Compressed Gases Compressed gas description v Definition of




















- Slides: 21

Compressed Gases

Compressed gas description v Definition of a compressed gas any gas, or mixture of gases, that is pressurized and contained in a cylinder or tank. 1 a

Compressed gas description v Examples of compressed gases • Acetylene • Anhydrous ammonia • Carbon dioxide • Helium v Types of containers in which compressed gas is stored 1 b

Cryogenic liquids v Definition of a cryogenic liquid matter that exists at minus 200 degrees Celsius v Examples of cryogenic liquids • Liquid nitrogen • Argon • Liquid oxygen 2 a

Handling and inspection procedures v Ensure gas containers are identified with a DOT or TC label v Cylinders must never be dropped, slid, or rolled 3 a

Handling and inspection procedures v Use carts or other material handling equipment to move cylinders v Regulators, pressure relief devices, valves, and hoses must be designed for the specific container 3 b

Handling and inspection procedures v Never interchange equipment between different types of gases v Pressure relief devices maintain pressure in cylinder at a desired level 3 c

Handling and inspection procedures v Inspect cylinders and devices, and remove defective equipment from service v Inspect threads on valves and regulators for damage, dirt, grease, and oil 3 d

Handling and inspection procedures v Threads on regulators and auxiliary equipment must match the container valve threads adapters are not allowed v Connections must not be tampered with, or forced together 3 e

Handling and inspection procedures v Valves should be opened slowly, with valve outlets pointed away from the user and other persons v Valves should be opened with the wrench supplied by the vendor 3 f

Handling and inspection procedures v Valves should not be hammered open or closed 3 g

Storage procedures v Containers upright must be stored v Storage areas must be wellventilated v Avoid storage temperatures above 125 degrees Fahrenheit 4 a

Storage procedures v Post the names of the gases in the storage area v Keep valve caps on containers when not connected to dispensing equipment 4 b

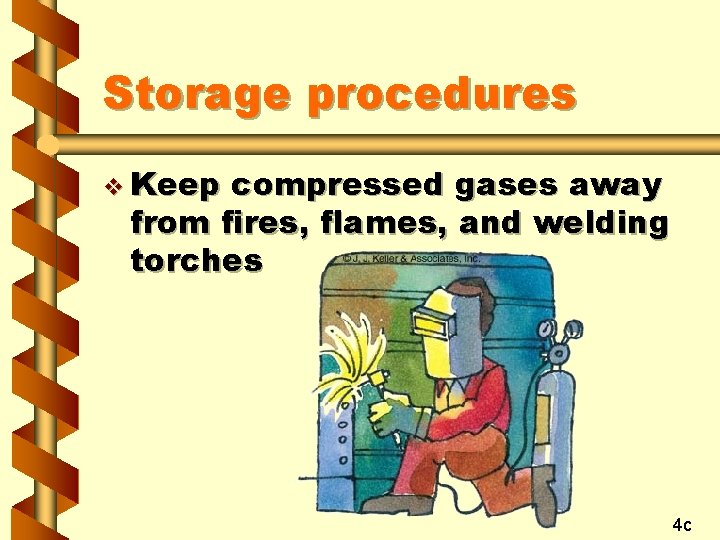
Storage procedures v Keep compressed gases away from fires, flames, and welding torches 4 c

Storage procedures v When containers are in storage, remove: • Regulators • Cylinder connections • Hoselines • Similar auxiliary devices 4 d

Storage procedures v Store oxygen away from flammable gas containers and combustibles • 20 -foot rule • Non-combustible 5 -foot high barrier 4 e

Storage procedures v Ensure that the storage area is: • properly labeled • secure • away from ignition sources • separate from oxidizing gases v Types of containers in which cryogenics are stored 4 f

Compressed gas hazards v Inert gases v Oxidizing gases v Fire hazards of flammable compressed gases 5 a

Compressed gas hazards v Health hazards of poison gases v Corrosive v Other gases hazards 5 b

Cryogenic liquid hazards v Extreme cold temperatures v Asphyxiation v Splashing transfer and boiling during 6 a

Personal protective equipment v Eye and face protection v Gloves v Aprons v Foot protection 7 a