Conductivity and Bonding EXPERIMENT 16 TESTING SOLUTIONS ALCOHOL
Conductivity and Bonding EXPERIMENT # 16
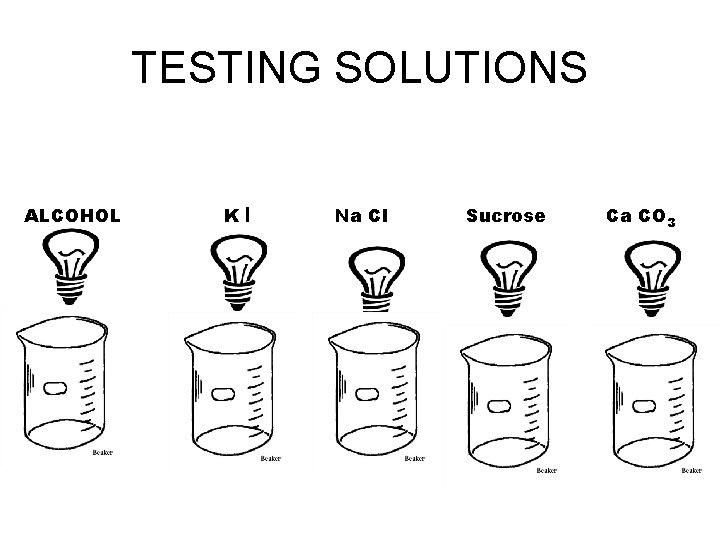
TESTING SOLUTIONS ALCOHOL KI Na Cl Sucrose Ca CO 3
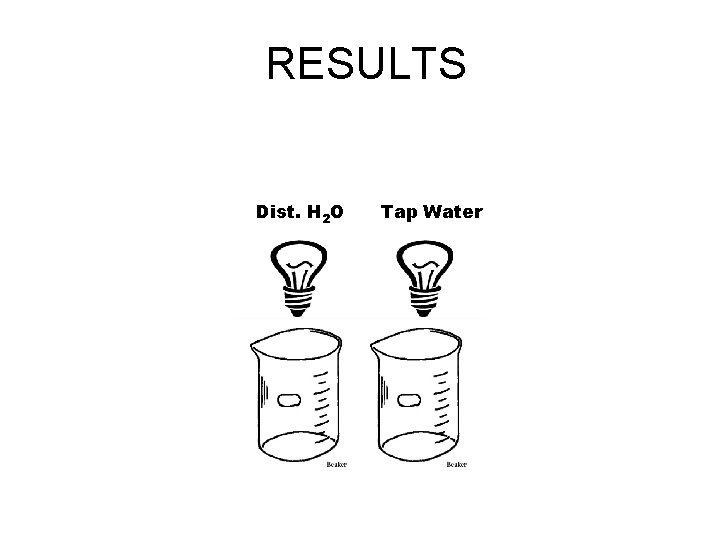
RESULTS Dist. H 2 O Tap Water
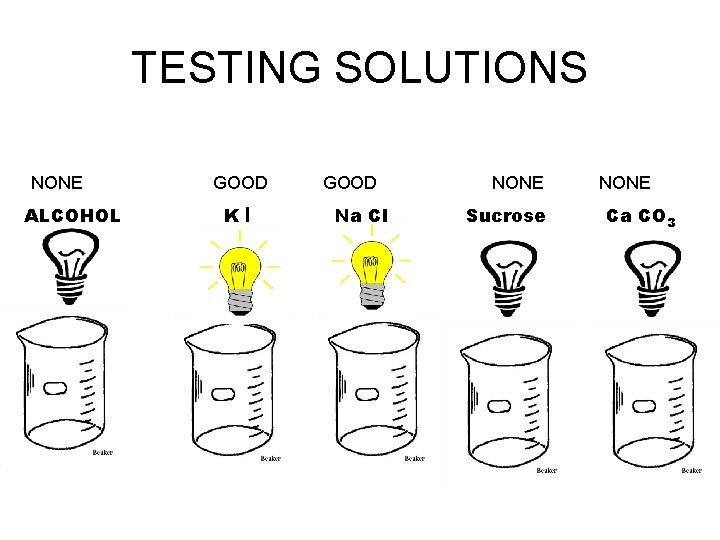
TESTING SOLUTIONS NONE ALCOHOL GOOD KI GOOD Na Cl NONE Sucrose NONE Ca CO 3
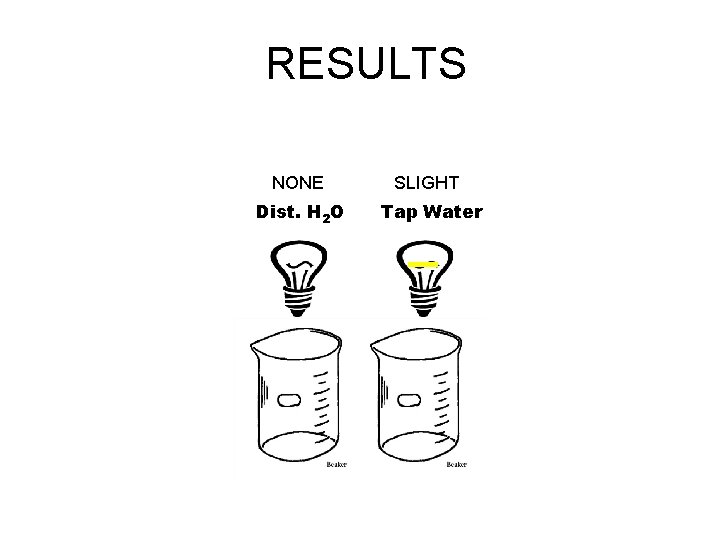
RESULTS NONE SLIGHT Dist. H 2 O Tap Water

1) Explain the difference between distilled water and tap water Distilled water Does not contain any minerals such as calcium and magnesium. They have been removed by boiling. Tap water Contains the minerals calcium and magnesium. They are in the rock layers that we get our water from. They slightly conduct an electric current
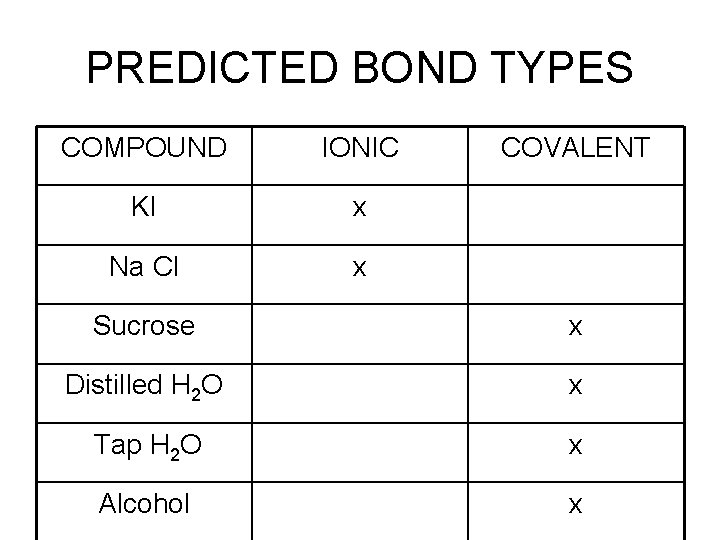
PREDICTED BOND TYPES COMPOUND IONIC KI x Na Cl x COVALENT Sucrose x Distilled H 2 O x Tap H 2 O x Alcohol x

POLAR MOLECULE THE ELECTRONS FROM THE HYDROGEN ARE PULLED INWARD TOWARD THE OXYGEN AND MAKE THE FAR END POSITIVE. THE MOLECULE LOOKS LIKE A MAGNET.

IONIC COMPOUNDS ELECTRONS ARE LOST SODIUM 10 ELECTRONS CHLORINE 18 ELECTRONS FROM ONE ATOM TO ANOTHER. MAKING THE ATOMS INTO IONS WITH A + OR - CHARGE

PI BONDS pi bonds (π bonds) are covalent chemical bonds where two lobes of an orbital on one atom overlap two lobes of an orbital on another atom.
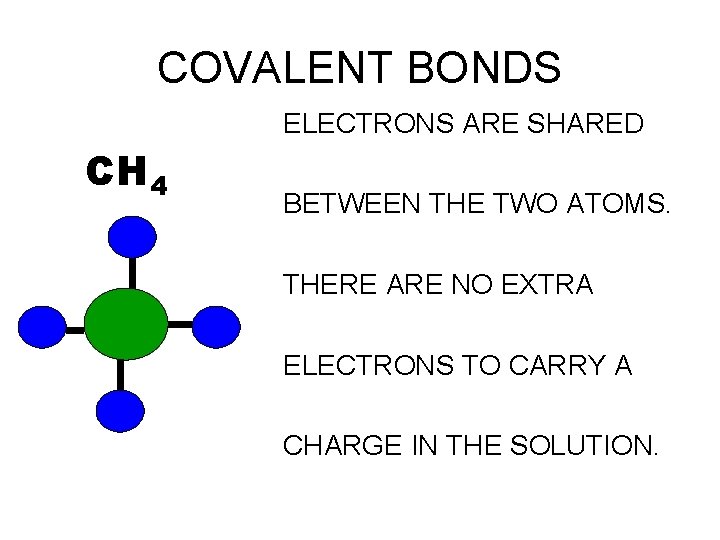
COVALENT BONDS ELECTRONS ARE SHARED CH 4 BETWEEN THE TWO ATOMS. THERE ARE NO EXTRA ELECTRONS TO CARRY A CHARGE IN THE SOLUTION.

HYDROGEN BONDS A hydrogen bond is a weak type of force that forms a special type of attraction. This occurs when a hydrogen atom bonded to a strongly electronegative atom is near another electronegative atom with a lone pair of electrons. These bonds are generally stronger than ordinary forces, but weaker than true covalent and ionic bonds.

COORDINATE COVALENT BONDS A coordinate covalent bond is a kind of covalent bond in which the two electrons come from the same atom. Examples= when metals from Group II and the Transition Elements give up two electrons. Ca+2 Ba+2 Sr+2 Fe +2

POLAR COVALENT BONDS In a polar covalent bond, the electrons shared by the atoms spend a greater amount of time, on the average, closer to the Oxygen nucleus than the Hydrogen nucleus. This is because of the geometry of the molecule and the great electronegativity difference between the Hydrogen atom and the Oxygen atom.
- Slides: 14