Conducting Longitudinal Cohort Studies in PBRNs Sandra Burge


Conducting Longitudinal Cohort Studies in PBRNs Sandra Burge Tamara Armstrong Robert Wood Jason Hill

Introductions

Objectives Participants will review and discuss design issues in longitudinal cohort studies conducted in practice-based research networks ◦ ◦ ◦ ◦ IRB & Consent Recruitment & Retention Data collection Measurement issues Statistical Analysis Workforce Issues Funding

Defined Practice-Based Research Networks ◦ "a group of ambulatory practices devoted principally to the primary care of patients. Typically, PBRNs draw on the experience and insight of practicing clinicians to identify and frame research questions whose answers can improve the practice of primary care. By linking these questions with rigorous research methods, the PBRN can produce research findings that are immediately relevant to the clinician and, in theory, more easily assimilated into everyday practice". [AHRQ PBRN Resource Center]

RRNe. T Residency Research Network of Texas ◦ We are a group of ten ambulatory practices devoted to the training of family physicians and the primary care of patients. We draw on the experience and insight of practicing clinicianteachers to identify and frame research questions whose answers can improve the practice and teaching of primary care. ◦ We have about 100 family physician faculty, 300 family medicine residents, and we see about 300, 000 patient visits per year. ◦ Most of our patients are low-income and ethnic minorities

RRNe. T Steering committee is comprised of one faculty representative from each residency program.

Simply Defined A longitudinal study is one conducted over the passage of time

Defined A longitudinal study is a correlational study involving repeated observations of the same phenomena over time In medical research, the key aims are to: ◦ assess changes over time ◦ uncover predictors of diseases http: //en. wikipedia. org/wiki/Longitudinal_study

Defined Observational Prospective ◦ Prospective Cohort Studies Retrospective ◦ Historical or Retrospective Cohort Studies ◦ Case Control Studies

Prospective Cohort Studies 1. 2. 3. 4. 5. Select a sample. Arrange to assess the sample multiple times. Measure demographic variables, predictors, and covariates at baseline (and perhaps at followup). Measure outcome variable (e. g. , disease) at baseline and followup. For data analysis, correlate predictors with changes in outcomes over time. Hulley S. B. et al. Designing Clinical Research 3 rd Ed. Philadelphia PA. Lippincott Williams & Wilkins, 2007.

Prospective Cohort Studies Advantages One can investigate causes of medical conditions by measuring predictor before outcome occurs. One can establish occurrence of variables in time. Measurement of predictors need not be influenced by outcomes. Disadvantages Confounding variables can decrease power. Loss of participants impacts study outcomes. Follow-up can be expensive and time consuming. This design is not suited for rare conditions.

Retrospective Cohort Studies 1. 2. 3. 4. Identify a sample From records, collect predictors measured in the past From records or subjects, collect outcomes measured in the present For data analysis, correlate predictors and outcomes Hulley S. B. et al. Designing Clinical Research 3 rd Ed. Philadelphia PA. Lippincott Williams & Wilkins, 2007.

Case Control Studies 1. 2. 3. 4. Identify a cohort with outcome condition (Cases) Identify a comparable cohort without the condition (Controls) From records, collect predictors measured in the past For data analysis, compare predictor variables in Cases versus Controls. Hulley S. B. et al. Designing Clinical Research 3 rd Ed. Philadelphia PA. Lippincott Williams & Wilkins, 2007.

Retrospective Studies Advantages Retrospective designs cost less in time and expense. One does not need to design measurements. Participant recruitment requires less effort. One does not need to follow patients. Case-Control is a good design for rare conditions. Disadvantages Investigator control is limited in terms of study design and sampling. Data may be incomplete. Measurements may not always suit investigator needs.

Today’s Focus Prospective Cohort Design

Key Design Issues Read the following longitudinal cohort study, and write down three design challenges that the investigators had to address. 1. . 2. . 3. .

Tuomilehto, J. et al. Coffee consumption and risk of type 2 diabetes. . . JAMA 2004; 291(10). Objective: To determine the relationship between coffee consumption and the incidence of type 2 DM among Finnish people, who are the world’s highest coffee consumers! Methods: A prospective survey conducted in 1982, 1987, 1992 included 14, 629 men and women age 35 to 64 who had no history of stroke, CHD, or DM at baseline. Coffee consumption and other study parameters were determined at baseline using standardized measurements. Hazard ratios for the incidence of type 2 DM were estimated for different levels of daily coffee consumption. Results: During a mean followup of 12 years, there were 381 new cases of DM. After controlling for confounding factors, hazard ratios were calculated, showing an inverse relationship between daily coffee consumption and DM; i. e. more coffee predicts lower incidence of diabetes.

Key Design Issues What challenges? 1. . 2. . 3. .

Key Design Issues IRB & Consent Recruitment & Retention Data Collection Measurement Statistical Analysis Workforce Issues Funding

RRNe. T Longitudinal Study of Chronic Low Back Pain Faced with the difficulty of caring for patients who do not get well, and who often place family physicians in the ethical quandary of prescribing or not prescribing narcotic medicines, RRNe. T physicians were interested in understanding patients with chronic low back pain.

RRNe. T Longitudinal Study of Chronic Low Back Pain RRNe. T identified 367 patients with chronic low back pain and gathered data using surveys and chart reviews. Patients were re-assessed at 1 -year and 2 year followup, using the same methods.
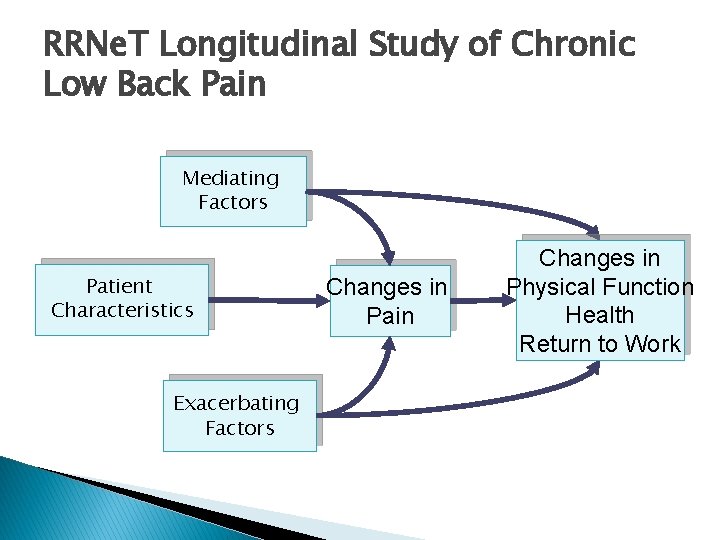
RRNe. T Longitudinal Study of Chronic Low Back Pain Mediating Factors Patient Characteristics Exacerbating Factors Changes in Pain Changes in Physical Function Health Return to Work

Key Design Issues 1. 2. 3. 4. 5. 6. 7. IRB & Consent Recruitment & Retention Data Collection Measurement Statistical Analysis Workforce Issues Funding

Key Design Issues 1. 2. 3. 4. 5. 6. 7. IRB & Consent Recruitment & Retention Data Collection Measurement Statistical Analysis Workforce Issues Funding

IRB & Consent RRNe. T has 10 IRBs! – one in each hospital. ◦ “Model protocol” and “Model Consent” are drafted and distributed to each practice. ◦ Steering Committee faculty is responsible for walking approval through their own IRB. ◦ We applied for Expedited Review. To follow patients, one must collect PHI, i. e. names & addresses. Consent form requires 2 signatures ◦ To participate in the study ◦ To consent to our use of PHI We required a HIPAA waiver for medical students to review patients’ records for study eligibility.

Key Design Issues 1. 2. 3. 4. 5. 6. 7. IRB & Consent Recruitment & Retention Data Collection Measurement Statistical Analysis Workforce Issues Funding

Recruitment & Retention RRNe. T - Recruitment ◦ “Free” assistance (medical students) is available each summer ◦ Medical students reviewed records of appointed patients to determine eligible individuals ◦ They approached patients in the waiting room or exam room and invited them to participate. ◦ After consent, patients completed a 5 -6 page survey. ◦ Later, medical students abstracted patient records for relevant information. ◦ We did not pay patients for participation. ◦ Refusal rate was 12. 5%

Recruitment & Retention RRNe. T – Retention One year later, we had a new group of students We sent anticipatory letters to patients. Those who returned to clinic were surveyed there. Others patients were telephoned for surveys. We did not pay patients for followup. Our followup rate was 62% for surveys, ranging from 39% to 85%. ◦ Our followup rate was 92% for medical records. ◦ ◦ ◦

Small Groups

Recruitment & Retention What are your recommendations? ◦ Gather in small groups ◦ List 2 – 5 strategies for recruiting patients into longitudinal studies. ◦ List 2 - 5 strategies for retaining patients in longitudinal studies.

Recruitment & Retention Your recommendations

Recruitment & Retention Recruitment ◦ ◦ ◦ ◦ Sample of records or visits Vary times of year Fliers Media Face-to-face Contact by letter Phone for interview appointment (not cold call) Incentives $$ Blumenthal DS, et al. Recruitment and retention of subjects for a longitudinal cancer prevention study … HSR: Health Services Research 1995; 30(1) Gilliss, CL et al. Recruitment and retention of healthy minority women… Journal of Women’s Health & Gender-based Medicine 2001; 10(1): 77 -85

Recruitment & Retention ◦ ◦ ◦ Alternative contact information gathered at baseline Frequent contact (birthday cards, newsletters) Incentives for followup $$ Anticipatory letter Phone to schedule interview appointment Home visit Blumenthal DS, et al. Recruitment and retention of subjects for a longitudinal cancer prevention study … HSR: Health Services Research 1995; 30(1) Gilliss, CL et al. Recruitment and retention of healthy minority women… Journal of Women’s Health & Gender-based Medicine 2001; 10(1): 77 -85

Key Design Issues 1. 2. 3. 4. 5. 6. 7. IRB & Consent Recruitment & Retention Data Collection Consistency of Measurement Statistical Analysis Workforce Issues Funding

Data Collection RRNe. T Data Collection at Baseline ◦ Patient Survey ◦ Chart Review RRNe. T Data Collection at 1 -year & 2 -Year ◦ Patient Survey, or ◦ Telephone Survey ◦ Chart Review

Key Design Issues 1. 2. 3. 4. 5. 6. 7. IRB & Consent Recruitment & Retention Data Collection Measurement Statistical Analysis Workforce Issues Funding
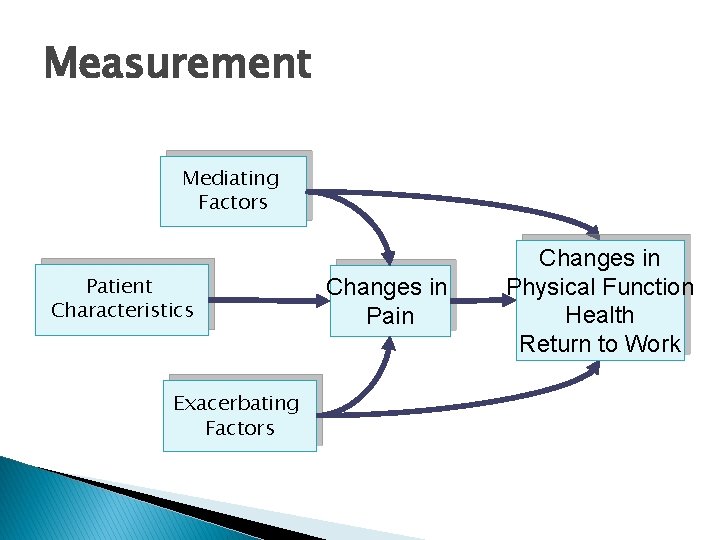
Measurement Mediating Factors Patient Characteristics Exacerbating Factors Changes in Pain Changes in Physical Function Health Return to Work

Measurement RRNe. T measured these variables each year: ◦ Patient characteristics ◦ Mediating factors – analgesics, other treatments, family support ◦ Exacerbating factors – depression, anxiety, family stress, risk for addiction ◦ Pain Severity ◦ Physical & Role Functioning ◦ General Health

Small Groups

Data Collection & Measurement What are your recommendations? ◦ Gather in small groups ◦ List 2 – 5 strategies to collect data at followup. ◦ List 2 - 5 recommendations about measurement decisions.

Data Collection & Measurement Your recommendations

Data Collection & Measurement Other followup methods of data collection ◦ ◦ ◦ Mailed Survey Web-based Survey “Catch-as-catch-can” during a routine office visit Scheduled return Automated telephone survey Medical records Van Weel

Data Collection & Measurement The Nijmegen network has recorded all episodes of morbidity encountered in Dutch family medicine since 1971 in a stable practice population. What helps in data collection? ◦ Stable population ◦ Continuity of care ◦ Motivated physicians and staff; ◦ Ownership of the data; ◦ Standardized data; ◦ Clinician-friendly means of collecting data; ◦ Training in data collection, reinforced routinely ◦ Feedback to practices on the research findings. van Weel C. Longitudinal research and data collection in primary care. Ann Fam Med. 2005 May-Jun; 3 Suppl 1: S 46 -51.

Data Collection & Measurement considerations ◦ Are the assessments of high quality? ◦ ◦ Reliable & Valid Consistently measured over time What if definitions change? (e. g. stds of care? ) What variables are expected to change over time? How often should one assess change? Who should assess the changes? (investigator bias) Will the assessments retain their meaning over time? (e. g. BMI from childhood to adulthood) Magnusson D. , Bergman LR. Data Quality in Longitudinal Research. Cambridge University Press, 1990.

Key Design Issues 1. 2. 3. 4. 5. 6. 7. IRB & Consent Recruitment & Retention Data Collection Measurement Statistical Analysis Workforce Issues Funding

Statistical Analysis Repeated Measures ANOVA or Random effects linear models Survival Analysis: Cox Model Regressions What to do about missing data? Small amounts may not seriously impact estimates of effects within some analyses. Large amounts may require missing value imputation (NORM or Proc MI in SAS).

Repeated Measures

Key Design Issues 1. 2. 3. 4. 5. 6. 7. IRB & Consent Recruitment & Retention Data Collection Measurement Statistical Analysis Workforce Issues Funding

Workforce Issues RRNe. T had a new workforce of medical students each year. 2 -day pre-study training included: ◦ ◦ Basic research methods Consent & Recruitment Followup Protocol Data Entry On site training by RRNe. T faculty ◦ Clinic personnel ◦ Local routines & resources ◦ Local patient population

Workforce Issues RRNe. T Steering Committee ◦ Train the trainers ◦ Re-visited study protocol each spring to prepare for students’ arrival

Key Design Issues 1. 2. 3. 4. 5. 6. 7. IRB & Consent Recruitment & Retention Consistency of Measurement Data Collection Statistical Analysis Workforce Issues Funding

Funding RRNe. T conducted this study on a shoestring ◦ ◦ Stipends for students from Dean’s office Additional small grants from TAFP Student housing from AHEC Later, HRSA funding for infrastructure NIH and large foundations generally have 3 -5 year funding limits.

Workforce & Funding Issues Final Recommendations ◦ Individually, write down 3 recommendations about workforce issues and/ or funding, as it pertains to Longitudinal research.

Summary 1. 2. 3. 4. 5. 6. 7. IRB & Consent Recruitment & Retention Data Collection Measurement Statistical Analysis Workforce Issues Funding

Future Plans We will collate your recommendations and post them on FMDRL. ◦ Please turn in your worksheets.

- Slides: 57