Conductivity and the Drude Model Objectives By the

Conductivity and the Drude Model Objectives By the end of this section you should be able to: p Understand how to apply the Drude model p Determine the density of conduction electrons p Calculate the electrical conductivity of a metal p Estimate or calculate the mean free path

Drude Model (1900) Za = atomic number = # of electrons simple model for conductivity isolated atom in a metal Z valence electrons are weakly bound to the nucleus (participate in reactions) Za – Z core electrons are tightly bound to the nucleus (less of a role) In a metal, the core electrons remain bound to the nucleus to form the metallic ion Valence electrons wander away from their parent atoms, called conduction electrons
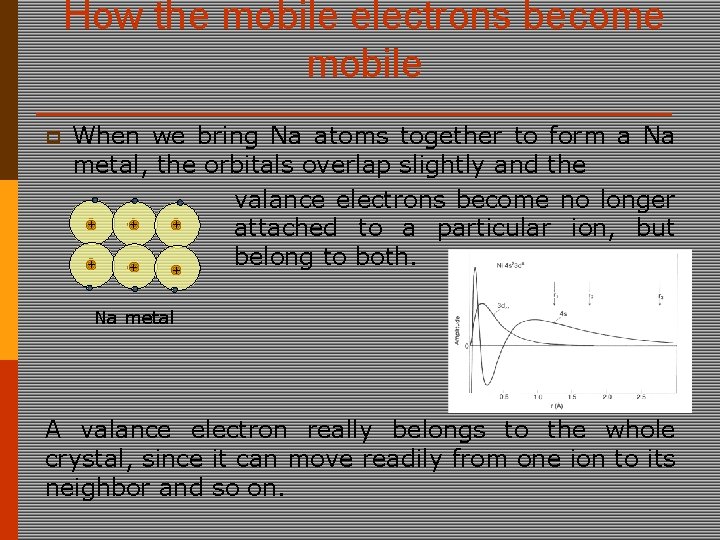
How the mobile electrons become mobile p When we bring Na atoms together to form a Na metal, the orbitals overlap slightly and the valance electrons become no longer + + + attached to a particular ion, but belong to both. + + + Na metal A valance electron really belongs to the whole crystal, since it can move readily from one ion to its neighbor and so on.
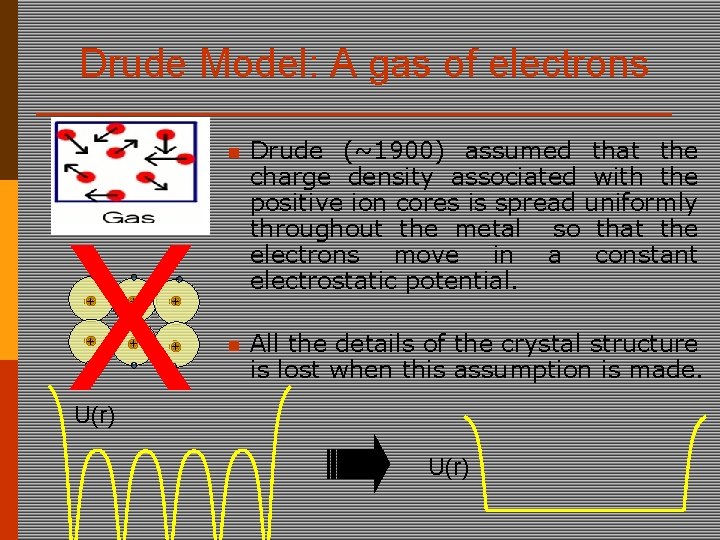
Drude Model: A gas of electrons X + + + n Drude (~1900) assumed that the charge density associated with the positive ion cores is spread uniformly throughout the metal so that the electrons move in a constant electrostatic potential. n All the details of the crystal structure is lost when this assumption is made. U(r)

Drude makes the free electron approximation U(r)=0 Neglect periodic potential & scattering between electrons Reasonable for “simple metals” (Alkali Li, Na, K, Cs, Rb) What does this remind you of?

Assumptions/Strengths/Weakness es p p p He also ignored electron-electron and electronion interactions (except to say electron-ion interaction were the cause of the scattering) If field applied, electron moves in straight line between collisions (no electron-electron collisions). Velocity changes instantaneously, but direction random. In no field<v>=0, but <v 2> T (gas) We will use a trick to use this model later (next) Positives: Strength and density of metals Negative: Predicts too high of a classical specific heat (100 x larger than observed at RT)

Conduction electron Density n = N/V 6. 022 x 1023 atoms per mole p Multiply by number of valence electrons (Z) p Convert moles to cm 3 (using mass density m and atomic mass A) p n = N/V = 6. 022 x 1023 Z m /A Similar to finding number of atoms
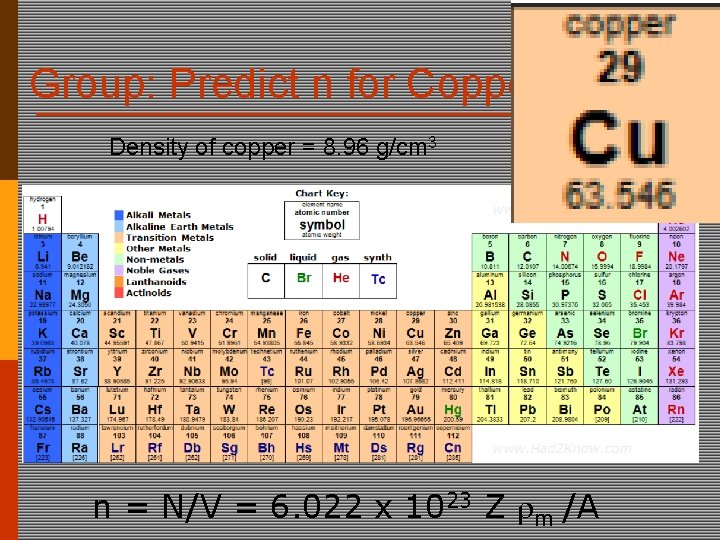
Group: Predict n for Copper Density of copper = 8. 96 g/cm 3 n = N/V = 6. 022 x 1023 Z m /A

How Far Apart are the Conduction Electrons? density of conduction electrons in metals ~1022 – 1023 cm-3 rs = radius of a sphere whose volume is equal to the volume per electron mean inter-electron spacing in metals rs ~ 1 – 3 Å

A model for electrical conduction p (a) Drude model in 1900 n In the absence of an electric field, the conduction electrons move in random directions through the conductor with large average speeds. The drift velocity of the free electrons is zero. There is no current in the conductor since there is no net flow of charge. (b) Estimate Average Speed n When an electric field is applied, in addition to the random motion, the free electrons drift slowly (vd ~ 10 -4 m/s) in a direction opposite that of the electric field.

“Quasi-Classical” Theory of Transport Microscopic Macroscopic Charge Current Density: J = I/A Depends on dimension = R A /length = 1/
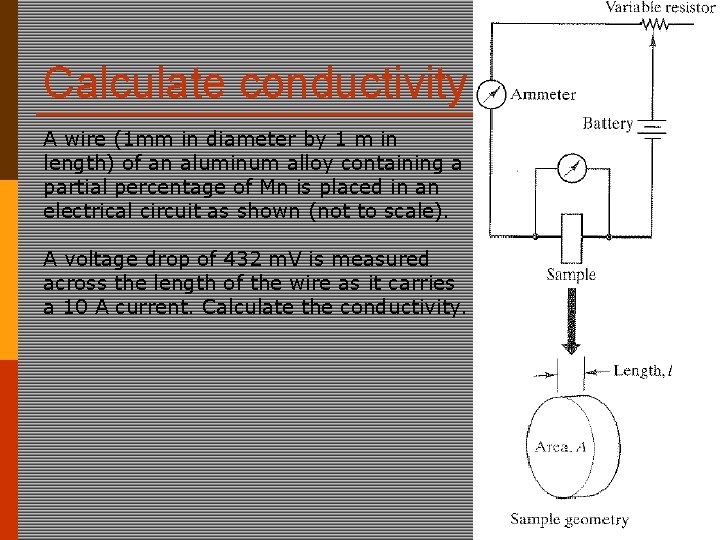
Calculate conductivity A wire (1 mm in diameter by 1 m in length) of an aluminum alloy containing a partial percentage of Mn is placed in an electrical circuit as shown (not to scale). A voltage drop of 432 m. V is measured across the length of the wire as it carries a 10 A current. Calculate the conductivity.
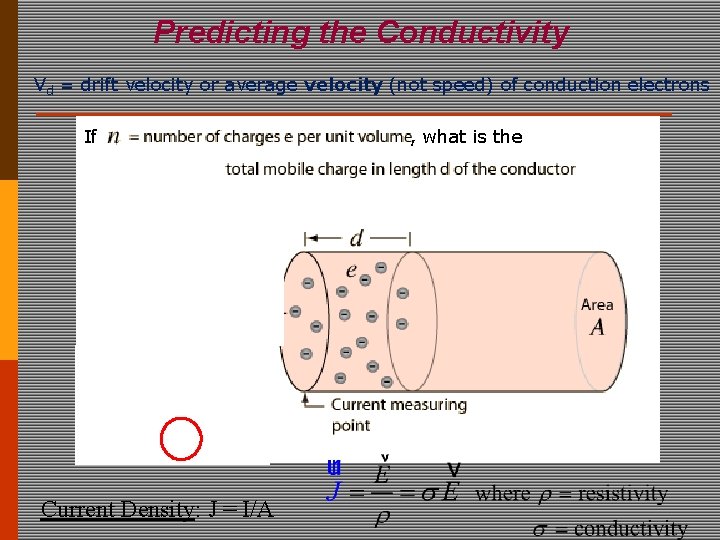
Predicting the Conductivity Vd = drift velocity or average velocity (not speed) of conduction electrons If Current Density: J = I/A , what is the

Determining Velocity When Applying an Electric Field E to a Metal F = ma = - e E, so acceleration a = - e E / m Integrating gives v = - e E t / m So the average vavg = - e E / m, where is “relaxation time” or time between collisions vavg = x/ , we can find the xavg or mean free path

Calculating Conductivity Ohm’s law vavg = - e E / m J = E = - n e vavg = - n e (- e E / m) Then conductivity = n e 2 / m ( = 1/ ) And = m / ( n e 2) ~10 -15 – 10 -14 s
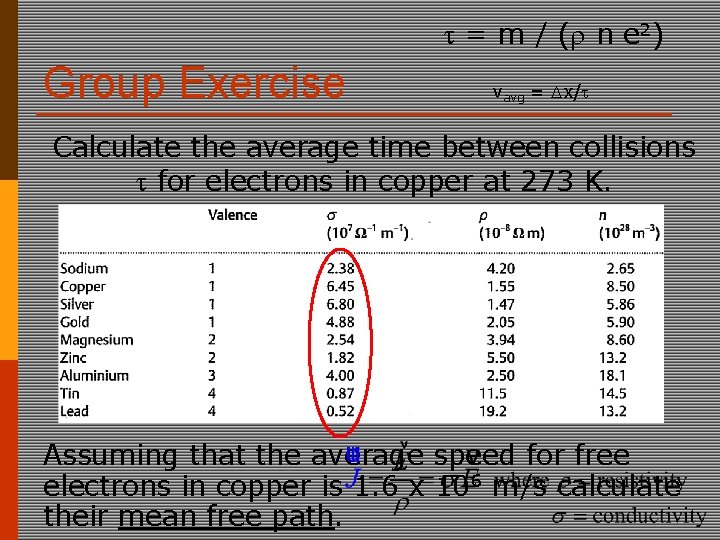
= m / ( n e 2) Group Exercise vavg = x/ Calculate the average time between collisions for electrons in copper at 273 K. Assuming that the average speed for free electrons in copper is 1. 6 x 106 m/s calculate their mean free path.
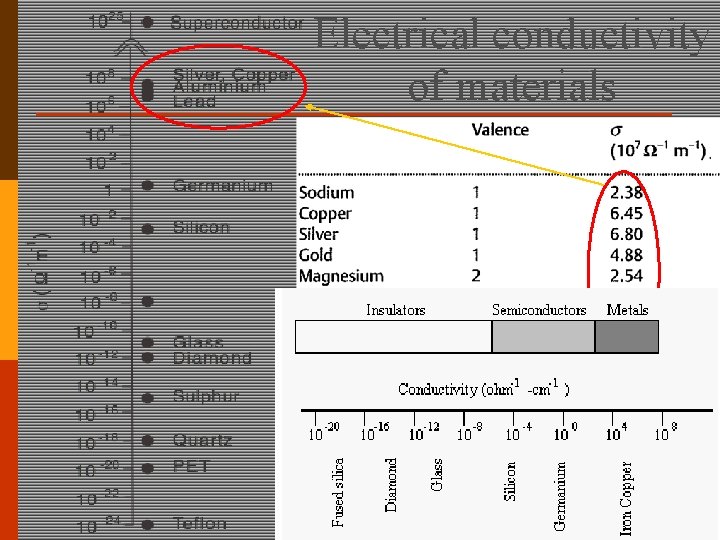
Electrical conductivity of materials
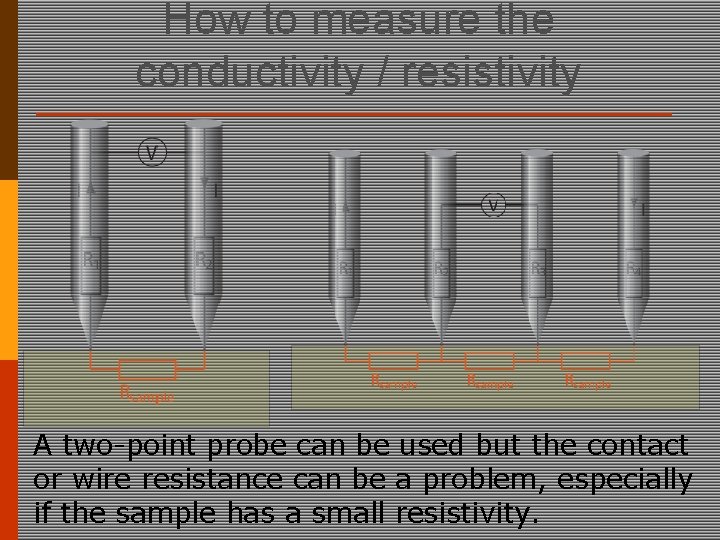
How to measure the conductivity / resistivity A two-point probe can be used but the contact or wire resistance can be a problem, especially if the sample has a small resistivity.

Resistivity vs Temperature not 10 accounted for in Drude model 20 Resistivity is temperature dependent mostly because of the temperature dependence of the scattering time . = m / ( n e 2) Resistivity (Ωm) Insulator Diamond 1010 Semiconductor Germanium 100 Metal 10 -100 200 Temperature (K) Copper 300 • In Metals, the resistivity increases with increasing temperature. The scattering time decreases with increasing temperature T (due to more phonons and more defects), so as the temperature increases ρ increases (for the same number of conduction electrons n) • We will find the opposite trend occurs in semiconductors but we will need more physics to understand why.

Struggles with: Sign of Hall Effect In a current carrying wire when in a perpendicular magnetic field, the current should be drawn to one side of the wire. As a result, the resistance will increase and a transverse voltage develops. H Lorentz force = ev/c x H --------- ++++++++ - +

If a current flows (of velocity v. D) in the positive x direction and a uniform magnetic field is applied in the positive z-direction, use the Lorentz force to determine the magnitude and direction of the resulting Hall field, first in terms of velocity, but then current density. Lorentz force = • Ey= vx. Hz/c = jx. Hz/nec ev/c x H • • Hall coefficient: induced E/current density * applied H field RH= Ey/jx. H = 1/nec how big? R is very small for metals as n is very large. Hz ---------++++++++ - x y

The Hall coefficient c Ohm’s law contains e 2 But for RH the sign of e is important. 10 k. G increases the resistance of bismuth 8 x, while only 0. 1% for silver A hole is the lack of an electron. It has the opposite charge so +e.

Application: Hall Probe The measurement of large magnetic fields (~1 T) is often done by the Hall effect. A sample is placed in the magnetic field and the transverse voltage is measured. For a 100 - m thick Cu film, in a 1. 0 T magnetic field and through which I = 0. 5 A is passing, the Hall voltage is 0. 737 V. Advantages: Hall effect devices are immune to dust, dirt, mud, and water, making them better for position sensing than other means such as optical and electromechanical sensing. Can also create non contact current sensors. Disadvantages: Magnetic flux from the surroundings (such as other wires) may diminish or enhance the field the Hall probe intends to detect, affecting results. Also, the output from this type of sensor cannot be used to directly drive actuators without amplification.
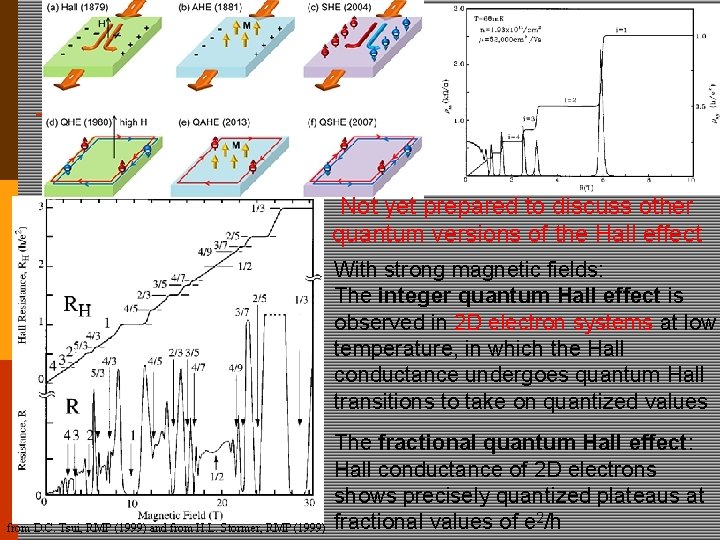
s ic t e n ld e i f g a m k a we Not yet prepared to discuss other quantum versions of the Hall effect With strong magnetic fields: The integer quantum Hall effect is observed in 2 D electron systems at low temperature, in which the Hall conductance undergoes quantum Hall transitions to take on quantized values from D. C. Tsui, RMP (1999) and from H. L. Stormer, RMP (1999) The fractional quantum Hall effect: Hall conductance of 2 D electrons shows precisely quantized plateaus at fractional values of e 2/h
- Slides: 24