Connective Tissue Metabolism Jana Novotna Connective Tissue Ubiquitous

Connective Tissue Metabolism Jana Novotna

Connective Tissue Ubiquitous, serves universal functions. 1. Primarily for support and for binding tissue together 2. Permits free diffusion of nutrients and metabolites 3. Immunological protection

Composition Connestive tissue cells: • Fibroblasts • Adipocytes • Fixed macrophages • Mast cells • Lymphocytes • Pigment cells • Other cells Fibers • Collagen fibers • Reticular fibers • Elastic fibers Ground substance • Proteoglycans (GAG) • glycoproteins

Cells • Fibroblasts - the most common resident cells in connective tissue. Responsible for normal development and woud healing. • Fibroblasts from different regions display extensively differentiated patterns of gene expression which may guide differentiated patterns of tissue organization. • Resting fibroblasts retain the ability to become active as during healing after injury. • The substance of the scar is collagen • Closely related cell types – chondroblasts (cartillage) and osteoblast (bone)

Types of Connective Tissue • "Ordinary" connective tissue - generalized form of connective tissue with all of the basic components (cells, fibers, ground substance). • Ground substance is the background material within which all other connective tissue elements are embedded. • Ground substance consists mainly of water whose major role is to provide a route for communication and transport (by diffusion) between tissues. • This water is stabilized by a complex of glycosaminoglycans (GAGs), proteoglycans, and glycoproteins. • "Special" connective tissue - very highly differentiated and localized forms (sharing many common features) – bone, cartillage, lymphoid tissue, blood.

Types of Connective Tissue • Dense/loose connective tissue - depending on the proportion of fibers. • Dense connective tissue - high density of extracellular fibers, relatively smaller proportions of ground substance and cells. – Dense collagenous connective tissues - tendons, ligaments, organ sheaths (top priority is strength) – Dense elastic connective tissue (top priority is elasticity) – the wall of the aorta, the elastic ligament of the spine (ligamentum flavum) • Loose (areolar) connective tissue - lacks the massive fibrous reinforcement that characterizes dense connective tissue. • Relatively large proportion of ground substance, cells or both (adipose tissue, lymphoid tissue)
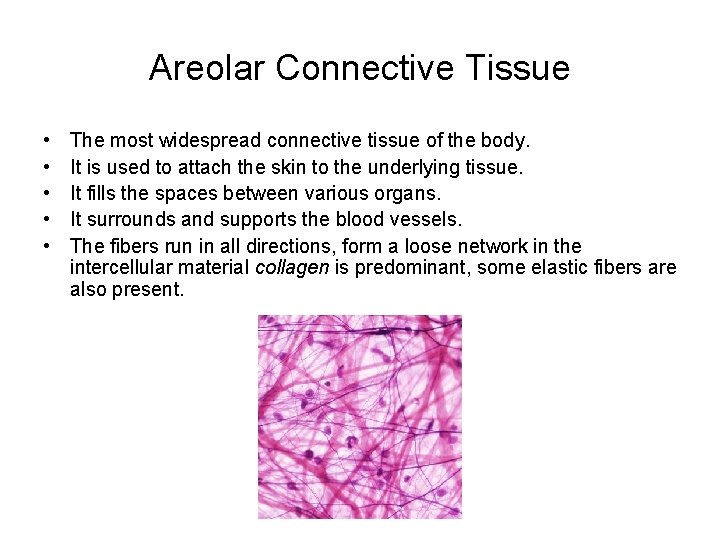
Areolar Connective Tissue • • • The most widespread connective tissue of the body. It is used to attach the skin to the underlying tissue. It fills the spaces between various organs. It surrounds and supports the blood vessels. The fibers run in all directions, form a loose network in the intercellular material collagen is predominant, some elastic fibers are also present.
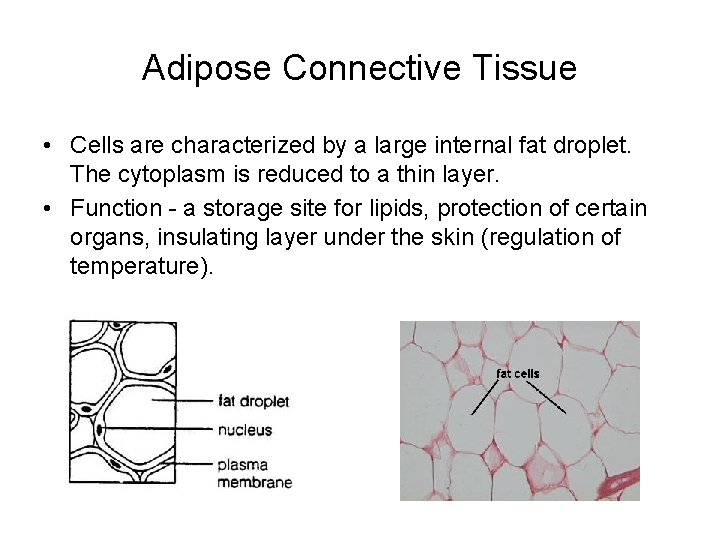
Adipose Connective Tissue • Cells are characterized by a large internal fat droplet. The cytoplasm is reduced to a thin layer. • Function - a storage site for lipids, protection of certain organs, insulating layer under the skin (regulation of temperature).
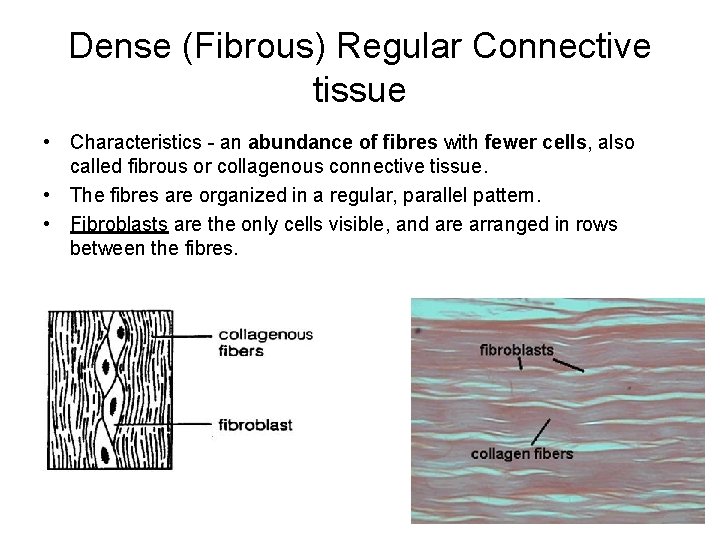
Dense (Fibrous) Regular Connective tissue • Characteristics - an abundance of fibres with fewer cells, also called fibrous or collagenous connective tissue. • The fibres are organized in a regular, parallel pattern. • Fibroblasts are the only cells visible, and are arranged in rows between the fibres.

Functions of Connective Tissue • Main functions: – – Transport Immunological defense Mechanical support Repair • Additional functions – Reserve energy store (fat) – Heat generation (brown fat) – Haemopoiesis
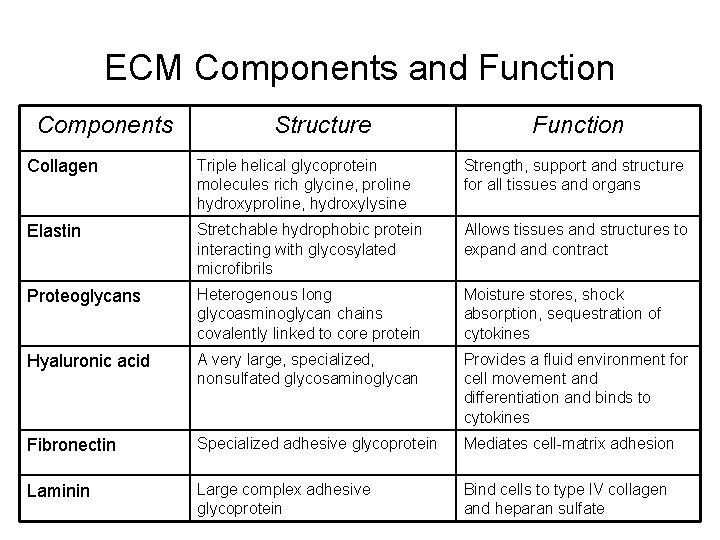
ECM Components and Function Components Structure Function Collagen Triple helical glycoprotein molecules rich glycine, proline hydroxyproline, hydroxylysine Strength, support and structure for all tissues and organs Elastin Stretchable hydrophobic protein interacting with glycosylated microfibrils Allows tissues and structures to expand contract Proteoglycans Heterogenous long glycoasminoglycan chains covalently linked to core protein Moisture stores, shock absorption, sequestration of cytokines Hyaluronic acid A very large, specialized, nonsulfated glycosaminoglycan Provides a fluid environment for cell movement and differentiation and binds to cytokines Fibronectin Specialized adhesive glycoprotein Mediates cell-matrix adhesion Laminin Large complex adhesive glycoprotein Bind cells to type IV collagen and heparan sulfate
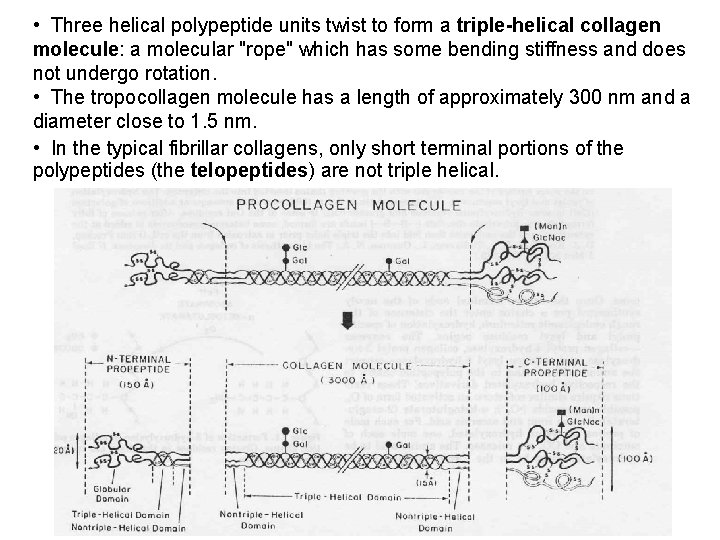
• Three helical polypeptide units twist to form a triple-helical collagen molecule: a molecular "rope" which has some bending stiffness and does not undergo rotation. • The tropocollagen molecule has a length of approximately 300 nm and a diameter close to 1. 5 nm. • In the typical fibrillar collagens, only short terminal portions of the polypeptides (the telopeptides) are not triple helical.
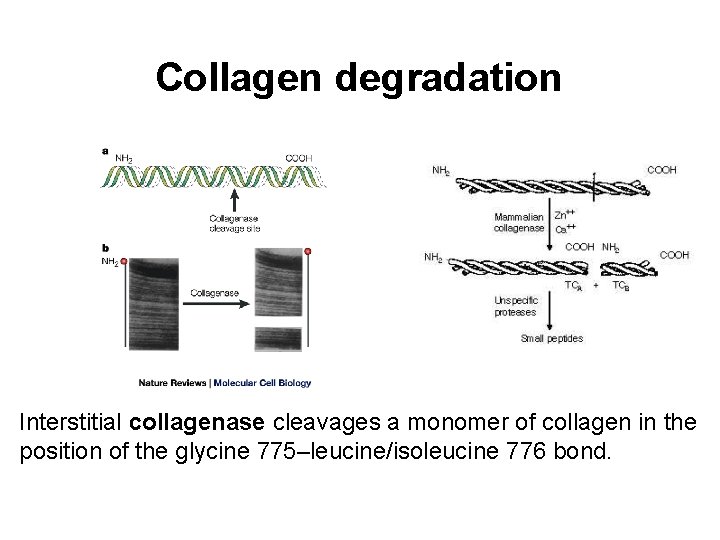
Collagen degradation Interstitial collagenase cleavages a monomer of collagen in the position of the glycine 775–leucine/isoleucine 776 bond.

Matrix Metalloproteinases (MMPs, matrixins) • Zn-dependent endopeptidases, proteolytic activity is preferentially targeted on extracellular matrix (ECM) and basal membranes. • It is involved in tissue degradation and remodeling of the ECM, both in physiological conditions, such as pathological. – embryogensis, morfogenesis, angiogenesis, healing etc. – Inflammatory diseases (arthritis), cardiovascular diseases, cancer • They have a broad substrate specificity and are present in the tissues of vertebrates, invertebrates and plants.

Matrix Metalloproteinases (MMPs, matrixins) • MMP´s belong to a larger superfamily of Zn-dependent metalloproteinases – metzincins (adamalysins, serralysins and astalysins). • Metzincins from other groups of enzymes differ, having highly conserved motif in the active center – three histidins with bound Zn 2+ • Activity of MMPs depends on the presence of Ca 2+ and neutral p. H. • To date - at least 26 known members of MMP´s, in humans are expressed 23 different MMP´s
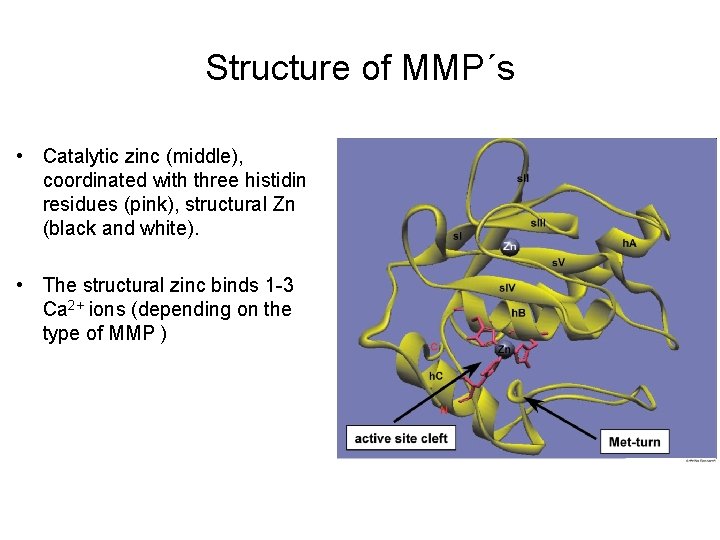
Structure of MMP´s • Catalytic zinc (middle), coordinated with three histidin residues (pink), structural Zn (black and white). • The structural zinc binds 1 -3 Ca 2+ ions (depending on the type of MMP )

Classification of MMP´s and their substrates (1) Collagenases Enzyme MMP# Substrate Collagenase-1 (fibroblast type) MMP-1 collagens (I-III, VIII, X), gelatine Collagenase-2 (neutrophile type) MMP-8 collagens (I-III, VIII, X), gelatine, fibronectin Collagenase-3 (breast cancer) MMP-13 collagens (I-IV, IX, X a XIV), gelatine, Collagenase-4 MMP-18 collagens I

Classification of MMP´s and their substrates (2) Gelatinases Enzyme MMP# Substrate Gelatinase A MMP-2 collagens (IV, V, VII, X a XIV), gelatine, fibronectin, osteonectin, Gelatinase B MMP-9 collagens (IV, V, VII, X a XIV), gelatine, elastin, fibronectin, osteonectin
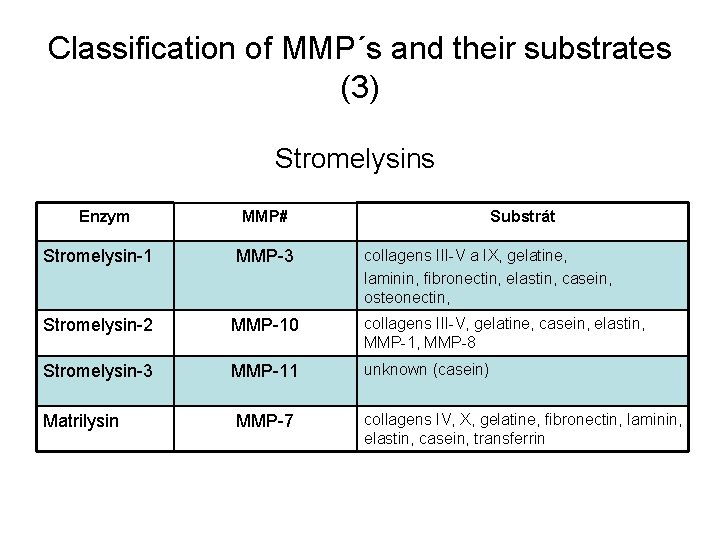
Classification of MMP´s and their substrates (3) Stromelysins Enzym MMP# Substrát collagens III-V a IX, gelatine, laminin, fibronectin, elastin, casein, osteonectin, Stromelysin-1 MMP-3 Stromelysin-2 MMP-10 collagens III-V, gelatine, casein, elastin, MMP-1, MMP-8 Stromelysin-3 MMP-11 unknown (casein) Matrilysin MMP-7 collagens IV, X, gelatine, fibronectin, laminin, elastin, casein, transferrin

Inhibitors of MMP´s – TIMP´s • MMP´s are inhibited by specific endogenous inhibitors – tissue inhibitors of matrix metalloproteinases. TIMP´s, inhibitors of metalloproteinases – IMPS, a 2 -makroglobulin • TIMP´s – group of four different proteases – TIMP-1, TIMP-2, TIMP 3 and TIMP-4 • Secreted either as soluble proteins (TIMP-1, TIMP-2 a TIMP-4), and TIMP-3 is bound to components of ECM • TIMP´s binds to the active site of enzyme by non-covalent bond. • It forms a complex with the catalytic zinc in a ratio of 1: 1

Proteoglycan degradation • • • Most glycoconjugates are degraded in lysosomes, and a portion of the liberated monosaccharides are reused for glycoconjugate synthesis Most of the endo- and exoglycosidases that degrade sugar chains and their modifications have p. H optima between 4. 0 and 5. 5. The endoglycosidases cleave internal glycosidic linkages of larger chains, yielding fragments that can then be degraded by exoglycosidases Exoglycosidases cleave the glycosidic linkage of terminal sugars from the nonreducing end of the chain. The exoglycosidases recognize only one (occasionally two) monosaccharide together with its anomeric linkage. Loss of a lysosomal enzyme leads to accumulation of non-degraded material in tissues – „lysosomal storage disease“ mucopolysaccharidoses

Normal and Pathological Response to Injury • Wound – disruption of normal anatomical structure and function • Healing – complex and dynamic process resulting in restoration of anatomical continuity and function. • Four basic response following an injury:
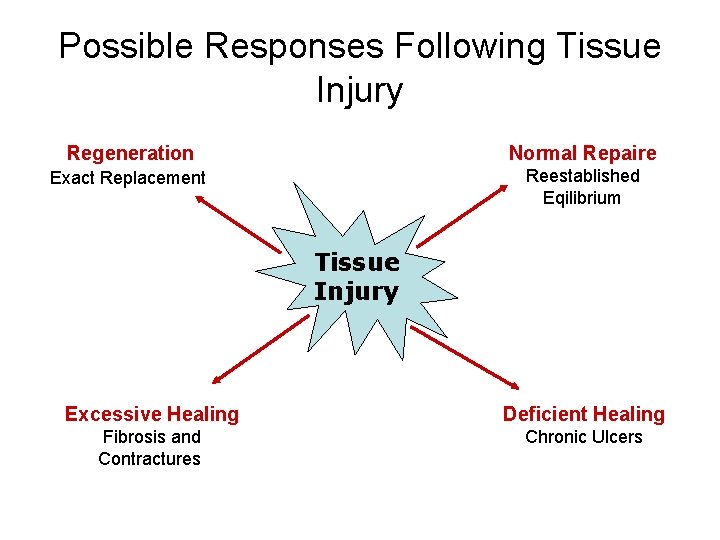
Possible Responses Following Tissue Injury Regeneration Normal Repaire Exact Replacement Reestablished Eqilibrium Tissue Injury Excessive Healing Deficient Healing Fibrosis and Contractures Chronic Ulcers

• Mechanisms of dermal wounds healing: 1. Connective tissue matrix deposition 2. Contraction 3. Epithelisation • • Primary Intension healing (simple wounds closed by sutures, tape or staples) – connective tissue matrix deposition (collagen, proteoglycans adhesive glycoproteins), formation of new ECM Wounds remain open – healing mainly by contraction (complex interaction between contractile fibroblasts – „myofibroblasts“ and matrix components).
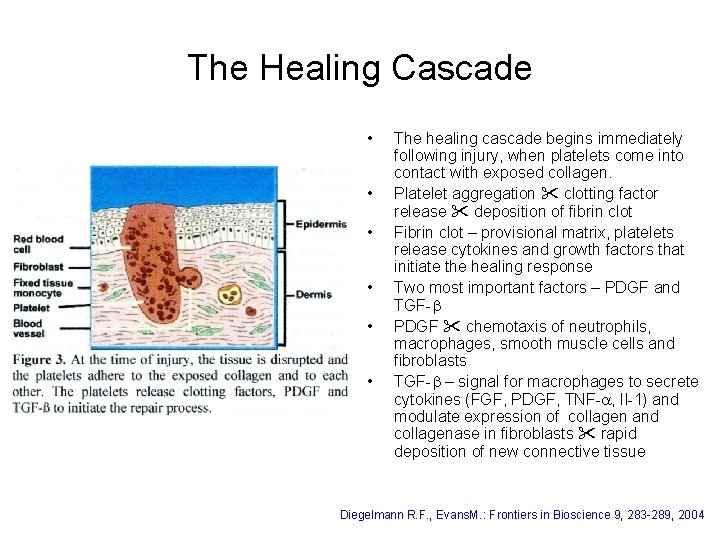
The Healing Cascade • • • The healing cascade begins immediately following injury, when platelets come into contact with exposed collagen. Platelet aggregation clotting factor release deposition of fibrin clot Fibrin clot – provisional matrix, platelets release cytokines and growth factors that initiate the healing response Two most important factors – PDGF and TGF-b PDGF chemotaxis of neutrophils, macrophages, smooth muscle cells and fibroblasts TGF-b – signal for macrophages to secrete cytokines (FGF, PDGF, TNF-a, Il-1) and modulate expression of collagen and collagenase in fibroblasts rapid deposition of new connective tissue Diegelmann R. F. , Evans. M. : Frontiers in Bioscience 9, 283 -289, 2004
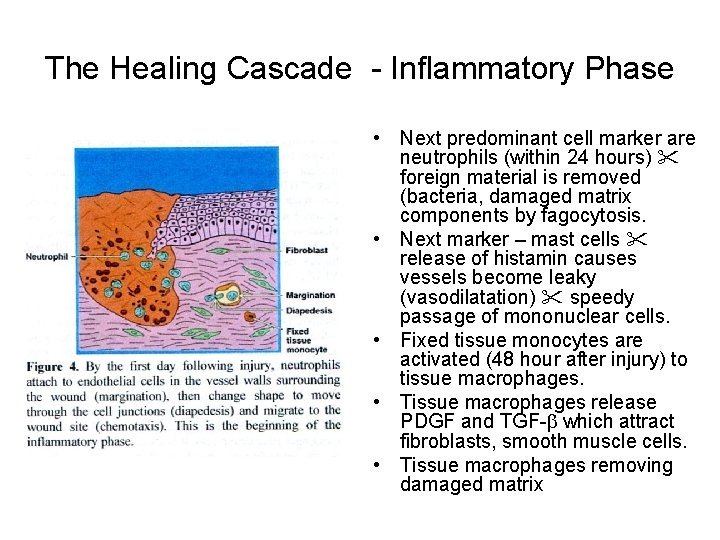
The Healing Cascade - Inflammatory Phase • Next predominant cell marker are neutrophils (within 24 hours) foreign material is removed (bacteria, damaged matrix components by fagocytosis. • Next marker – mast cells release of histamin causes vessels become leaky (vasodilatation) speedy passage of mononuclear cells. • Fixed tissue monocytes are activated (48 hour after injury) to tissue macrophages. • Tissue macrophages release PDGF and TGF-b which attract fibroblasts, smooth muscle cells. • Tissue macrophages removing damaged matrix
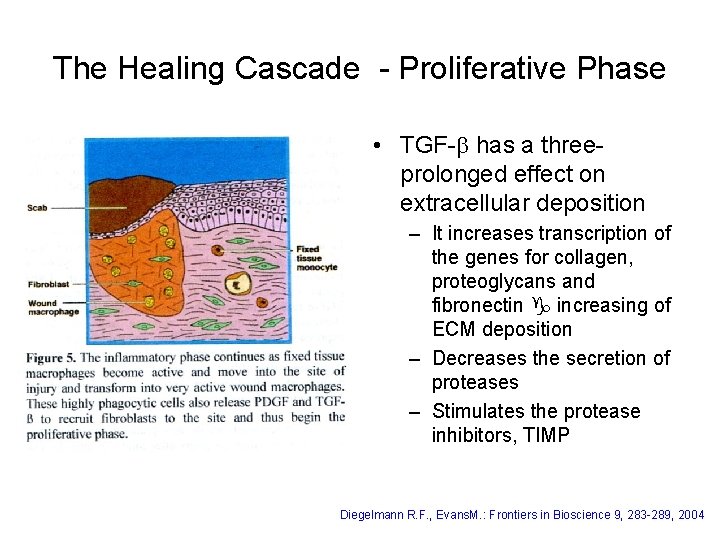
The Healing Cascade - Proliferative Phase • TGF-b has a threeprolonged effect on extracellular deposition – It increases transcription of the genes for collagen, proteoglycans and fibronectin increasing of ECM deposition – Decreases the secretion of proteases – Stimulates the protease inhibitors, TIMP Diegelmann R. F. , Evans. M. : Frontiers in Bioscience 9, 283 -289, 2004

The Healing Cascade - Remodelling Phase • Predominant cell type in the wound site – fibroblasts. • Fibroblasts are attached to the provisional fibrin matrix – start to produce collagen. The first type synthesized - type III collagen (fine reticular fibers) and then is substituted for type I. • Type I collagen is predominant type in the scar tissue.
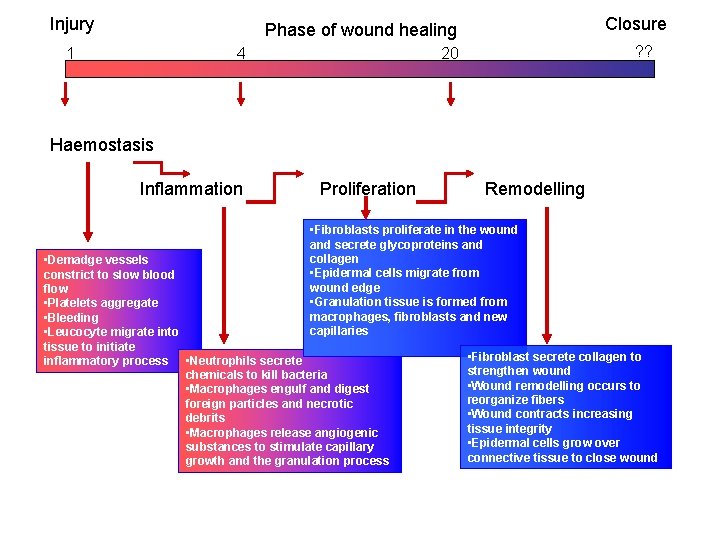
Injury Closure Phase of wound healing 1 4 ? ? 20 Haemostasis Inflammation Proliferation Remodelling • Fibroblasts proliferate in the wound and secrete glycoproteins and collagen • Epidermal cells migrate from wound edge • Granulation tissue is formed from macrophages, fibroblasts and new capillaries • Demadge vessels constrict to slow blood flow • Platelets aggregate • Bleeding • Leucocyte migrate into tissue to initiate inflammatory process • Neutrophils secrete chemicals to kill bacteria • Macrophages engulf and digest foreign particles and necrotic debrits • Macrophages release angiogenic substances to stimulate capillary growth and the granulation process • Fibroblast secrete collagen to strengthen wound • Wound remodelling occurs to reorganize fibers • Wound contracts increasing tissue integrity • Epidermal cells grow over connective tissue to close wound
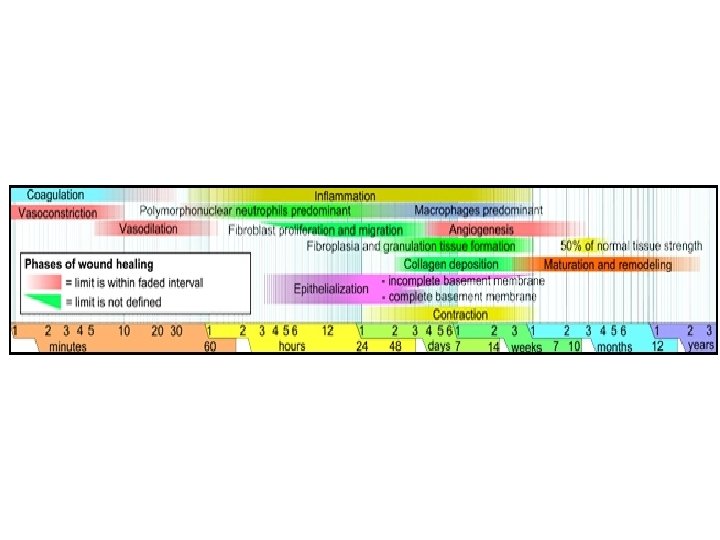

Mechanisms of dermal wounds healing - local factors in the wound microenvironment • High metabolic activity in the wound site – increasing demand for oxygen and nutrients. • Local factors - low p. H, reduced oxygen tension, increased lactate initiate the release of factors activating angiogenesis (vasculogenesis, neovascularisation). • epidermal cells, fibroblasts, macrophages and vascular endothelial cells produce mediator iniating angiogenesis - Vasoactive Endothelial Growth Factor (VEGF ), basic Fibroblast Growth Factor (b. FGF) and TGFb.
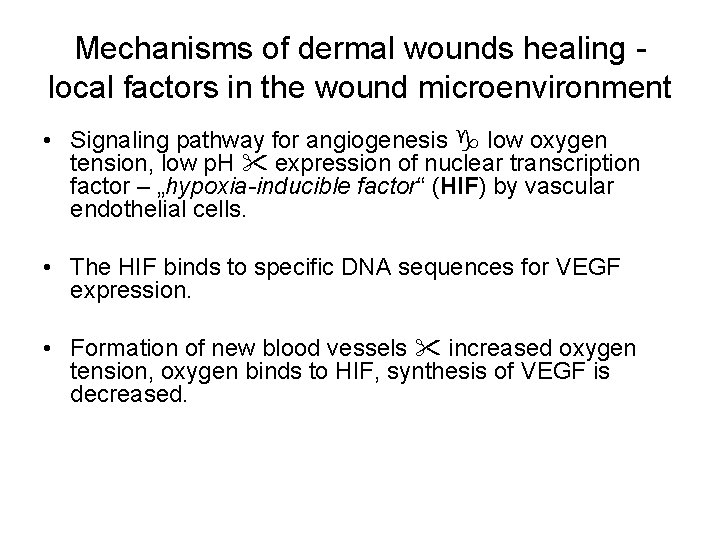
Mechanisms of dermal wounds healing - local factors in the wound microenvironment • Signaling pathway for angiogenesis low oxygen tension, low p. H expression of nuclear transcription factor – „hypoxia-inducible factor“ (HIF) by vascular endothelial cells. • The HIF binds to specific DNA sequences for VEGF expression. • Formation of new blood vessels increased oxygen tension, oxygen binds to HIF, synthesis of VEGF is decreased.

Chronic wound (chronic ulcer) • In acute wounds, there is a precise balance between production and degradation of collagen and other extracellular matrix molecules. • Chronic wounds often remain in the inflammatory stage for too long. • Ischemia is an important factor in the formation and persistence of wounds - causes tissue to become inflamed and cells to release factors that attract neutrophils (cytokines, interleukins, chemokines, leukotrienes, complement components).

Chronic wound (chronic ulcer) • A significant biological marker appears to be excessive infiltration by neutrophils, the over-abundant neutrophil infiltration is responsible for the chronic inflammation. • The neutrophils release significant amounts of proteases – collagenase (matrix metalloproteinase-8) responsible for destruction of the connective tissue matrix – elastase capable of destroying important healing factors - PDGF and TGF-ß – excess matrix metalloproteinases may also cause wounds to become chronic • Excessive reactive oxygen species (ROS) - another marker of chronic ulcers further damage of the cells and prevent cell proliferation and wound closure.
• One of the principal feature of wound healing is formation of new small blood vessels at the site of injury. • Endothelial Progenitor Cells (EPCs) from bone marrow in a process vasculogenesis form new vessels. • EPCs are mobilized circulation repair site. • The cascade of vasculogenesis begins when ischemic tissue releases VEGF (Vasoactive Endothelial Growth Factor). • VEGF goes to the bone marrow, activates NO synthase to produce NO.
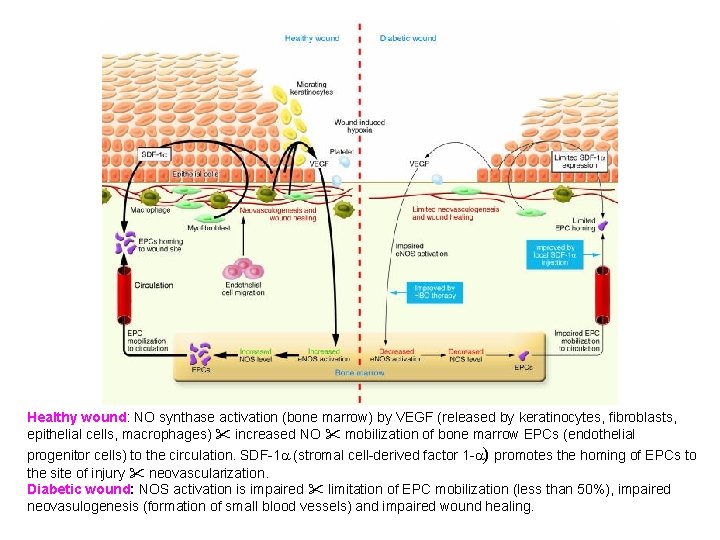
Healthy wound: NO synthase activation (bone marrow) by VEGF (released by keratinocytes, fibroblasts, epithelial cells, macrophages) increased NO mobilization of bone marrow EPCs (endothelial progenitor cells) to the circulation. SDF-1 a (stromal cell-derived factor 1 -a) promotes the homing of EPCs to the site of injury neovascularization. Diabetic wound: NOS activation is impaired limitation of EPC mobilization (less than 50%), impaired neovasulogenesis (formation of small blood vessels) and impaired wound healing.

Fibrosis • Fibrosis can be defined as the replacement of the normal structural elements of the tissue by distorted, non-functional and excessive accumulation of scar tissue. • A clinical example of fibrosis - keloids, hypertrophic scars in the skin. • Fibroblasts isolated from keloids produce about 2 to 3 times more collagen compared to fibroblasts isolated from normal skin in the same patients. • Keloids have increased expression of TGFß and also an upregulation of receptors for TGFß.

Fibrosis • Hypertrophic scars are characterized by excessive accumulation of scar collagen. • • Very significant biological marker that distinguishes keloids from hypertrophic scars the absence of myofibroblasts in keloids and an abundance of these contractile cells in hypertrophic scars. • Most conditions of fibrosis are characterized by an increased density of mast cells. • Diseases characterized by fibrotic processes - scleroderma, Crohn’s disease, liver cirrhosis, lung fibrosis, atherosclerosis, esophageal or urethral strictures.

Diseases caused by collagen gene mutation • More than 1000 mutation have been characterized in 29 collagen genes (COL 1 A 1, COL 1 A 2, COL 2 A 1, COL 3 A 1, COL 4 A 5, COL 7 A 1 a COL 8 A 2). • Most of the mutation are single base substitutions for obligatory glycine to a bigger amino acid, alter folding of triple helix structure. • Bone disorders – osteogenesis imperfecta (OI) (COL 1 A 1 and COL 1 A 2), some form of osteoporosis, • Cartillage disorders – chondrodisplasias (COL 2 A 1), some form of osteoarthritis (COL 11 A 1, COL 11 A 2), intervertebral discs. • Ehlers-Danlos syndrome (COL 5 A 1 and COL 5 A 2, COL 3 A 1 ) – a heterogenous group, several subtypes – joint and skin changes, the rupture of arteries. • Other mutation – duplication, insertion, complex rearrangements.

UV rays and skin damage • Understanding the difference between UV-A and UV-B rays: – UV-A (320 -400 nm) – tanning rays/ indirect DNA damage – UV-B (280 -320 nm) – vitamin D-making rays/ primary cause of sunburn and direct DNA damage

UV rays and skin damage • Penetration ability: UV-A’s longer wavelength has a greater penetration ability than UV-B – UV-A penetrates earth’s ozone layer, clouds, smog – penetrates to dermis in skin – i. e. deeper than UV-B, which only reaches surface level (epidermis) – 70 -90% UV-A penetrates window glass. • UV-A oxidizes melanin from melanocytes to create the tan color in the skin. • UV-A can cause oxidative stress leading to cancerous mutations – creates ROS (Reactive Oxygen Species) indirect DNA/RNA damage
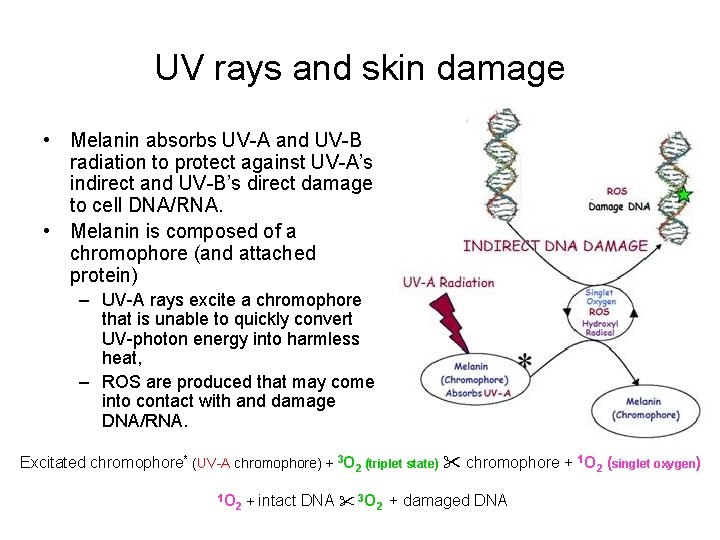
UV rays and skin damage • Melanin absorbs UV-A and UV-B radiation to protect against UV-A’s indirect and UV-B’s direct damage to cell DNA/RNA. • Melanin is composed of a chromophore (and attached protein) – UV-A rays excite a chromophore that is unable to quickly convert UV-photon energy into harmless heat, – ROS are produced that may come into contact with and damage DNA/RNA. Excitated chromophore* (UV-A chromophore) + 3 O 2 (triplet state) chromophore + 1 O 2 (singlet oxygen) 1 O 2 + intact DNA 3 O 2 + damaged DNA

UV rays and skin damage • UV-A’s indirect damage is pain-free and has no other warning signals the ROS (hydroxyl radical and singlet oxygen) traveling through the body, affecting inner organs. – E. g. 92% of malignant melanoma is caused by indirect DNA damage • The adequate presence in the skin of antioxidants (vitamin A and astaxanthin) can neutralize ROS produced. • UV-A can cause photo-aging - by damaging collagen and elastin fibers and destroying vitamin A in skin – Actinic (or solar) elastosis is an accumulation of abnormal elastin (elastic tissue) in the skin’s dermis layer, and in the conjunctiva of the eye, as a result of prolonged / excessive sun exposure.

UV-B (280 -320 nm) UV-B has both beneficial and harmful health effects – partially absorbed by ozone layer – only affects skin’s epidermis (surface) layer • UV-B produces vitamin D 3 - potent defense against melanoma - calcitriol causes growth inhibition and apoptotic cell death of cancer cells
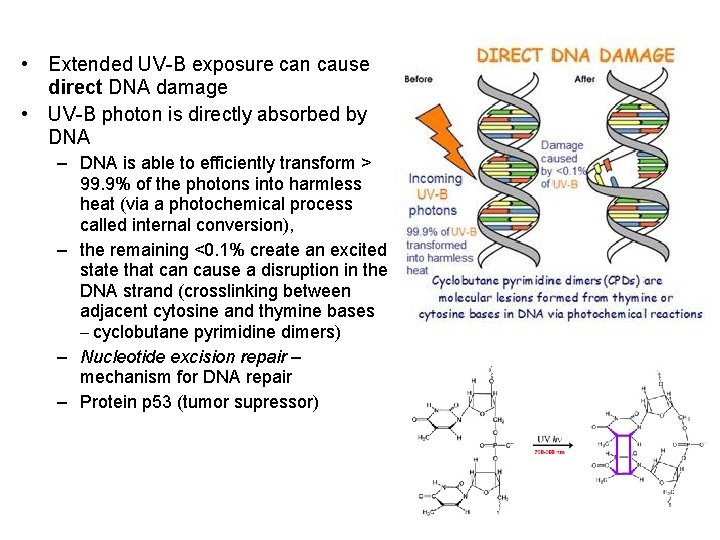
• Extended UV-B exposure can cause direct DNA damage • UV-B photon is directly absorbed by DNA – DNA is able to efficiently transform > 99. 9% of the photons into harmless heat (via a photochemical process called internal conversion), – the remaining <0. 1% create an excited state that can cause a disruption in the DNA strand (crosslinking between adjacent cytosine and thymine bases – cyclobutane pyrimidine dimers) – Nucleotide excision repair – mechanism for DNA repair – Protein p 53 (tumor supressor)
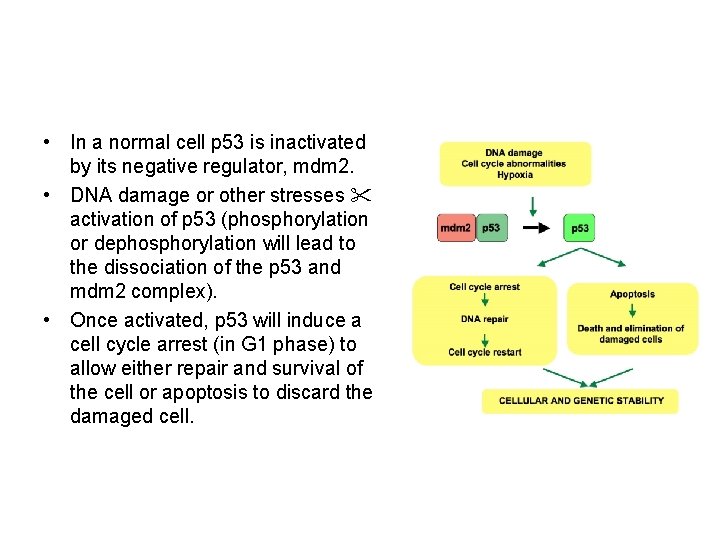
• In a normal cell p 53 is inactivated by its negative regulator, mdm 2. • DNA damage or other stresses activation of p 53 (phosphorylation or dephosphorylation will lead to the dissociation of the p 53 and mdm 2 complex). • Once activated, p 53 will induce a cell cycle arrest (in G 1 phase) to allow either repair and survival of the cell or apoptosis to discard the damaged cell.
- Slides: 46