CONCEPT 3 ANALYZING CELL METABOLISM AND ENZYME FUNCTION

CONCEPT 3: ANALYZING CELL METABOLISM AND ENZYME FUNCTION (CH 8, AP LAB 2) Holtzclaw: “Metabolism” pg 53 -57 Campbell: Read pg 142 -145, 155 -156 Look at figures on pg 147, 157 -159

Fig. 8 -1

CONCEPT 3: LEARNING INTENTIONS An Introduction to Energy Metabolism The key role of ATP in energy coupling. That enzymes work by lowering the energy of activation. The catalytic cycle of an enzyme that results in the production of a final product. The factors that influence the efficiency of enzymes. Lab 2: Enzyme Catalysis The factors that affect the rate of an enzyme reaction such as temperature, p. H, enzyme concentration. How the structure of an enzyme can be altered, and how p. H and temperature affect enzyme function. How to name an enzyme, its substrate and products, and then design a controlled experiment to measure the activity of a specific enzyme under varying conditions. How to calculate the rate of reaction.

ENZYMES A catalyst is a chemical agent that speeds up a reaction without being consumed by the reaction An enzyme is a catalytic protein Hydrolysis of sucrose by the enzyme sucrase is an example of an enzyme-catalyzed reaction Even though the reaction is “spontaneous”/exergonic/ releases free energy, it would happen WAY to slow (years) without help from the enzyme due to its very high energy of activation. With the enzyme, the reaction takes seconds.
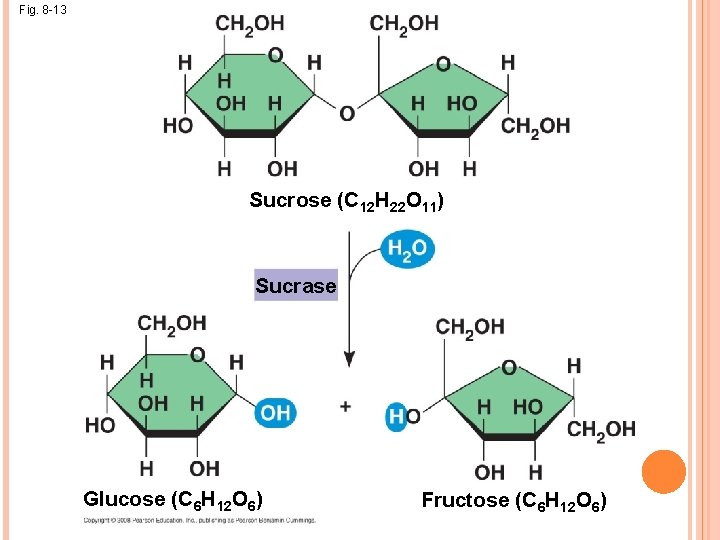
Fig. 8 -13 Sucrose (C 12 H 22 O 11) Sucrase Glucose (C 6 H 12 O 6) Fructose (C 6 H 12 O 6)

THE ACTIVATION ENERGY BARRIER Every chemical reaction between molecules involves bond breaking and bond forming The initial energy needed to start a chemical reaction is called the free energy of activation, or activation energy (EA) (to contort the bonds) Activation energy is often supplied in the form of heat from the surroundings Enzymes catalyze reactions by lowering the EA barrier; they do not affect the change in free energy (∆G)
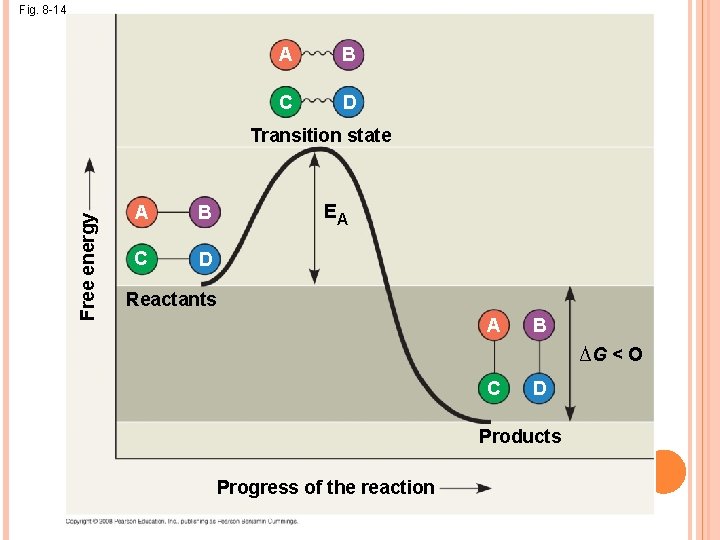
Fig. 8 -14 A B C D Free energy Transition state A B C D EA Reactants A B ∆G < O C D Products Progress of the reaction
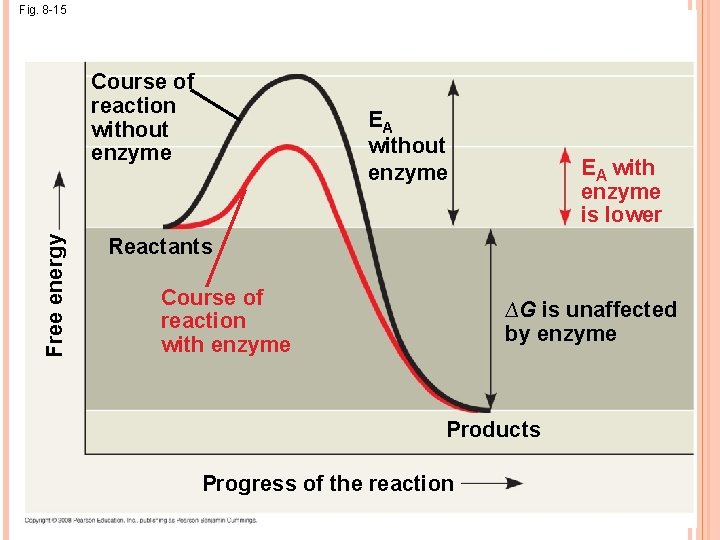
Fig. 8 -15 Free energy Course of reaction without enzyme EA with enzyme is lower Reactants Course of reaction with enzyme ∆G is unaffected by enzyme Products Progress of the reaction

SUBSTRATE SPECIFICITY OF ENZYMES The reactant that an enzyme acts on is called the enzyme’s substrate The enzyme binds to its substrate, forming an enzymesubstrate complex The active site is the region on the enzyme where the substrate binds Induced fit of a substrate brings chemical groups of the active site into positions that enhance their ability to catalyze the reaction Most enzymes are capable of 1000 substrate actions per second!
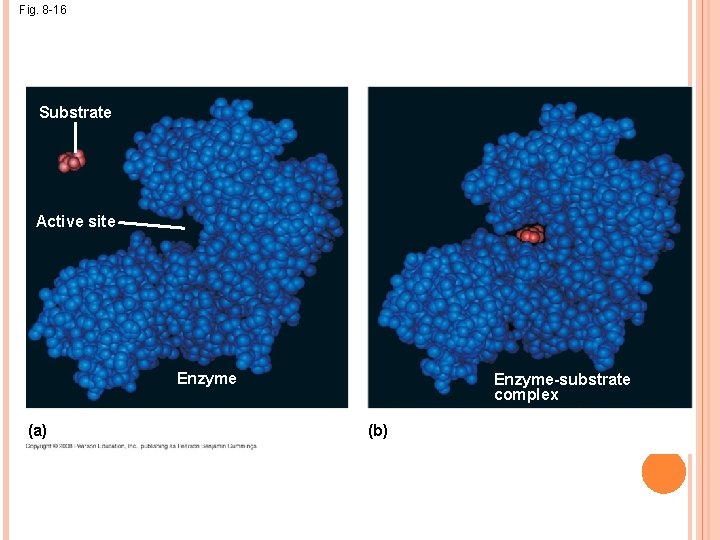
Fig. 8 -16 Substrate Active site Enzyme (a) Enzyme-substrate complex (b)

CATALYSIS IN THE ENZYME’S ACTIVE SITE In an enzymatic reaction, the substrate binds to the active site of the enzyme. Think of four mechanisms which allow the active site to lower an EA barrier…

CATALYSIS IN THE ENZYME’S ACTIVE SITE Four mechanisms which allow the active site to lower an EA barrier: Orienting substrates correctly – providing a place for reactants to find each other Straining substrate bonds – contorting reactants towards transition state through weak H and ionic interactions from the R groups in the protein Providing a favorable microenvironment – ex) acidic conditions via acidic R-groups Covalently bonding to the substrate – this covalent bonding is temporary… it is released in subsequent reactions
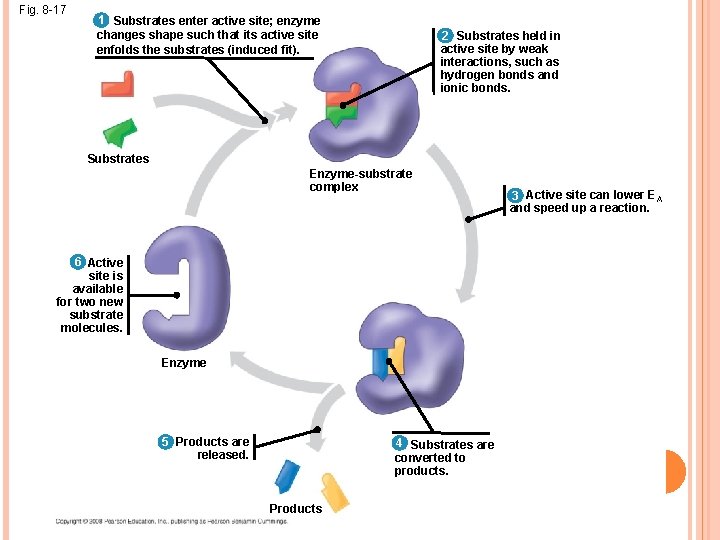
Fig. 8 -17 1 Substrates enter active site; enzyme changes shape such that its active site enfolds the substrates (induced fit). 2 Substrates held in active site by weak interactions, such as hydrogen bonds and ionic bonds. Substrates Enzyme-substrate complex 6 Active site is available for two new substrate molecules. Enzyme 5 Products are released. 4 Substrates are converted to products. Products 3 Active site can lower EA and speed up a reaction.

EFFECTS OF LOCAL CONDITIONS ON ENZYME ACTIVITY An enzyme’s activity can be affected by: Substrate concentration: activity increases with increasing substrate concentration until the saturation point is reached (where all active sites are occupied) General environmental factors, such as temperature and p. H Chemicals that specifically influence the enzyme

EFFECTS OF TEMPERATURE AND PH Each enzyme has an optimal temperature in which it can function Each enzyme has an optimal p. H in which it can function
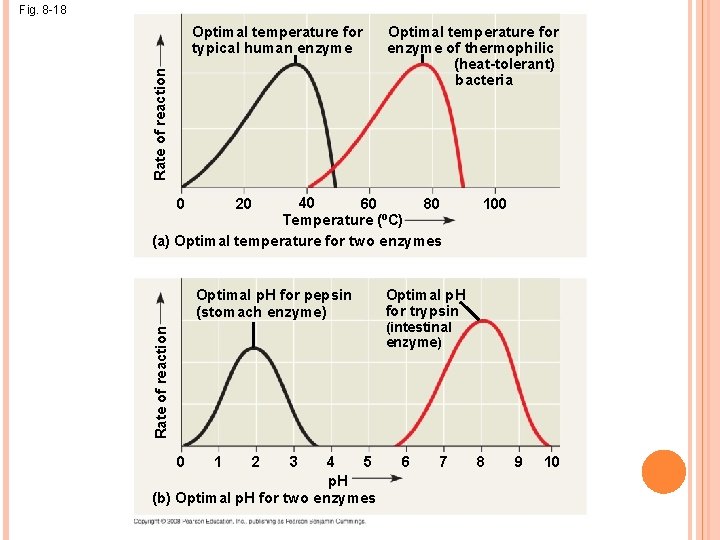
Fig. 8 -18 Rate of reaction Optimal temperature for typical human enzyme Optimal temperature for enzyme of thermophilic (heat-tolerant) bacteria 40 60 80 Temperature (ºC) (a) Optimal temperature for two enzymes 0 20 Rate of reaction Optimal p. H for pepsin (stomach enzyme) 4 5 p. H (b) Optimal p. H for two enzymes 0 1 2 3 100 Optimal p. H for trypsin (intestinal enzyme) 6 7 8 9 10

COFACTORS Cofactors are nonprotein enzyme helpers Cofactors may be inorganic (such as a metal in ionic form) or organic An organic cofactor is called a coenzyme Coenzymes include vitamins

ENZYME INHIBITORS Competitive inhibitors bind to the active site of an enzyme, competing with the substrate Noncompetitive inhibitors bind to another part of an enzyme, causing the enzyme to change shape and making the active site less effective Examples of inhibitors include toxins, poisons, pesticides, and antibiotics
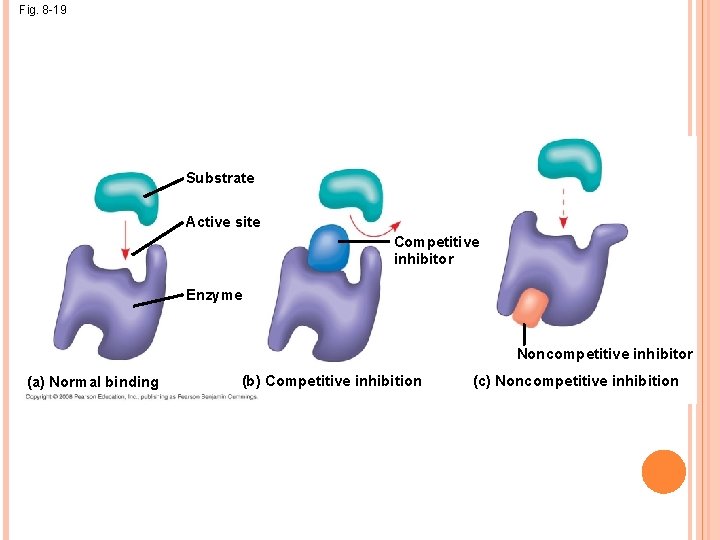
Fig. 8 -19 Substrate Active site Competitive inhibitor Enzyme Noncompetitive inhibitor (a) Normal binding (b) Competitive inhibition (c) Noncompetitive inhibition

REGULATION OF ENZYME ACTIVITY HELPS CONTROL METABOLISM Chemical chaos would result if a cell’s metabolic pathways were not tightly regulated A cell does this by switching on or off the genes that encode specific enzymes or by regulating the activity of enzymes

ALLOSTERIC REGULATION OF ENZYMES Allosteric regulation may either inhibit or stimulate an enzyme’s activity Allosteric regulation occurs when a regulatory molecule binds to a protein at one site and affects the protein’s function at another site

ALLOSTERIC ACTIVATION AND INHIBITION Most allosterically regulated enzymes are made from polypeptide subunits Each enzyme has active and inactive forms The binding of an activator stabilizes the active form of the enzyme The binding of an inhibitor stabilizes the inactive form of the enzyme
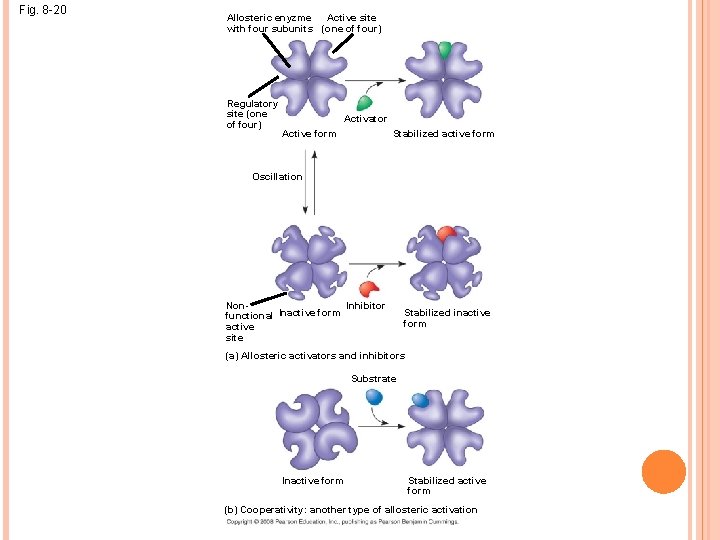
Fig. 8 -20 Active site Allosteric enyzme with four subunits (one of four) Regulatory site (one of four) Activator Active form Stabilized active form Oscillation Non. Inhibitor functional Inactive form active site Stabilized inactive form (a) Allosteric activators and inhibitors Substrate Inactive form Stabilized active form (b) Cooperativity: another type of allosteric activation

Cooperativity is a form of allosteric regulation that can amplify enzyme activity In cooperativity, binding by a substrate to one active site stabilizes favorable conformational changes at all other subunits
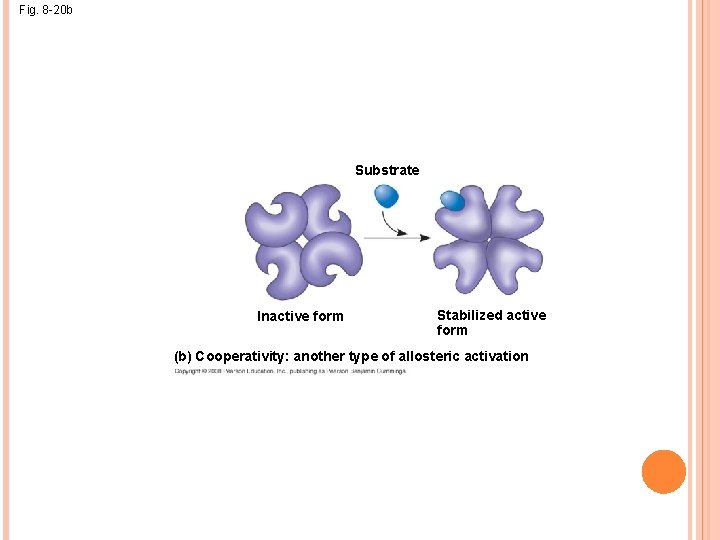
Fig. 8 -20 b Substrate Inactive form Stabilized active form (b) Cooperativity: another type of allosteric activation

IDENTIFICATION OF ALLOSTERIC REGULATORS Allosteric regulators are attractive drug candidates for enzyme regulation Inhibition of proteolytic enzymes called caspases may help management of inappropriate inflammatory responses

FEEDBACK INHIBITION In feedback inhibition, the end product of a metabolic pathway shuts down the pathway Feedback inhibition prevents a cell from wasting chemical resources by synthesizing more product than is needed
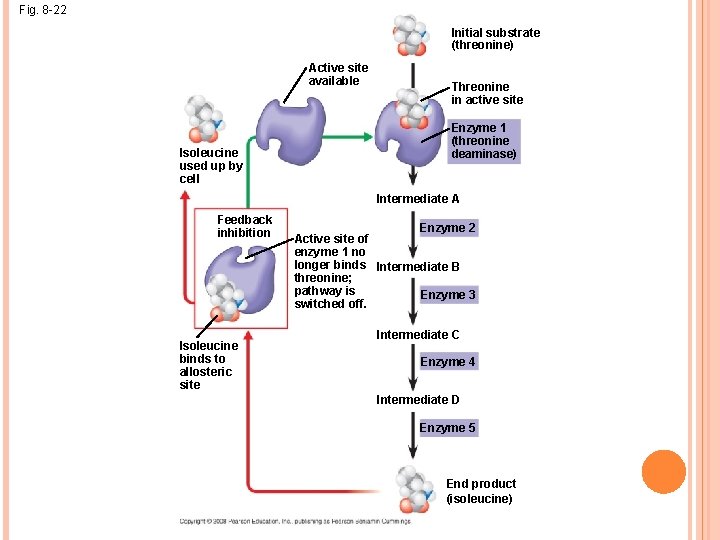
Fig. 8 -22 Initial substrate (threonine) Active site available Isoleucine used up by cell Threonine in active site Enzyme 1 (threonine deaminase) Intermediate A Feedback inhibition Isoleucine binds to allosteric site Enzyme 2 Active site of enzyme 1 no longer binds Intermediate B threonine; pathway is Enzyme 3 switched off. Intermediate C Enzyme 4 Intermediate D Enzyme 5 End product (isoleucine)

SPECIFIC LOCALIZATION OF ENZYMES WITHIN THE CELL Structures within the cell help bring order to metabolic pathways Some enzymes act as structural components of membranes In eukaryotic cells, some enzymes reside in specific organelles; for example, enzymes for cellular respiration are located in mitochondria
- Slides: 29