Computeraided Drug Design Ligand Structure database In silico

Computer-aided Drug Design Ligand Structure database (In silico) Course: Drug Design Course code: 0510412 Dr. Balakumar Chandrasekaran Dr. Bilal Al-Jaidi Assistant Professors, Faculty of Pharmacy, Philadelphia University-Jordan

Learning Outcomes At the end of this lesson students will be able to Ø Outline the entire process involved in the computer-aided drug design methodologies. Ø Describe about molecular mechanics and quantum mechanics. Ø Explain structure-based drug design including the molecular docking technique. Ø Describe about QSAR and pharmacophore modeling in ligand-based drug design. Ø Explain the steric-electronic modifications in de novo drug design method.

DRUG Medicine or substance which has a physiological effect when administered into the body. Drug acting on different targets • Enzyme - inhibitors • Receptor - agonists or antagonists • Ion channels - blockers • Transporter - update inhibitors • DNA – blockers.
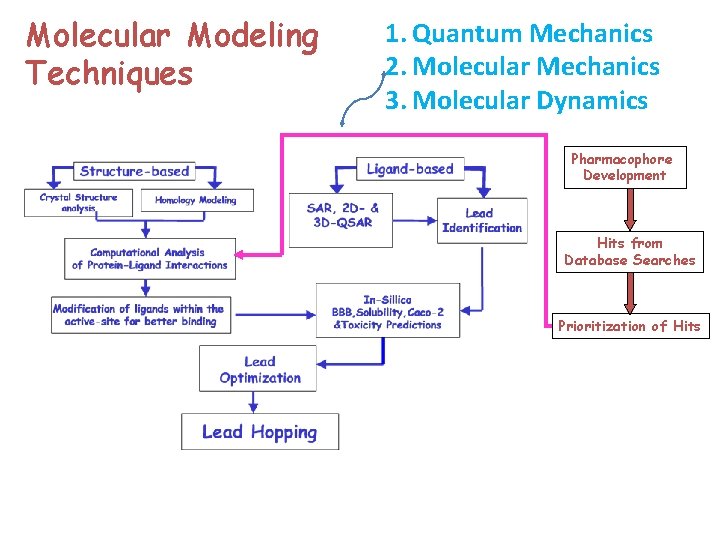
Molecular Modeling Techniques 1. Quantum Mechanics 2. Molecular Mechanics 3. Molecular Dynamics Pharmacophore Development Hits from Database Searches Prioritization of Hits
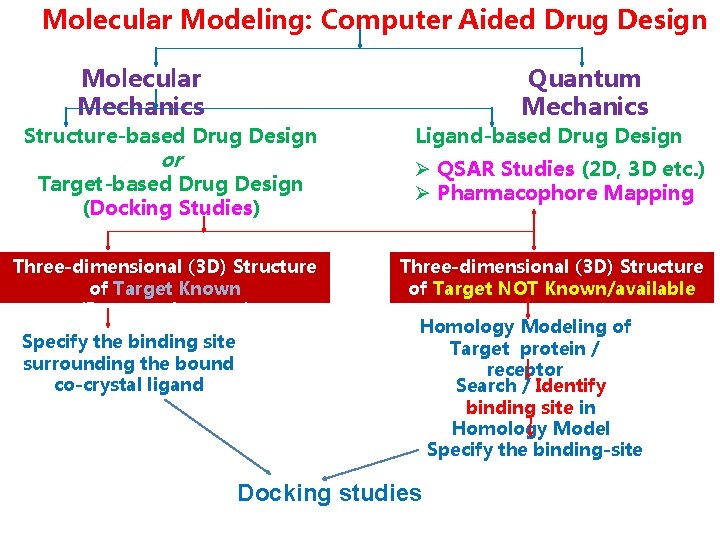
Molecular Modeling: Computer Aided Drug Design Molecular Mechanics Structure-based Drug Design or Target-based Drug Design (Docking Studies) Three-dimensional (3 D) Structure of Target Known (Receptor/enzyme) Specify the binding site surrounding the bound co-crystal ligand Quantum Mechanics Ligand-based Drug Design Ø QSAR Studies (2 D, 3 D etc. ) Ø Pharmacophore Mapping Three-dimensional (3 D) Structure of Target NOT Known/available Homology Modeling of Target protein / receptor Search / Identify binding site in Homology Model Specify the binding-site Docking studies

Molecular Mechanics It is the more popular method used to obtain molecular models as Ø it is simpler to use and Ø requires considerably less computing time to produce a model. It assumes that the total POTENTIAL ENERGY (ETotal) of a molecule is given by the sum of all the energies of the attractive and repulsive forces between the atoms in the structure. These energies are calculated using a Mechanical Model in which Ø atoms are represented by balls whose mass is proportional to their relative atomic masses Ø atoms are joined by mechanical springs corresponding to the covalent bonds in the structure.
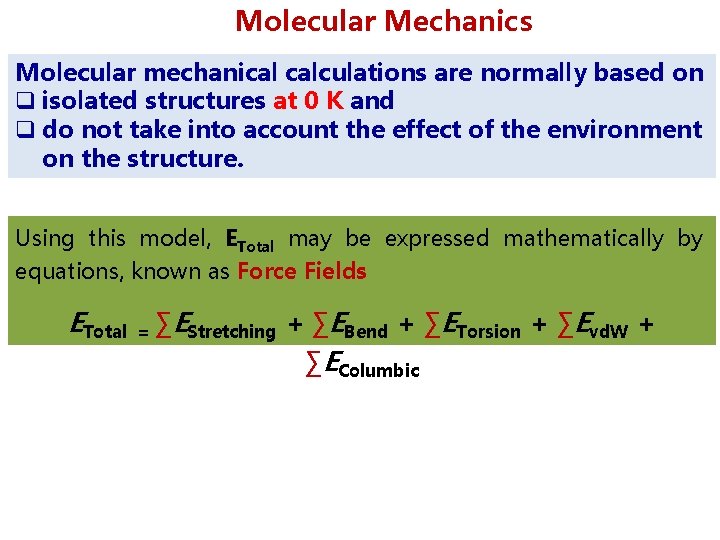
Molecular Mechanics Molecular mechanical calculations are normally based on q isolated structures at 0 K and q do not take into account the effect of the environment on the structure. Using this model, ETotal may be expressed mathematically by equations, known as Force Fields ETotal = ∑EStretching + ∑EBend + ∑ETorsion + ∑Evd. W + ∑EColumbic

Quantum Mechanics Ø It does not require the use of parameters similar to those used in molecular mechanics. Ø It is based on the realisation that electrons and all material particles exhibit wave-like properties. Ø Mathematics of wave motions are applied to electrons, atomic and molecular structure. Ø The basis of these calculations is the Schrodinger wave equation: Hѱ=Eѱ ѱ = time dependent wave function, which defines the state (nature and properties) of a system. E ѱ = represents the total potential and kinetic energy of all the particles (nuclei and electrons) in the structure H = Hamiltonium operator acting on the wave function ѱ

Quantum Mechanics Applications: Quantum mechanics is useful Ø for calculating the values of ionisation potentials, electron affinities, heats of formation, dipole moments and other physical properties of atoms and molecules. Ø to calculate the relative probabilities of finding electrons (the electron density) in a structure. This makes it possible to determine the most likely points at which a structure will react with electrophiles and nucleophiles. Ø to assess the nature of the binding of a possible drug to a target site.

Molecular Mechanics Vs Quantum Mechanics A. Quantum mechanical methods Ø deal with electrons in a system, and Ø the calculations are very MUCH time consuming. B. Molecular mechanics (force-field methods) on the other hand Ø ignore the electronic motions and Ø calculates the energy of the system as a function of the nuclear position only. Ø the calculations are very LESS time consuming. However, molecular mechanics cannot provide answers

Drug Design Strategies Based on Molecular Mechanics Target-Based Ligand-based Target Protein Known Docking Lead Pharmacophore & 3 D-QSAR Study Ligands Target Protein Unknown Ligands Ligand Structure database (In silico) 11

Structure-based Drug Design • Structure-based drug design: – The macromolecular target can be isolated and crystallized…then the structure will be determined using X-ray crystallography. – This structure will not give information about the binding site. – The co-crystal structure (structure of protein with the inhibitor inside) is better (WHY? ): • Where is the active site? • The distance between inhibitor and binding site boundaries. • The possible bonds between inhibitor and binding site.

How to use co-crystal structure in drug design • First the inhibitor will be removed from the active site (in silico). • The enzyme structure will be minimized to get the lowest energy state. • Then lead compounds will be inserted (docked) into the active site to see how they fit. • Best fit compounds will be synthesized and tested for activity.

Structure-based (target-based) Drug Design Ø It is used to design a new drug molecule based on the knowledge of the three-dimensional (3 D) structure of the biological target. Active site / binding site
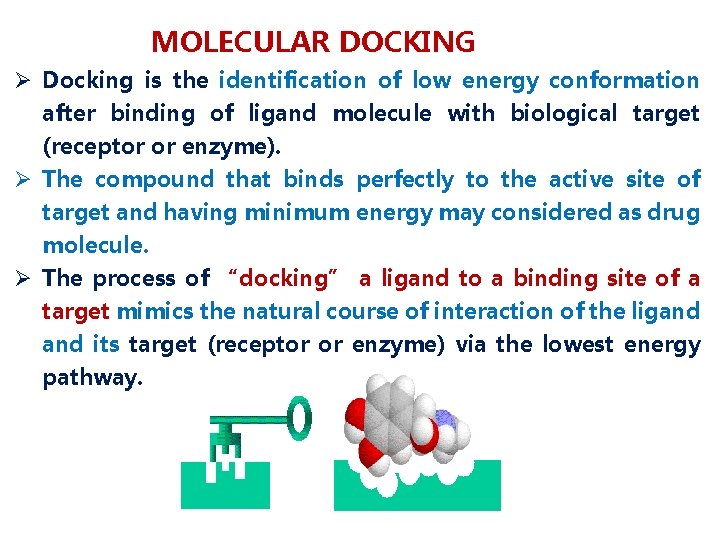
MOLECULAR DOCKING Ø Docking is the identification of low energy conformation after binding of ligand molecule with biological target (receptor or enzyme). Ø The compound that binds perfectly to the active site of target and having minimum energy may considered as drug molecule. Ø The process of “docking” a ligand to a binding site of a target mimics the natural course of interaction of the ligand its target (receptor or enzyme) via the lowest energy pathway.

WHY DOCKING? Ø Drug work by interacting with biological target (receptor/enzyme) Ø Docking helps to decide a candidate drug will interact appropriately with a target protein? Ø Examine binding model of known ligands to suggest modification. Ø Screen databases of 3 D structure to find novel ligands. Ø Drug interaction with receptor can be as an agonist or an antagonist.
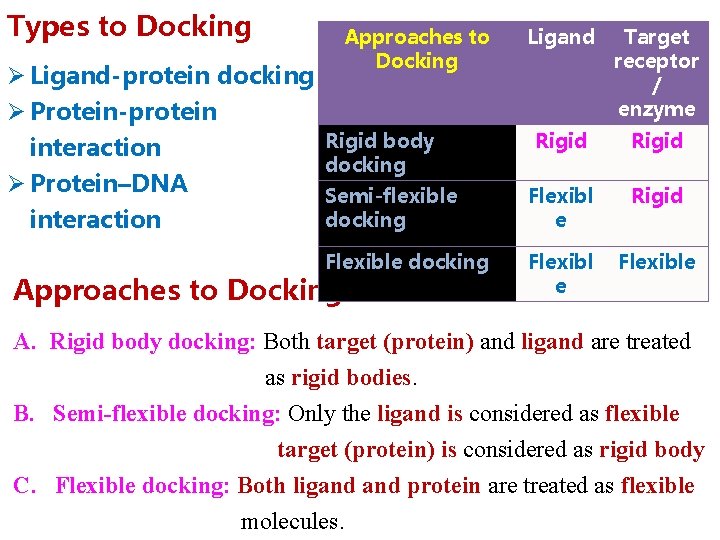
Types to Docking Ø Ligand-protein docking Ø Protein-protein interaction Ø Protein–DNA interaction Approaches to Docking Ligand Target receptor / enzyme Rigid Semi-flexible docking Flexibl e Rigid Flexible docking Flexibl e Flexible Rigid body docking Approaches to Docking A. Rigid body docking: Both target (protein) and ligand are treated as rigid bodies. B. Semi-flexible docking: Only the ligand is considered as flexible target (protein) is considered as rigid body C. Flexible docking: Both ligand protein are treated as flexible molecules.

Scoring Functions During the docking process, they serve as fitness functions in the optimization of the placement of the ligand. When the docking is completed, the scoring function is used to rank each ligand in the database. Some common scoring functions are: 1. Force-field methods 2. Empirical free energy scoring functions 3. Knowledge-based potential of mean

Docking Softwares Free for Academics: • DOCK • Auto. DOCK • Surflex • FRED • e. Hits Commercial: • GOLD/Glide/Flex. X/Discovery studio-CDOCKER.

If you do not have the crystal structure of your target enzyme • Three options: – Use recombinant DNA technology to produce the enzyme using bacterial cell. – Use the homologue of this enzyme from other organism such as bacteria or parasite (homology modeling/comparative modeling).

Ligand-based Drug Design • Here the crystal structure of the target enzyme or receptor is not available. • But their ligands are well defined and characterized. • This method not involves molecular docking or homology modeling methods. • This method works based on the concept of ‘similar chemical structures have similar chemical

Ligand-based Drug Design This method will v Begin with biologically active compounds. v Describe what chemistry those compounds have in common. v Few new compounds that match this description. v Compounds that match theprocess description will Initial two steps involving the called also building’ be active. while the final two steps are ‘model known as ‘database screening’.
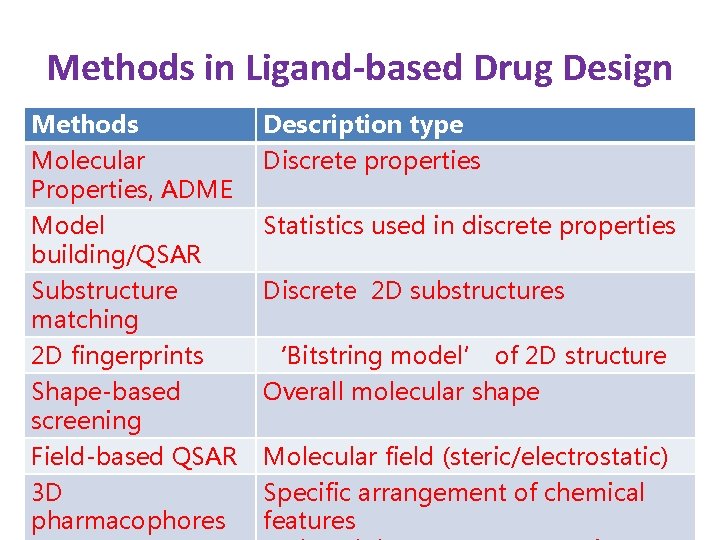
Methods in Ligand-based Drug Design Methods Molecular Properties, ADME Description type Discrete properties Model building/QSAR Statistics used in discrete properties Substructure matching 2 D fingerprints Shape-based screening Field-based QSAR 3 D pharmacophores Discrete 2 D substructures ‘Bitstring model’ of 2 D structure Overall molecular shape Molecular field (steric/electrostatic) Specific arrangement of chemical features

(Quantitative Structure-Activity QSAR is a mathematical relationship between a biological Relationship) activity of a molecular system and its geometric and chemical characteristics. A general formula for a quantitative structure-activity relationship (QSAR) can be given by the following: Activity = f (molecular or fragmental properties) QSAR attempts to find consistent relationship between biological activity and molecular properties, so that these “rules” can be used to evaluate the activity of new

QSAR (Quantitative Structure-Activity Relationship) ü QSAR is a statistical approach that attempts to relate physico-chemical properties of molecules to their biological activities. ü Various descriptors like molecular weight, number of rotatable bonds, Log. P etc. are commonly used in QSAR. Example: Hydrophobicity Vs Biological Activity • Activity of drugs is often related to P e. g. binding of drugs to serum albumin (straight line - limited range of log P) Log (1/C) . . 0. 78 3. 82 Log æè 1 C öø = mlog. P Log P +C
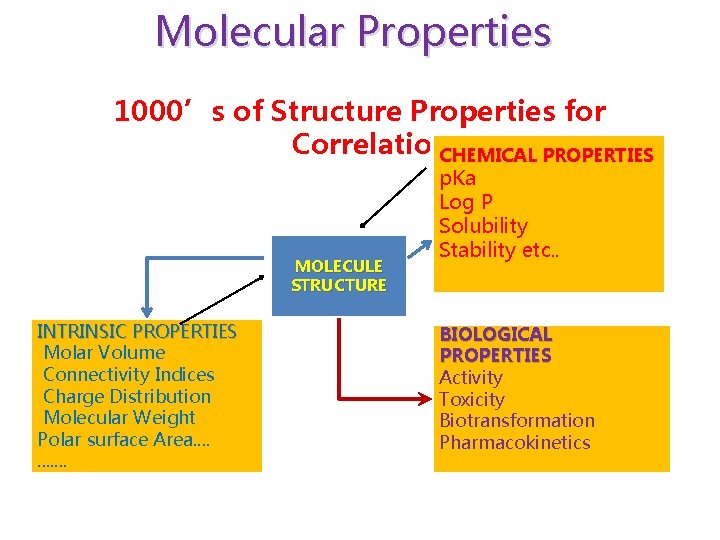
Molecular Properties 1000’s of Structure Properties for Correlation. CHEMICAL PROPERTIES MOLECULE STRUCTURE INTRINSIC PROPERTIES Molar Volume Connectivity Indices Charge Distribution Molecular Weight Polar surface Area. . . p. Ka Log P Solubility Stability etc. . BIOLOGICAL PROPERTIES Activity Toxicity Biotransformation Pharmacokinetics
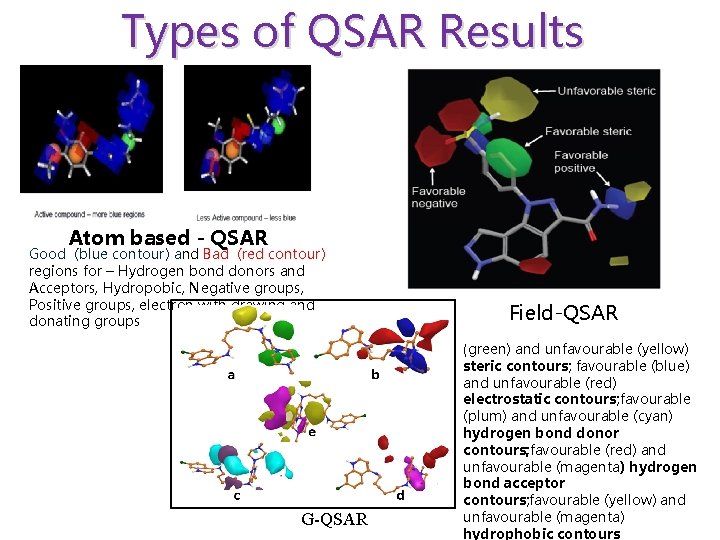
Types of QSAR Results Atom based - QSAR Good (blue contour) and Bad (red contour) regions for – Hydrogen bond donors and Acceptors, Hydropobic, Negative groups, Positive groups, electron with drawing and donating groups G-QSAR Field-QSAR (green) and unfavourable (yellow) steric contours; favourable (blue) and unfavourable (red) electrostatic contours; favourable (plum) and unfavourable (cyan) hydrogen bond donor contours; favourable (red) and unfavourable (magenta) hydrogen bond acceptor contours; favourable (yellow) and unfavourable (magenta) hydrophobic contours
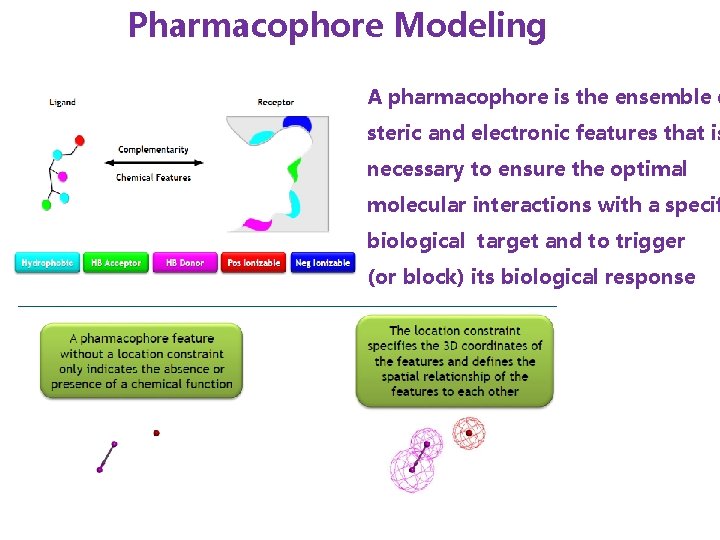
Pharmacophore Modeling A pharmacophore is the ensemble o steric and electronic features that is necessary to ensure the optimal molecular interactions with a specif biological target and to trigger (or block) its biological response
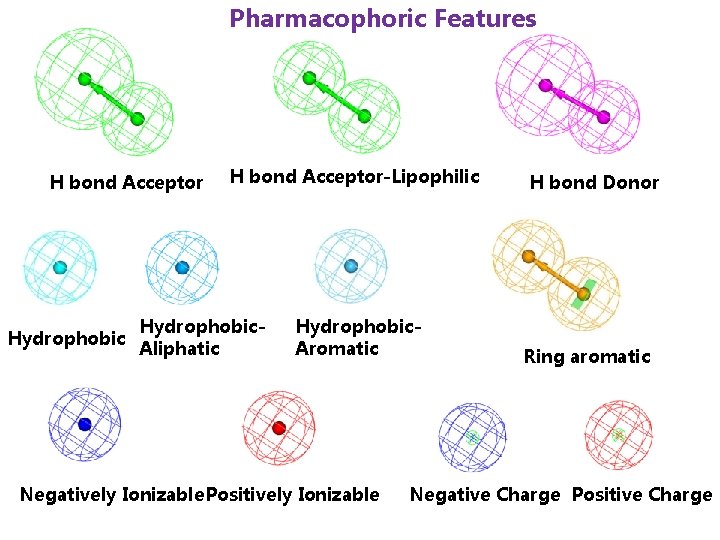
Pharmacophoric Features H bond Acceptor Hydrophobic H bond Acceptor-Lipophilic Hydrophobic. Aliphatic Hydrophobic. Aromatic Negatively Ionizable Positively Ionizable H bond Donor Ring aromatic Negative Charge Positive Charge
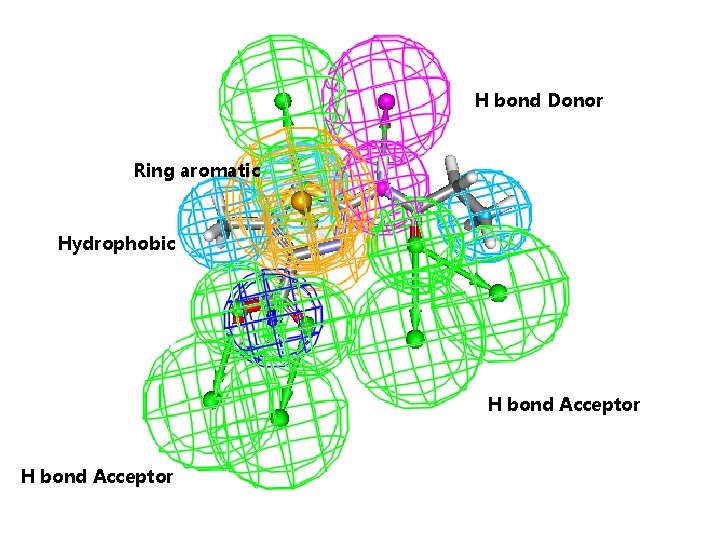
Pharmacophore H-bonding donor H bond Donor Ring aromatic Hydrophobic H-bonding acceptor H H-bonding bond Acceptor H bond Acceptor

GENERATION of a pharmacophore model
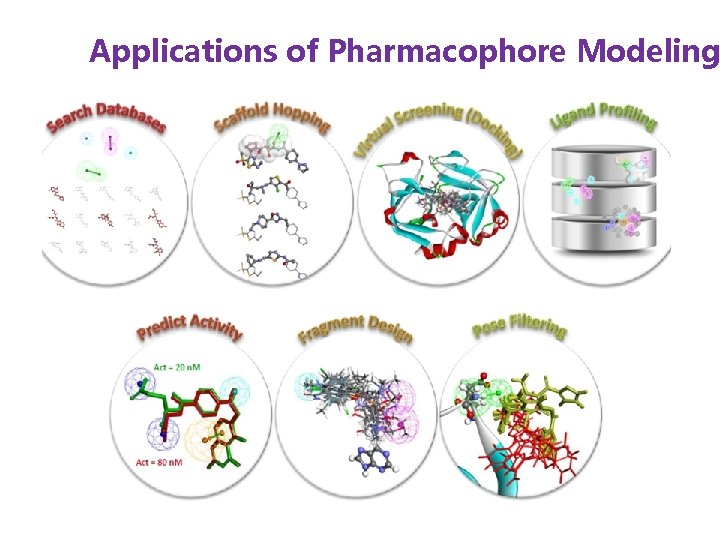
Applications of Pharmacophore Modeling

De-novo Drug Design • Involves the design of a novel drug structure based on a knowledge of the binding site alone. – In this approach, you must have good supply of enzyme. – Sometimes the bacterial version of enzyme will be used (obtained by recombinant DNA technology). – The major drawback of this, is the bacterial version is not identical to the human enzyme…. But it can be considered very similar.
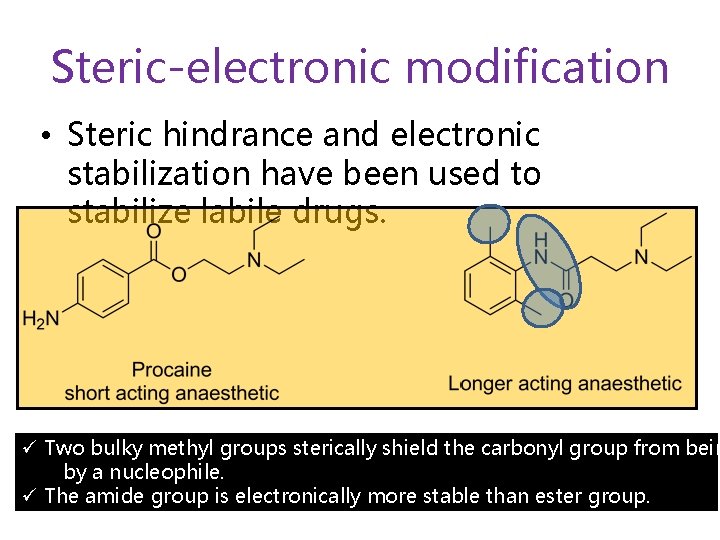
Steric-electronic modification • Steric hindrance and electronic stabilization have been used to stabilize labile drugs. ü Two bulky methyl groups sterically shield the carbonyl group from bein by a nucleophile. ü The amide group is electronically more stable than ester group.
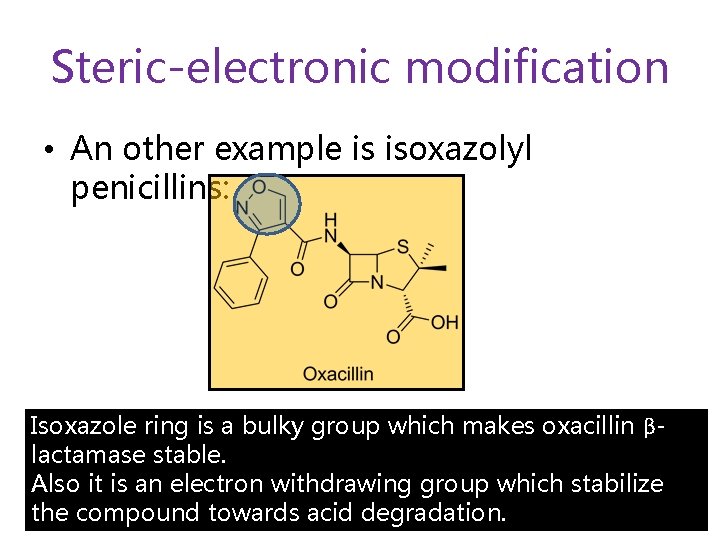
Steric-electronic modification • An other example is isoxazolyl penicillins: Isoxazole ring is a bulky group which makes oxacillin βlactamase stable. Also it is an electron withdrawing group which stabilize the compound towards acid degradation.
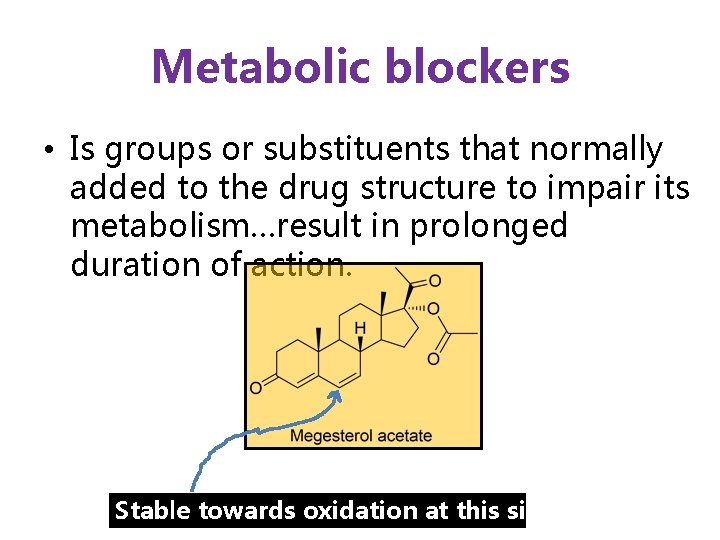
Metabolic blockers • Is groups or substituents that normally added to the drug structure to impair its metabolism…result in prolonged duration of action. Stable towards oxidation at this site
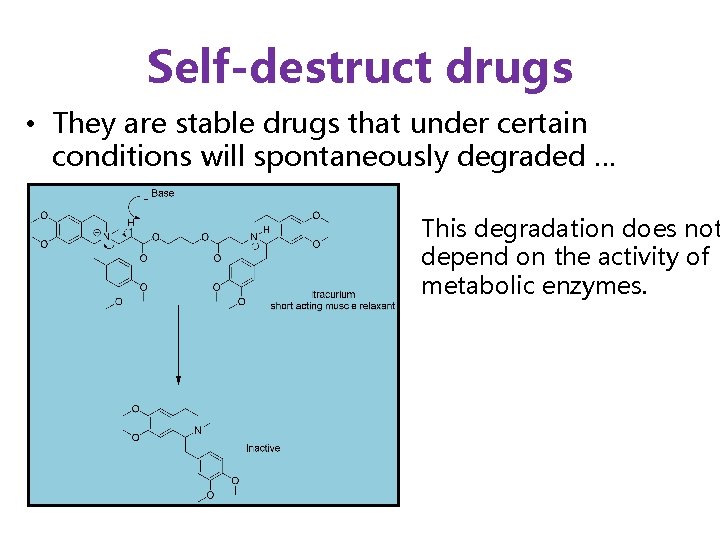
Self-destruct drugs • They are stable drugs that under certain conditions will spontaneously degraded … This degradation does not depend on the activity of metabolic enzymes.

Sleeping drugs • An inactive drug which is only converted to the active form by external stimuli such as light, heat, … • Example: Anticancer porphyrin drugs… large compound that will pass through the leaky membrane into the tumor cells… then the site of tumor will be exposed to red-laser beam to activate the drug…form a reactive oxygen radicals that will attack DNA and membranes.

Sentry Drugs • An other drug is co-administered with the principal drug to guard or assist it. • Examples: – Clavulanic acid + Amoxicillin. – Kaletra (combination of Ritonavir and Lopinavir): Ritonavir is a potent CYP 450 inhibitors as well as a good antiviral agent…prevent Lopinavir oxidation. – The combination of Carbidopa and Levodopa.

Recommended Books 1. The organic chemistry of drug design by Richard B. Silverman. Second edition, Elsevier, 2004. 2. An introduction to Medicinal Chemistry by Graham L. Patrick. Fourth edition, Oxford, 2009. 42

- Slides: 41