Computational Thermodynamics ByeongJoo Lee Computational Materials Science Engineering

Computational Thermodynamics Byeong-Joo Lee Computational Materials Science & Engineering Lab. Pohang University of Science & Technology Byeong-Joo Lee www. postech. ac. kr/~calphad
R&D in Materials Science and Engineering Structure Evolution Process Condition Materials Property Research Type I : experiments first, then thinking Research Type II: think first, then do experiments Byeong-Joo Lee www. postech. ac. kr/~calphad

Thermodynamic Modelling Byeong-Joo Lee www. postech. ac. kr/~calphad
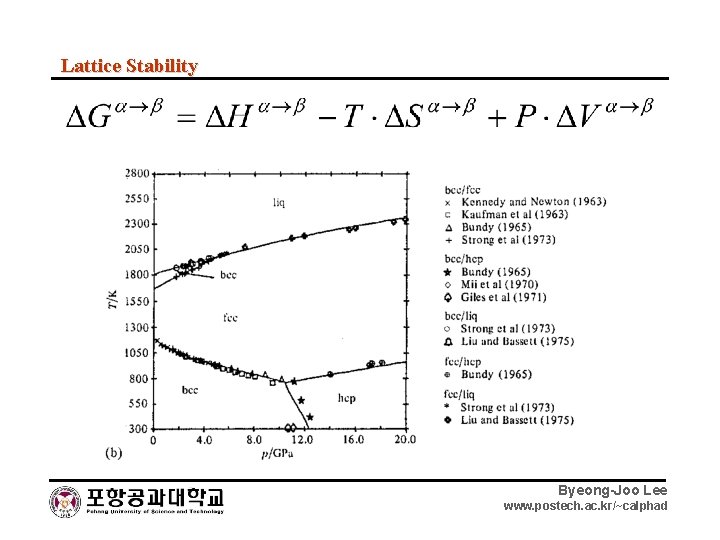
Lattice Stability Byeong-Joo Lee www. postech. ac. kr/~calphad

Regular Solution vs. Quasi-Chemical Model Byeong-Joo Lee www. postech. ac. kr/~calphad
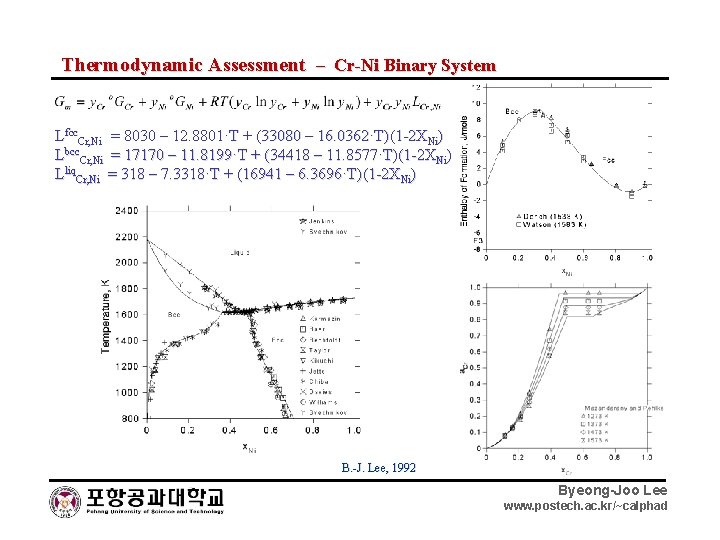
Thermodynamic Assessment – Cr-Ni Binary System Lfcc. Cr, Ni = 8030 – 12. 8801·T + (33080 – 16. 0362·T)(1 -2 XNi) Lbcc. Cr, Ni = 17170 – 11. 8199·T + (34418 – 11. 8577·T)(1 -2 XNi) Lliq. Cr, Ni = 318 – 7. 3318·T + (16941 – 6. 3696·T)(1 -2 XNi) B. -J. Lee, 1992 Byeong-Joo Lee www. postech. ac. kr/~calphad
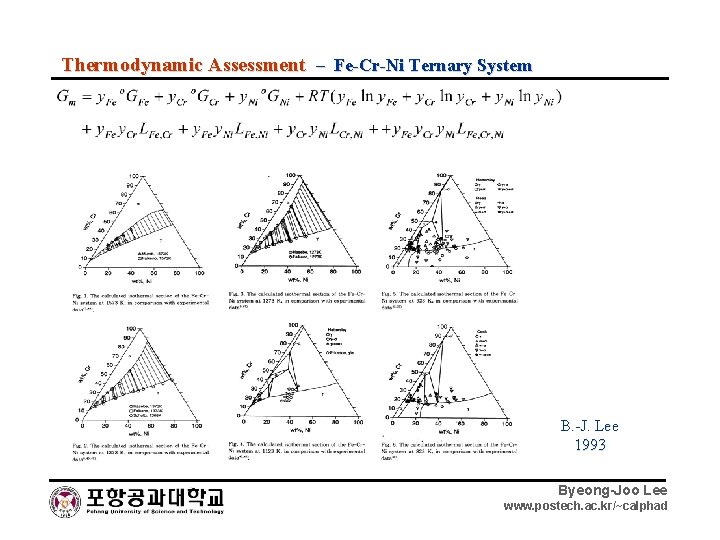
Thermodynamic Assessment – Fe-Cr-Ni Ternary System B. -J. Lee 1993 Byeong-Joo Lee www. postech. ac. kr/~calphad

Thermodynamic Parameters (Fe, Cr, Mo)(Va, B, C, N) Byeong-Joo Lee www. postech. ac. kr/~calphad

Thermodynamic Modeling – Gibbs Energy For a Phase with Formula Unit, (M 1, M 2, …, Mi, …)a(Va, C, N)c Byeong-Joo Lee www. postech. ac. kr/~calphad

Thermodynamic Modeling – Gibbs Energy For a Phase with Formula Unit, (M 1, M 2, …, Mi, …)a(Va, C, N)c Byeong-Joo Lee www. postech. ac. kr/~calphad

Thermodynamic Database for Steels – TCFe 2000 → TCFe 2 → TCFe 3 Fe-Cr-Ni-Mo-Mn-Si-C-N +Nb-Ti-V-W-Al-Co-Cu-B-O-P-S 8 C 2 = 28 Binary Systems 8 C 3 = 56 Ternary Systems 19 C 2 = 171 Binary Systems 19 C 3 = 969 Ternary Systems Byeong-Joo Lee www. postech. ac. kr/~calphad
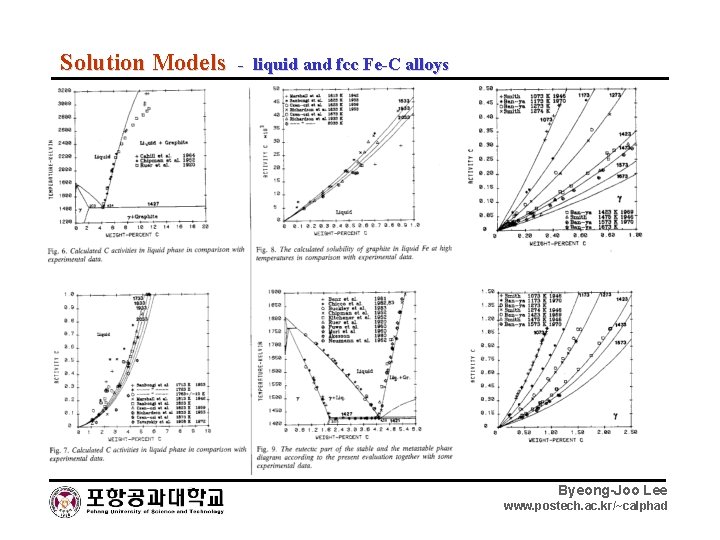
Solution Models - liquid and fcc Fe-C alloys Byeong-Joo Lee www. postech. ac. kr/~calphad

Solution Model - liquid and fcc Fe-C alloys fcc : (Fe)1(Va, C) 1 Liquid : (Fe, C) Ternary (Fe, Mn)a(Va, C) c Byeong-Joo Lee www. postech. ac. kr/~calphad

Applications of Computational Thermodynamics Byeong-Joo Lee www. postech. ac. kr/~calphad
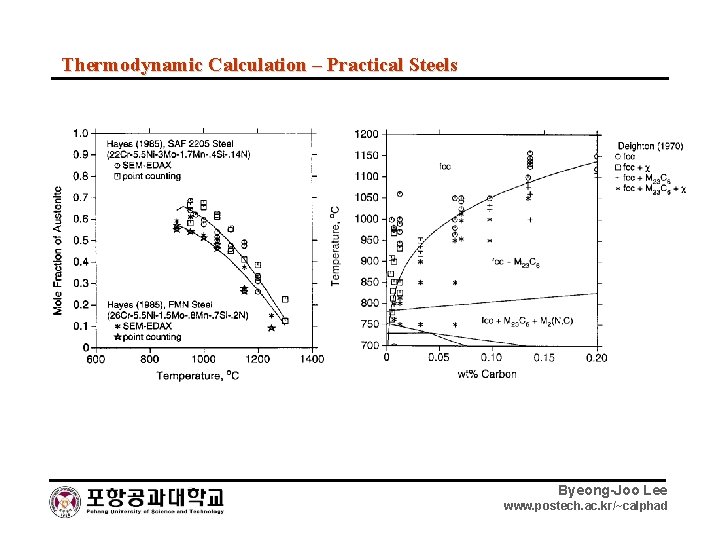
Thermodynamic Calculation – Practical Steels Byeong-Joo Lee www. postech. ac. kr/~calphad
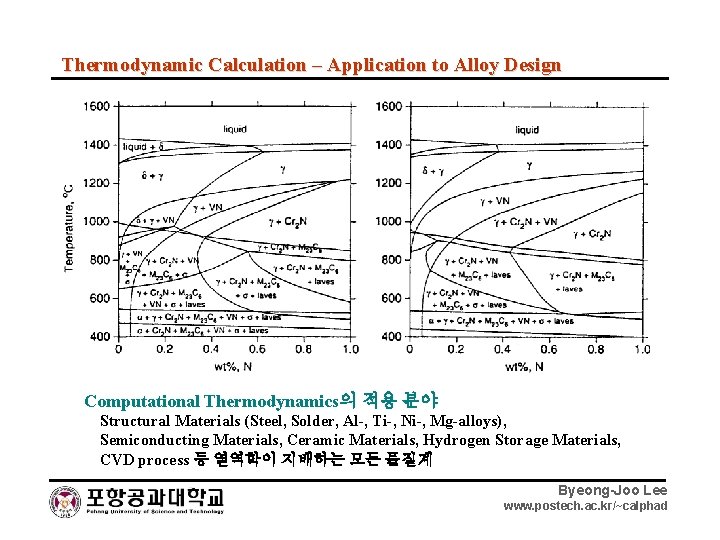
Thermodynamic Calculation – Application to Alloy Design Computational Thermodynamics의 적용 분야 Structural Materials (Steel, Solder, Al-, Ti-, Ni-, Mg-alloys), Semiconducting Materials, Ceramic Materials, Hydrogen Storage Materials, CVD process 등 열역학이 지배하는 모든 물질계 Byeong-Joo Lee www. postech. ac. kr/~calphad
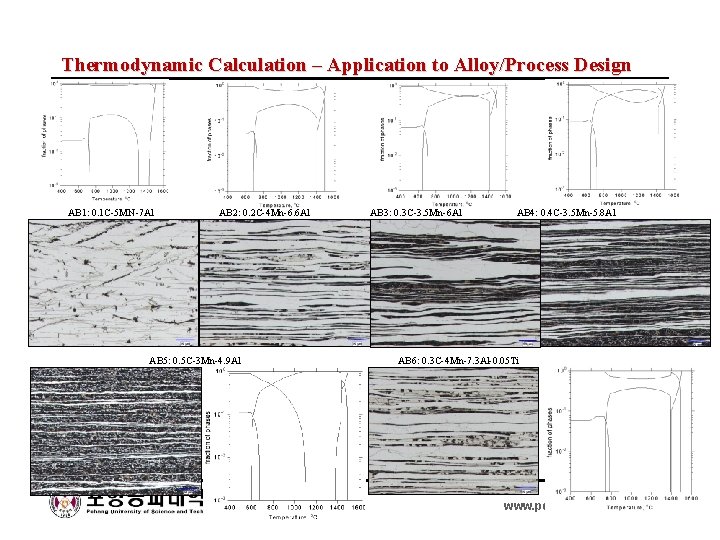
Thermodynamic Calculation – Application to Alloy/Process Design AB 1: 0. 1 C-5 MN-7 Al AB 2: 0. 2 C-4 Mn-6. 6 Al AB 3: 0. 3 C-3. 5 Mn-6 Al AB 4: 0. 4 C-3. 5 Mn-5. 8 Al AB 5: 0. 5 C-3 Mn-4. 9 Al AB 6: 0. 3 C-4 Mn-7. 3 Al-0. 05 Ti Byeong-Joo Lee www. postech. ac. kr/~calphad

Thermodynamics Assessment - Na-Al-H system Byeong-Joo Lee www. postech. ac. kr/~calphad
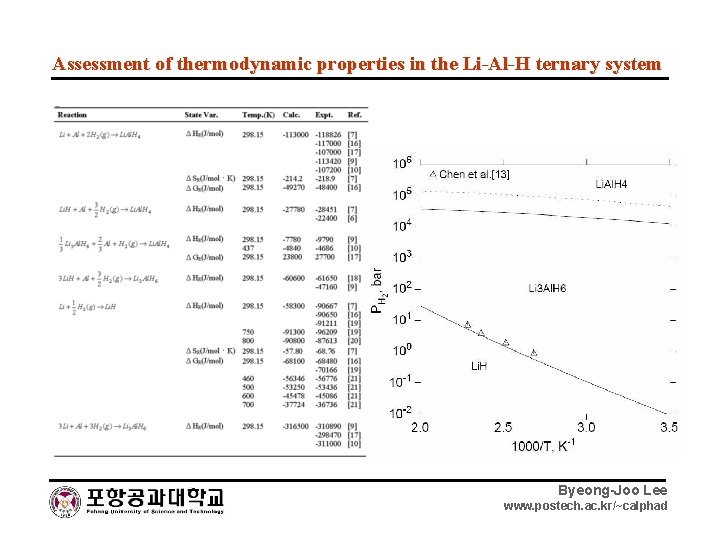
Assessment of thermodynamic properties in the Li-Al-H ternary system Byeong-Joo Lee www. postech. ac. kr/~calphad
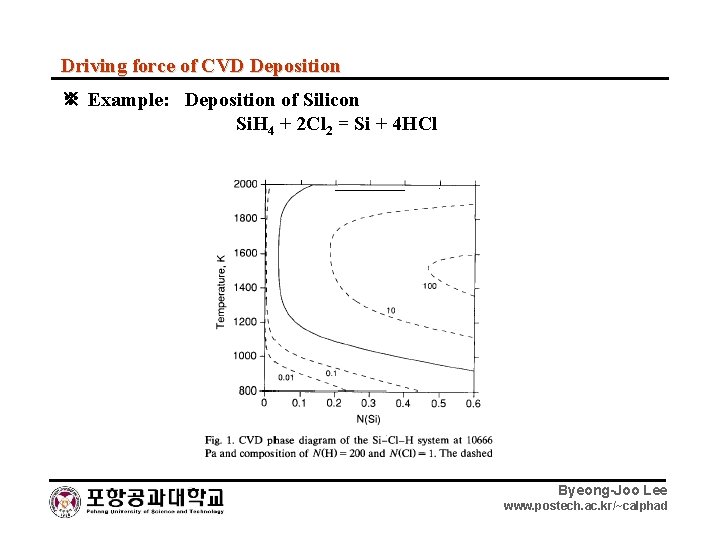
Driving force of CVD Deposition ※ Example: Deposition of Silicon Si. H 4 + 2 Cl 2 = Si + 4 HCl Byeong-Joo Lee www. postech. ac. kr/~calphad
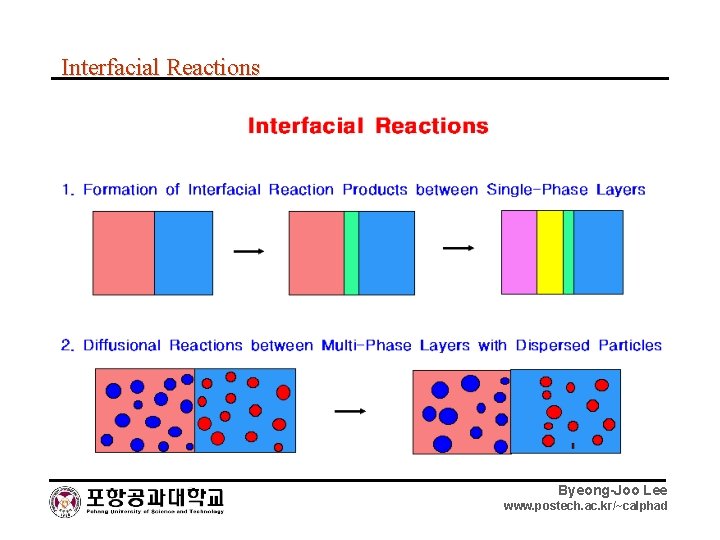
Interfacial Reactions Byeong-Joo Lee www. postech. ac. kr/~calphad

Interfacial Reaction between Cu and Various Solder -Experimental Observation ▶ Cu/Sn : Cu 6 Sn 5 ▶ Cu/Sn-Pb eutectic : Cu 6 Sn 5 ▶ Cu/Sn-Ag eutectic : Cu 6 Sn 5 ▶ Cu/Sn-Zn eutectic : Cu. Zn_γ ▶ Cu/Sn-In eutectic : Cu 2(Sn, In) or Cu 2 In 3 Sn Byeong-Joo Lee www. postech. ac. kr/~calphad
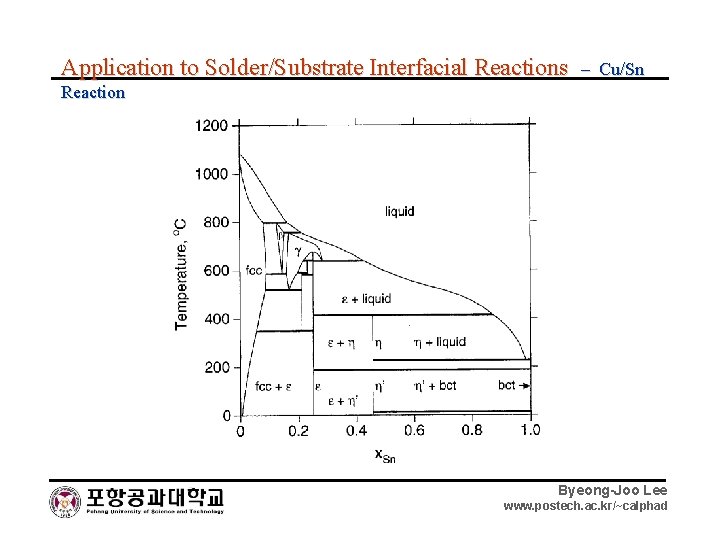
Application to Solder/Substrate Interfacial Reactions – Cu/Sn Reaction Byeong-Joo Lee www. postech. ac. kr/~calphad
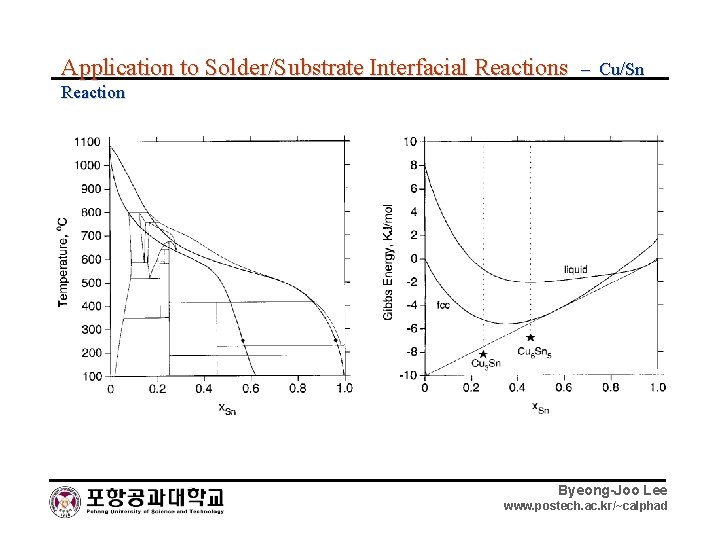
Application to Solder/Substrate Interfacial Reactions – Cu/Sn Reaction Byeong-Joo Lee www. postech. ac. kr/~calphad
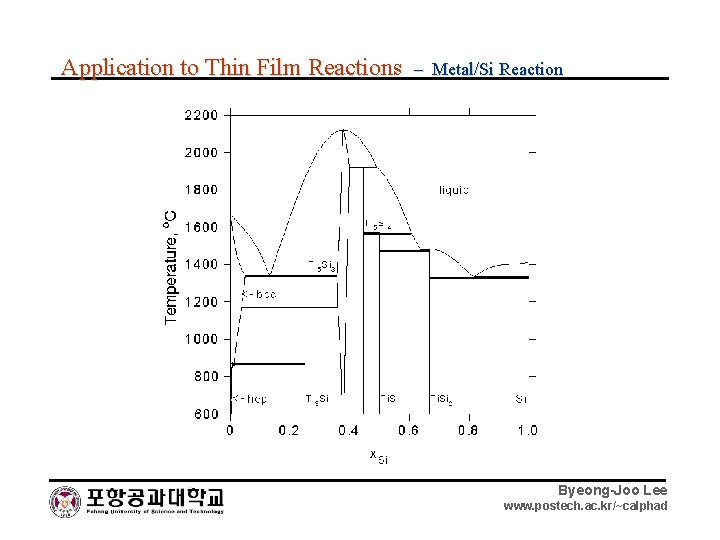
Application to Thin Film Reactions – Metal/Si Reaction Byeong-Joo Lee www. postech. ac. kr/~calphad
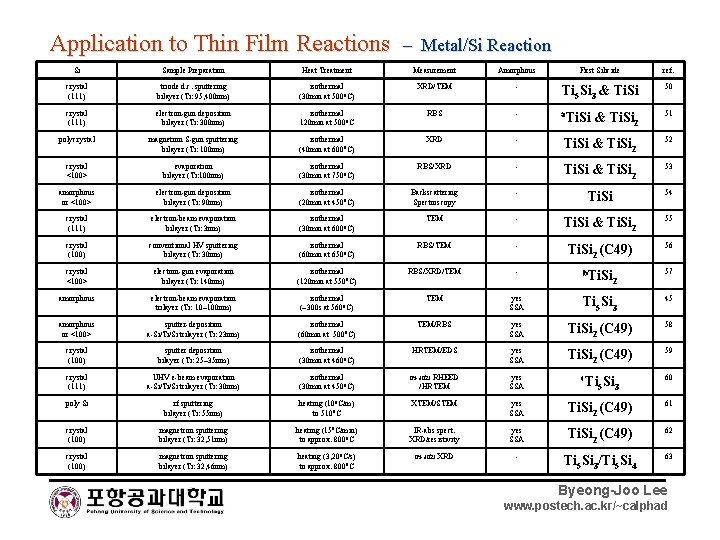
Application to Thin Film Reactions – Metal/Si Reaction Si Sample Preparation Heat Treatment Measurement Amorphous First Silicide ref. crystal (111) triode d. c. sputtering bilayer (Ti: 95, 400 nm) isothermal (30 min at 500 o. C) XRD/TEM - Ti 5 Si 3 & Ti. Si 50 crystal (111) electron-gun deposition bilayer (Ti: 300 nm) isothermal 120 min at 500 o. C RBS - a. Ti. Si & Ti. Si 2 51 polycrystal magnetron S-gun sputtering bilayer (Ti: 100 nm) isothermal (40 min at 600 o. C) XRD - Ti. Si & Ti. Si 2 52 crystal <100> evaporation bilayer (Ti: 100 nm) isothermal (30 min at 750 o. C) RBS/XRD - Ti. Si & Ti. Si 2 53 amorphous or <100> electron-gun deposition bilayer (Ti: 90 nm) isothermal (20 min at 450 o. C) Backscattering Spectroscopy - Ti. Si 54 crystal (111) electron-beam evaporation bilayer (Ti: 3 nm) isothermal (30 min at 600 o. C) TEM - Ti. Si & Ti. Si 2 55 crystal (100) conventional HV sputtering bilayer (Ti: 30 nm) isothermal (60 min at 650 o. C) RBS/TEM - Ti. Si 2 (C 49) 56 crystal <100> electron-gun evaporation bilayer (Ti: 140 nm) isothermal (120 min at 550 o. C) RBS/XRD/TEM - b. Ti. Si 57 amorphous electron-beam evaporation trilayer (Ti: 10~100 nm) isothermal (~300 s at 560 o. C) TEM yes SSA Ti 5 Si 3 45 amorphous or <100> sputter-deposition a-Si/Ti/Si trilayer (Ti: 23 nm) isothermal (60 min at 500 o. C) TEM/RBS yes SSA Ti. Si 2 (C 49) 58 crystal (100) sputter deposition bilayer (Ti: 25~35 nm) isothermal (30 min at 460 o. C) HRTEM/EDS yes SSA Ti. Si 2 (C 49) 59 crystal (111) UHV e-beam evaporation a-Si/Ti/Si trilayer (Ti: 30 nm) isothermal (30 min at 450 o. C) in-situ RHEED /HRTEM yes SSA poly Si rf sputtering bilayer (Ti: 55 nm) heating (10 o. C/m) to 510 o. C XTEM/STEM yes SSA Ti. Si 2 (C 49) 61 crystal (100) magnetron sputtering bilayer (Ti: 32, 51 nm) heating (15 o. C/min) to approx. 800 o. C IR-abs spect. XRD/resistivity yes SSA Ti. Si 2 (C 49) 62 crystal (100) magnetron sputtering bilayer (Ti: 32, 46 nm) heating (3, 20 o. C/s) to approx. 800 o. C in-situ XRD - Ti 5 Si 3/Ti 5 Si 4 63 c. Ti 2 5 Si 3 60 Byeong-Joo Lee www. postech. ac. kr/~calphad
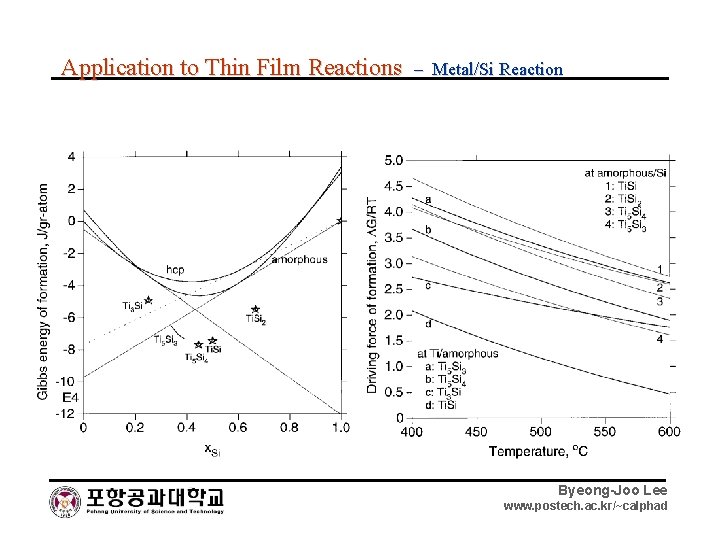
Application to Thin Film Reactions – Metal/Si Reaction Byeong-Joo Lee www. postech. ac. kr/~calphad
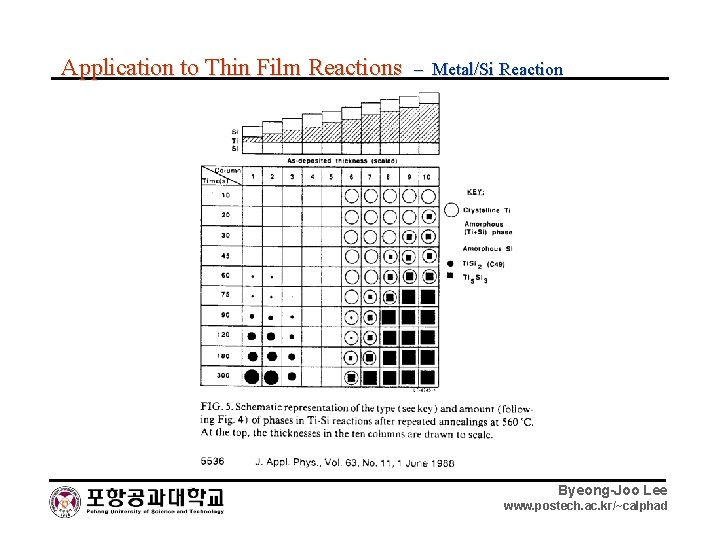
Application to Thin Film Reactions – Metal/Si Reaction Byeong-Joo Lee www. postech. ac. kr/~calphad
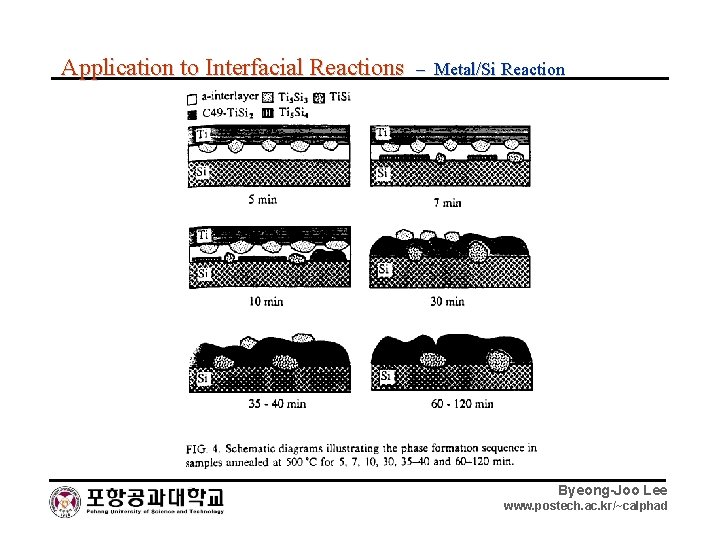
Application to Interfacial Reactions – Metal/Si Reaction Byeong-Joo Lee www. postech. ac. kr/~calphad

Thermodynamics Nano Materials Eunha Kim Inyoung Sa Byeong-Moon Lee and Byeong-Joo Lee Computational Materials Science & Engineering Lab. Pohang University of Science & Technology, Korea Byeong-Joo Lee www. postech. ac. kr/~calphad

Curvature Effect – Capillary Pressure System condition T = constant Vα = Vβ = V = constant @ equilibrium Byeong-Joo Lee www. postech. ac. kr/~calphad

Curvature Effect – Capillary Pressure Effect on Melting Point of Nano Particle Byeong-Joo Lee www. postech. ac. kr/~calphad
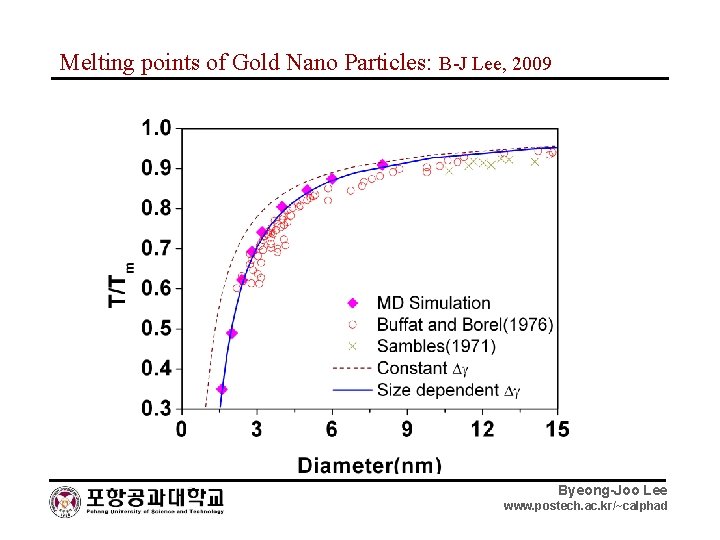
Melting points of Gold Nano Particles: B-J Lee, 2009 Byeong-Joo Lee www. postech. ac. kr/~calphad
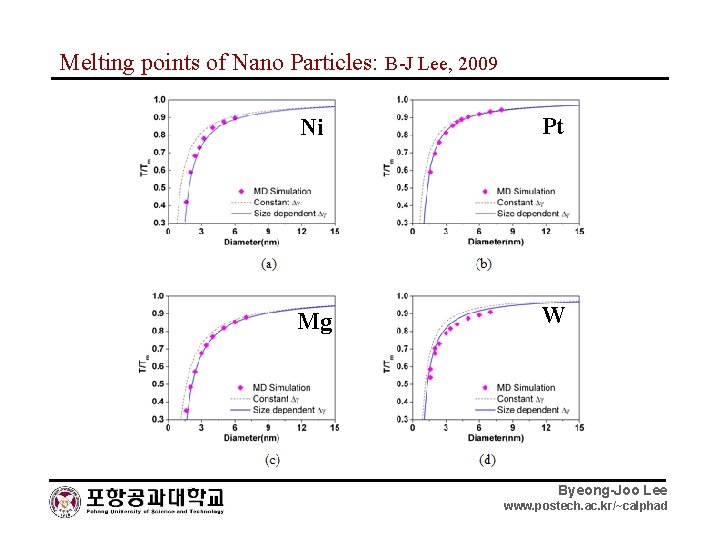
Melting points of Nano Particles: B-J Lee, 2009 Ni Pt Mg W Au Pt Byeong-Joo Lee www. postech. ac. kr/~calphad
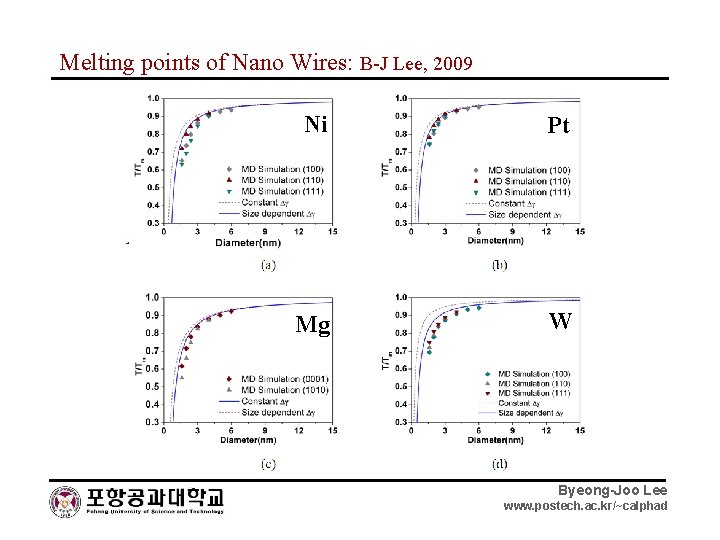
Melting points of Nano Wires: B-J Lee, 2009 Ni Pt Mg W Au Pt Byeong-Joo Lee www. postech. ac. kr/~calphad
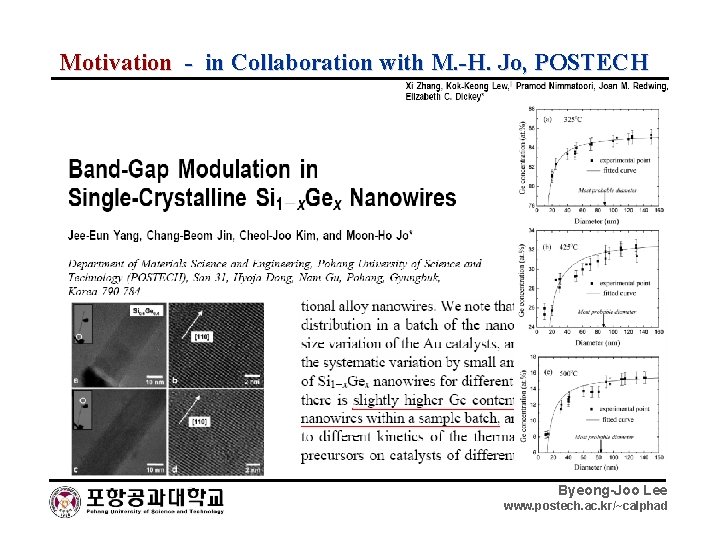
Motivation - in Collaboration with M. -H. Jo, POSTECH Byeong-Joo Lee www. postech. ac. kr/~calphad
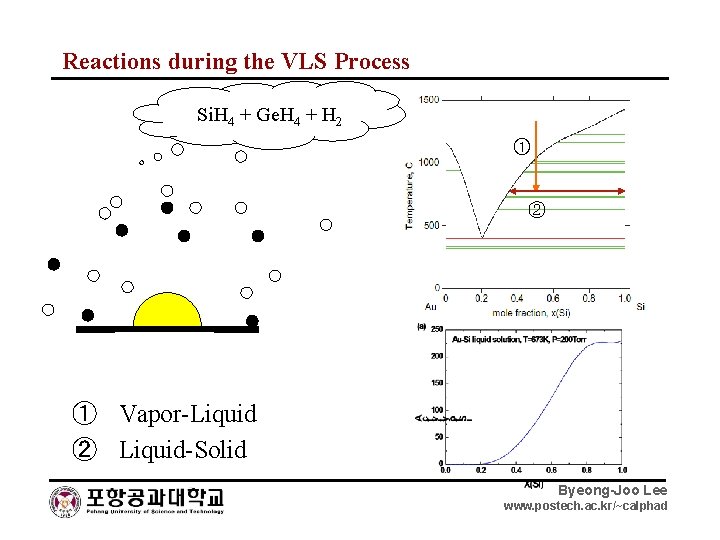
Reactions during the VLS Process Si. H 4 + Ge. H 4 + H 2 ① ② ① Vapor-Liquid ② Liquid-Solid Byeong-Joo Lee www. postech. ac. kr/~calphad
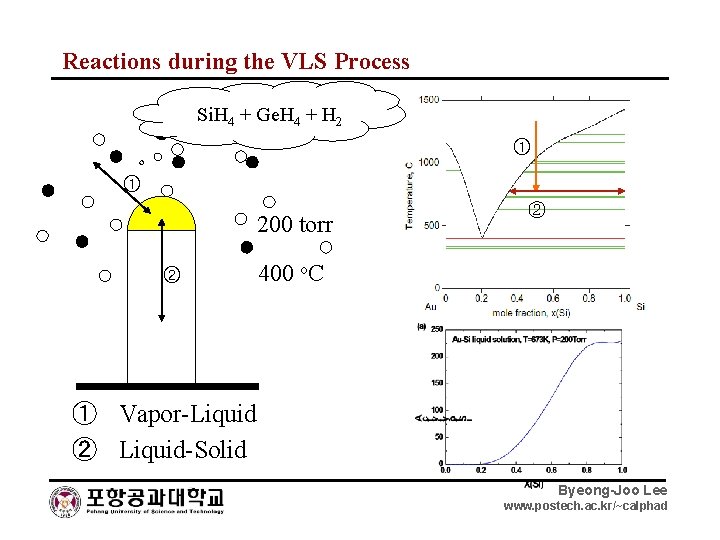
Reactions during the VLS Process Si. H 4 + Ge. H 4 + H 2 ① ① 200 torr ② ② 400 o. C ① Vapor-Liquid ② Liquid-Solid Byeong-Joo Lee www. postech. ac. kr/~calphad
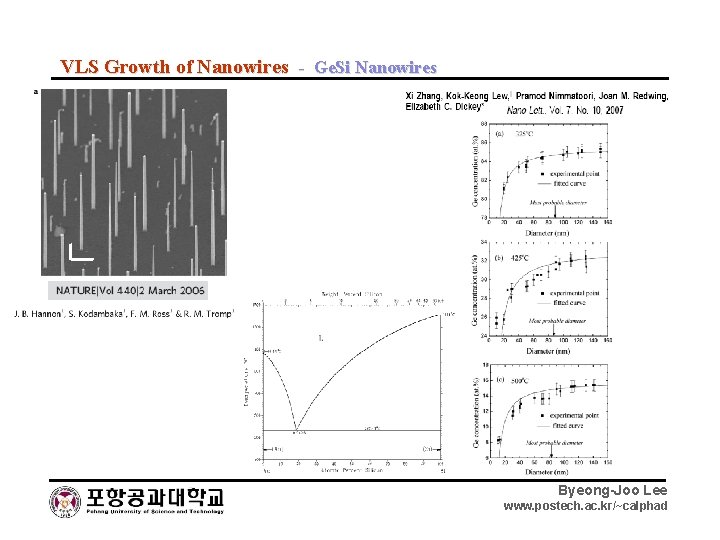
VLS Growth of Nanowires - Ge. Si Nanowires Byeong-Joo Lee www. postech. ac. kr/~calphad
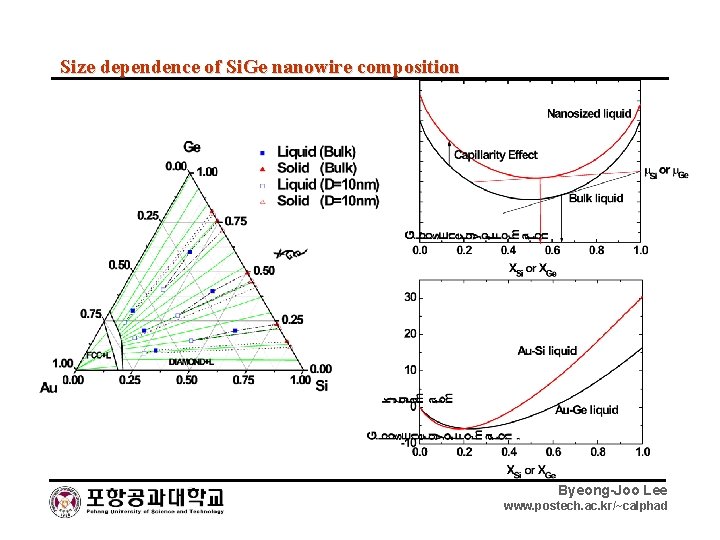
Size dependence of Si. Ge nanowire composition Byeong-Joo Lee www. postech. ac. kr/~calphad
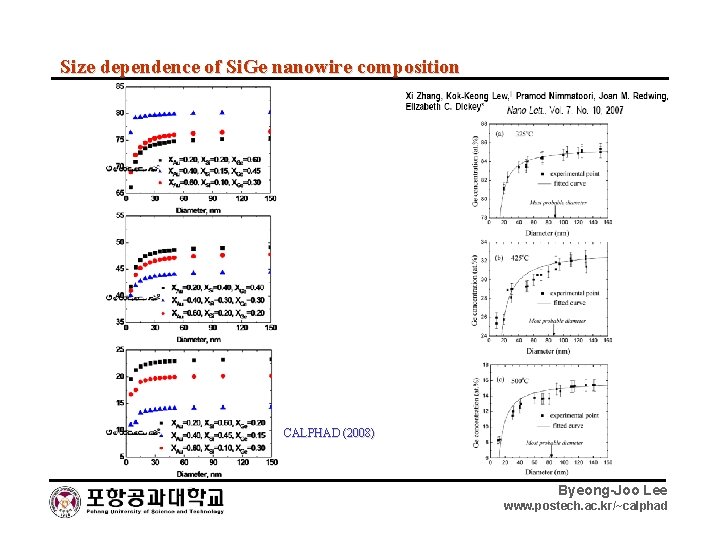
Size dependence of Si. Ge nanowire composition CALPHAD (2008) Byeong-Joo Lee www. postech. ac. kr/~calphad

Interfacial Phenomena – Segregation (Guttmann, Butler/Tanaka) Assume a one atomic layer surface phase and consider equilibrium between bulk and surface where ωi is the molar surface area Assume ωi = ωj = … = ω Byeong-Joo Lee www. postech. ac. kr/~calphad
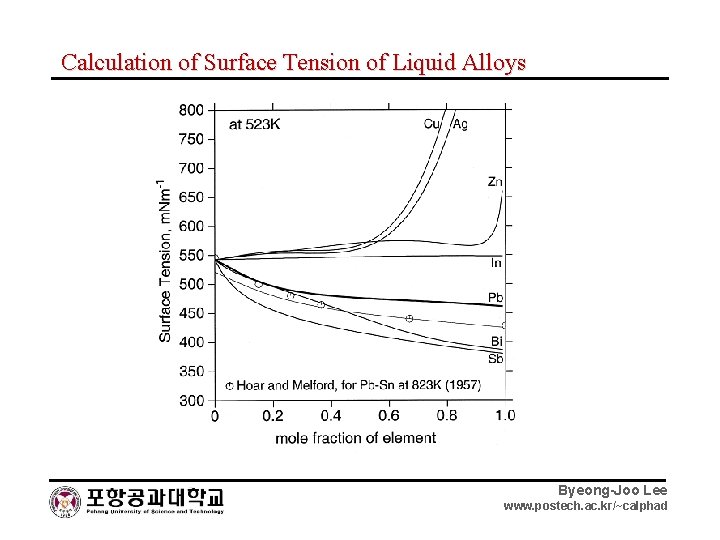
Calculation of Surface Tension of Liquid Alloys Byeong-Joo Lee www. postech. ac. kr/~calphad
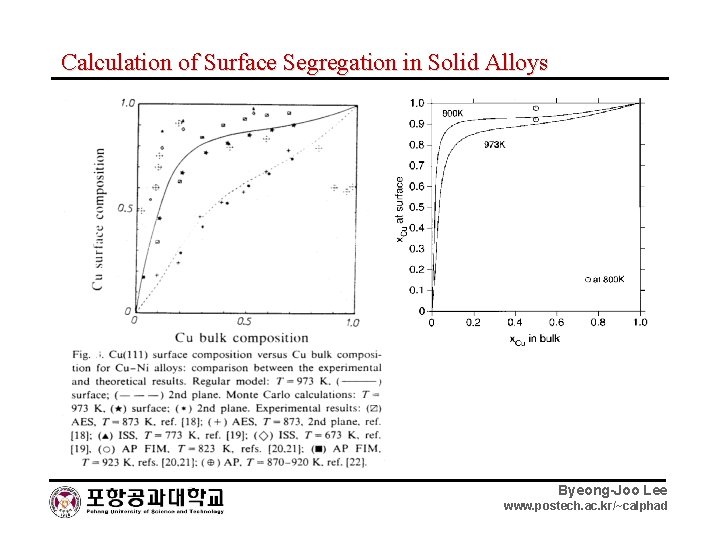
Calculation of Surface Segregation in Solid Alloys Byeong-Joo Lee www. postech. ac. kr/~calphad
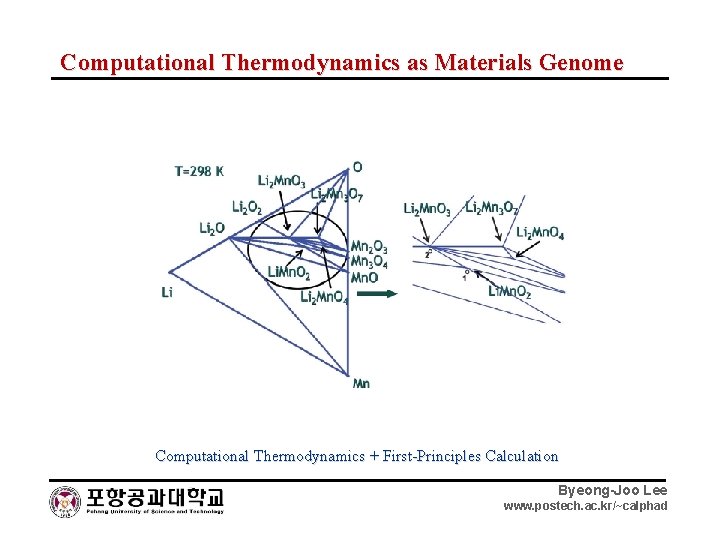
Computational Thermodynamics as Materials Genome Computational Thermodynamics + First-Principles Calculation Byeong-Joo Lee www. postech. ac. kr/~calphad
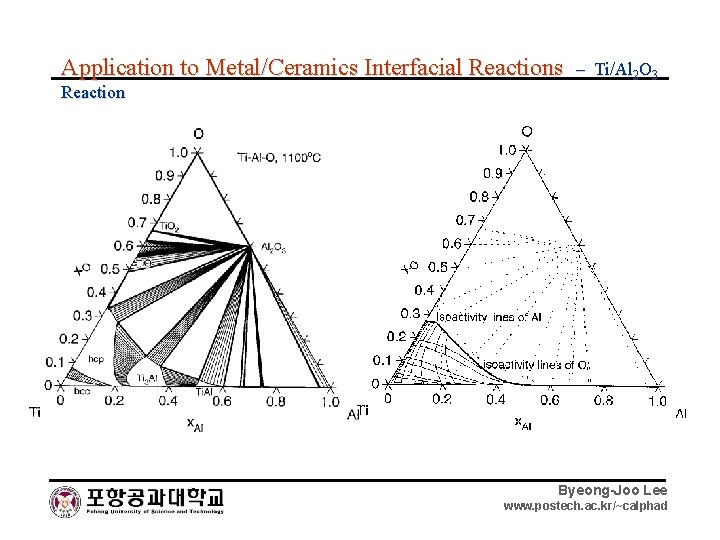
Application to Metal/Ceramics Interfacial Reactions – Ti/Al 2 O 3 Reaction Byeong-Joo Lee www. postech. ac. kr/~calphad

Application to Metal/Ceramics Interfacial Reactions – Ti/Al 2 O 3 Reaction Byeong-Joo Lee www. postech. ac. kr/~calphad
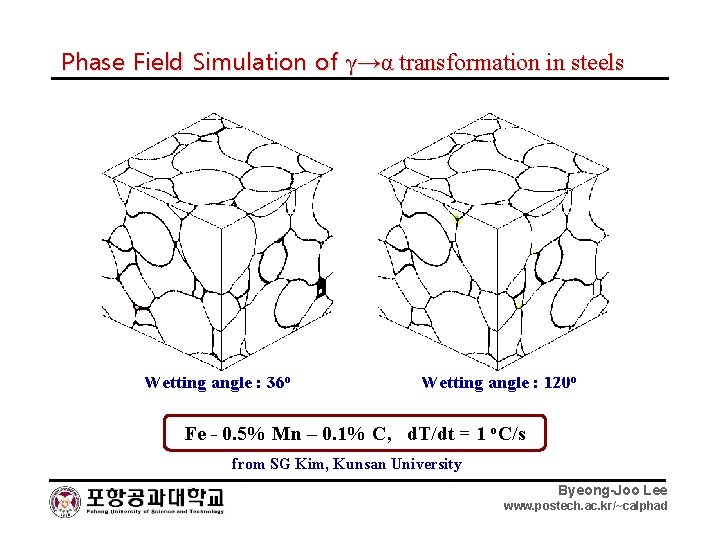
Phase Field Simulation of γ→α transformation in steels Wetting angle : 36 o Wetting angle : 120 o Fe - 0. 5% Mn – 0. 1% C, d. T/dt = 1 o. C/s from SG Kim, Kunsan University Byeong-Joo Lee www. postech. ac. kr/~calphad

Summary Computational Thermodynamics • • Calculation of Multi-component Phase Diagrams Interfacial Reactions – Metal/Liquid Solder, Metal/Ceramics Thin Films Reactions – Metal/Silicon Thermodynamics of Nano Materials – Capillarity Effect Byeong-Joo Lee www. postech. ac. kr/~calphad
- Slides: 49