Concentrations DoNow Make four vocab cards Concentration The

Concentrations Do-Now: Make four vocab cards. Concentration: The mass of solid dissolved in one cubic centimeter of liquid. Solute: The dissolved solid Solvent: The dissolving liquid Solubility: The concentration of a saturated solution.

Standard: �SC. 8. P. 8. 4 Classify and compare substances on the basis of characteristic physical properties that can be demonstrated or measured; for example, density, thermal or electrical conductivity, solubility, magnetic properties, melting and boiling points, and know that these properties are independent of the amount of the sample.

Concentration – Notes on ISN pg: �The amount of “stuff” in a given volume. �Where have we seen this before?

Example: Yesterday’s lab �Orange solid at the beginning: 0. 30 g = 0. 060 g/cm 3 & 0. 30 g = 0. 015 g/cm 3 5. 0 cm 3 20 cm 3 Units are similar to density (g/cm 3), but in concentration, it is not the mass and volume of the same substance. It is the mass of the solid and volume of the liquid.

Confusion? To avoid confusion, concentrations are often given in g/100 cm 3. For example, a concentration of 0. 015 g/cm 3 means that 0. 15 g of a solute is dissolved in 1 cm 3 of water. Therefore, a volume of 100 cm 3 of water will contain 100 x 0. 015 g = 1. 5 g
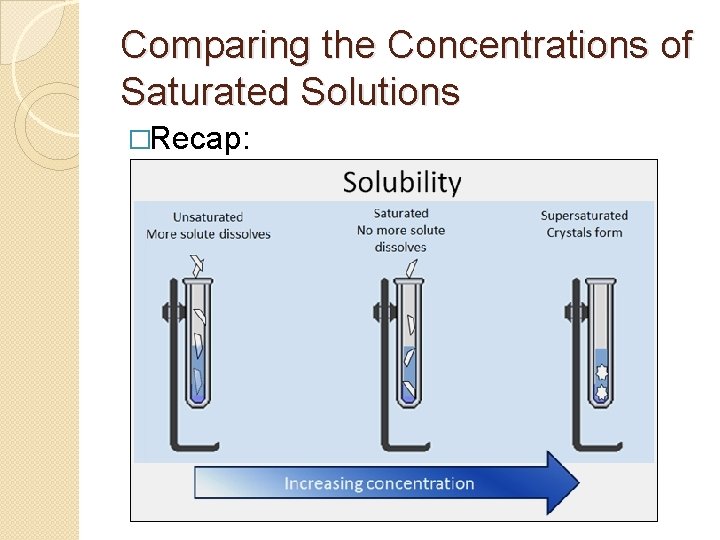
Comparing the Concentrations of Saturated Solutions �Recap:

Concentration �To find the concentration of a saturated solution, you can add the solid a tiny amount at a time and see whether it dissolves. �Better? : Saturate it and decant (separate) the liquid from the saturated solid. �You can find mass of solution, evaporate liquid, and remass dry solid.

Concentration Use what you know: Mass solution – mass solid = mass of water Then, using density of water (1 g/cm 3), we can calculate the volume of water used. This will give you information needed for calculate the concentration of the saturated solution.

Dry Lab: �Purpose: To calculate the concentration of an unknown solution. �Materials: ◦ ◦ ◦ 5 g solid 5 cm 3 water Evaporating dish Scale Stopper

Procedure 1. 2. 3. 4. 5. 6. Add solid into liquid. Shake. Mass the evaporating dish. Decant almost all of the saturated solution into it. Find mass of evaporating dish and solution. Slowly evaporate the solution to dryness. Find mass of remaining solid.

Data & Questions: Mass of Evaporating Dish (g) • • Mass of Dish and Solution (g) Mass of Dish and Solid (g) 23. 2 g 34. 2 g 26. 0 What is the mass of the solid remaining in the dish? What was the mass of the water it was dissolved in? (This is the water you evaporated. ) What was the volume of the water? What is the concentration of the saturated solution? How would the calculate concentration be affected if some of the undissolved solid had been added to the dish? How would the calculate concentration be affected if some of the solid had splattered out of the evaporating dish? What is the concentration of each of the saturated
- Slides: 11