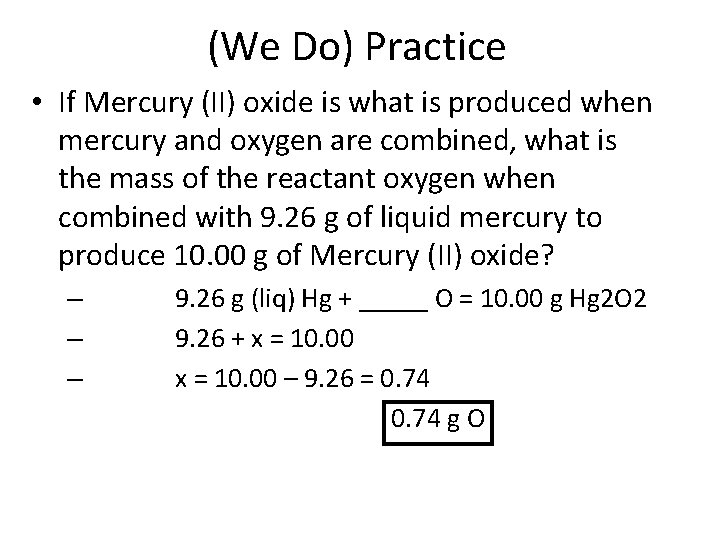
(We Do) Practice • If Mercury (II) oxide is what is produced when mercury and oxygen are combined, what is the mass of the reactant oxygen when combined with 9. 26 g of liquid mercury to produce 10. 00 g of Mercury (II) oxide? – – – 9. 26 g (liq) Hg + _____ O = 10. 00 g Hg 2 O 2 9. 26 + x = 10. 00 – 9. 26 = 0. 74 g O

(You Do) Practice • A 28. 0 g sample of nitrogen gas combines completely with 6. 0 g of hydrogen gas to form ammonia. What is the mass of the ammonia formed? • 28. 0 g + 6. 0 g = 34 g of ammonia

Practice with your partner ( 7 minutes) • Page 65 • # 12 • A&B

Pg 65 # 12 A • 22. 99 g Na + 35. 45 g Cl = _____ Na. Cl • 22. 99 + 35. 45 = 58. 44 g Na. Cl

Pg. 65 # 12 B • 12. 2 g X + ____ Y = 78. 9 g XY • 12. 2 + x = 78. 9 • X = 78. 9 – 12. 2 = 66. 7 g Y

Review • The law of conservation of mass states that created or ____ destroyed in any mass is neither ____ process – it is _____. conserved • Mass of Reactants = _________ Mass of Products • Be prepared to work some problems tomorrow as a review!

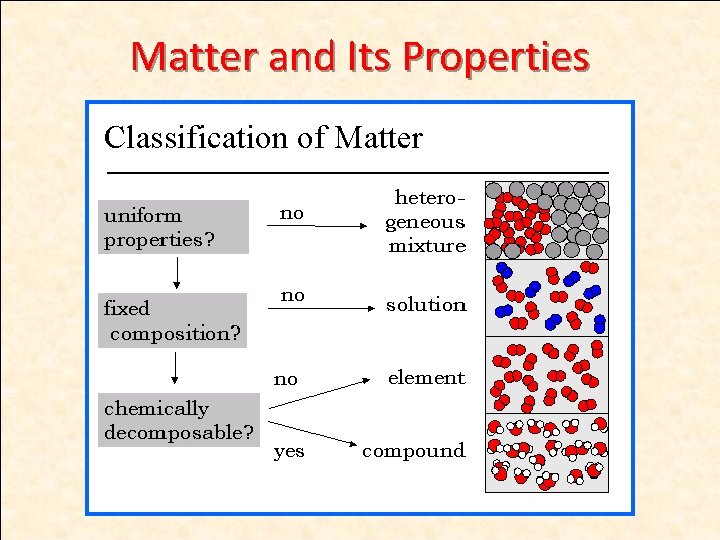
Types of Matter 1. Pure substances (homogeneous) a) Elements b) Compounds 2. Mixtures (homogeneous or heterogeneous)

Pure Substances • Matter that is uniform and unchanging in composition – Salt and water – But NOT ocean water

Elements • Simplest type of matter - one type of atom (homogeneous) • Elements arranged according to increasing atomic mass and recurring properties in PERIODIC TABLE OF THE ELEMENTS • Each element has unique one or two letter symbol

Elements • 91 elements occur naturally • Example: Copper (Cu)

Element • Each element has a unique chemical name and symbol. • The first letter is always capitalized and the remaining letters are lowercase. • Ex: – Carbon – Cu – Copper – Cs – Cesium – Cr – Chromium – Co - Cobalt
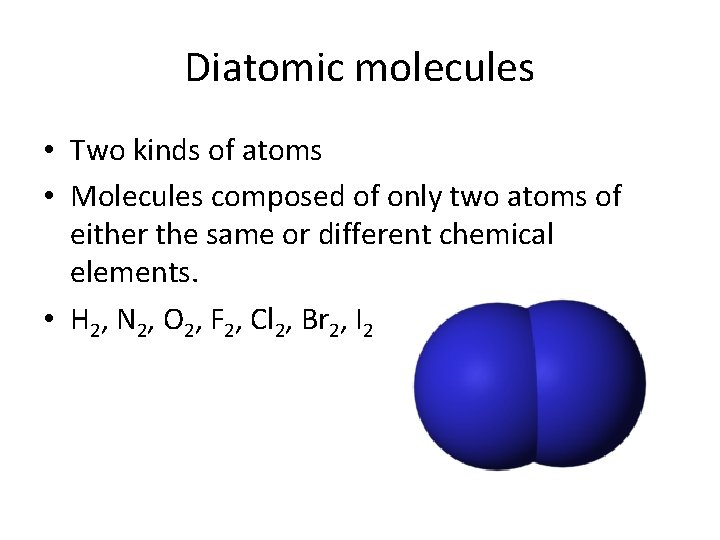
Diatomic molecules • Two kinds of atoms • Molecules composed of only two atoms of either the same or different chemical elements. • H 2, N 2, O 2, F 2, Cl 2, Br 2, I 2
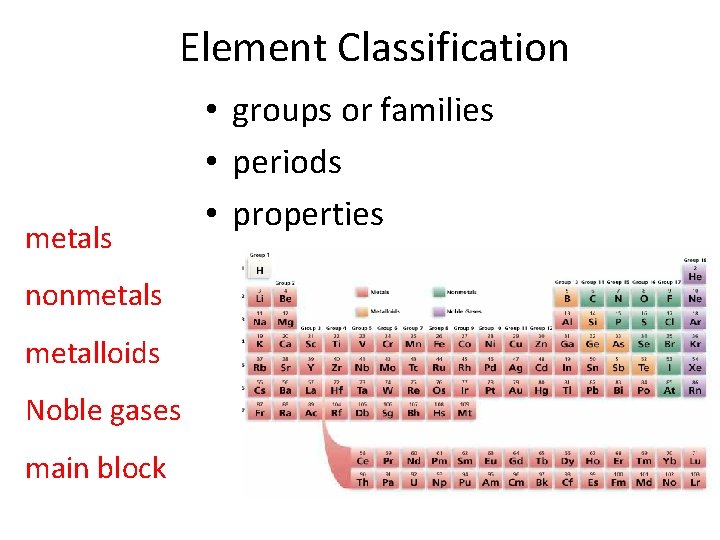
Element Classification metals nonmetals metalloids Noble gases main block • groups or families • periods • properties

Pure substances • Compounds – Salt (always composed of Na and Cl) – Water (always composed of H and O)

Compounds • Chemical combinations of two or more elements • Most familiar substances are compounds • Can be broken down into simpler substances by chemical reactions.

Compounds • Table salt isn’t an element! • It’s a compound (a substance composed of atoms of two or more different elements that are chemically combined) • EX: Na. Cl • They combine ONLY when the compound is more stable than the individual atoms. • Substances go through a chemical change to form a compound, and the different elements seem to lose their individual characteristics. • Na (explosive alone) + Cl (poisonous gas alone) = Na. Cl (safe to eat when bonded together)

Law of definite proportions • Elements that make up compounds combine in definite proportions by mass. • Percent by mass (%) = Mass of element x 100 Mass of compound

Example • 500. 0 g of sucrose isolated from a sample of sugar cane • Sucrose has C, H, O • What is the percent by mass of carbon in the sucrose molecule if the mass is 210. 5 g C? • 210. 5 g C x 100 = 42. 10 % Carbon 500. 0 g Sucrose

Example • 500. 0 g of sucrose isolated from a sample of sugar cane • Sucrose has C, H, O • What is the percent by mass of Hydrogen in the sucrose molecule if the mass is 32. 4 g H? • 32. 4 g H x 100 = 6. 48 % Hydrogen 500. 0 g Sucrose

Example • 500. 0 g of sucrose isolated from a sample of sugar cane • Sucrose has C, H, O • What is the percent by mass of Oxygen in the sucrose molecule if the mass is 257. 1 g O? • 257. 1 g O x 100 = 51. 4 % Oxygen 500. 0 g Sucrose

You try • Pg. 76 20 -24

Mixtures (heterogeneous)

Remember that compounds are when elements combine with other elements through CHEMICAL BONDING! • Covalent bonding • Ionic Bonding Example: A baked cake, table salt, water molecules Compound

Mixture Elements are NOT BONDING CHEMICALLY in mixtures. • They just mix together, retaining their original properties. • They can be physically separated after they are combined. Examples: 1. A salad in a bowl where the different vegetables can be put together in a bowl, but can be easily separated. 2. The blue sand salt we combined in lab. 3. Salt dissolved in water. Can pour the mixture through a filter and remove the salt from the water.

Homogeneous Mixtures • two or more kinds of matter with uniform composition • air, vinegar, salt water, and Sprite • liquid homogeneous mixtures - SOLUTIONS • solid homogeneous mixtures of metals - ALLOYS

Heterogeneous Mixtures • combination of two or more kinds of matter with different parts or properties; composition is NOT uniform • blood, whole milk, granite, and chocolate chip cookies

Separation of Matter • Elements – cannot be broken down • Compounds – can be broken down by chemical methods • Mixtures – can be separated by physical means • Separation depends on properties of the matter

Separation Techniques • • • Distillation Chromatography Density Centrifuge Filtration

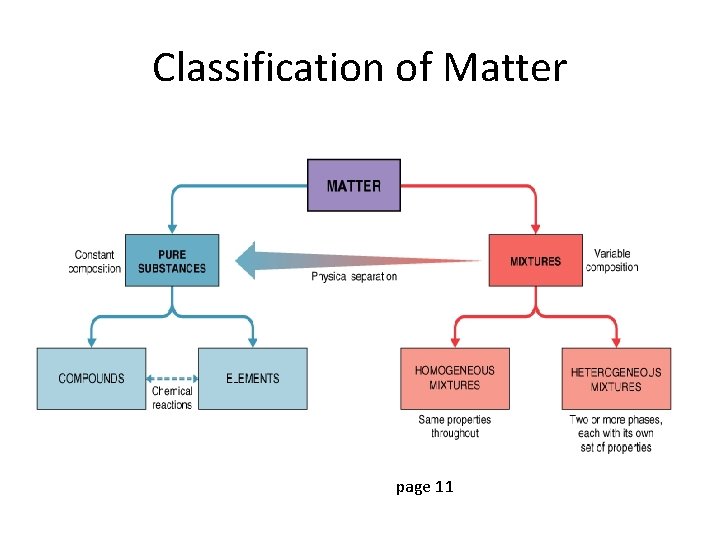
Classification of Matter page 11
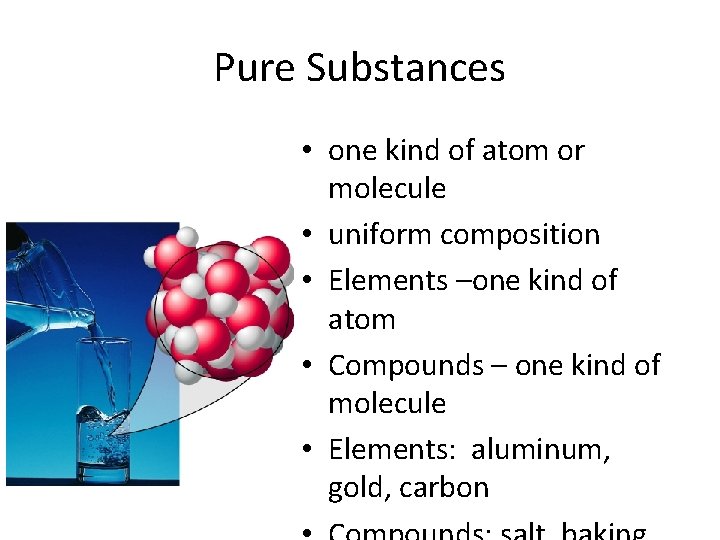
Pure Substances • one kind of atom or molecule • uniform composition • Elements –one kind of atom • Compounds – one kind of molecule • Elements: aluminum, gold, carbon
Matter and Its Properties
- Slides: 41