Comprehensive Trainings to support the National Integrated TBHIV

Comprehensive Trainings to support the National Integrated TB/HIV Information System Implementation Facility Integrated TB/HIV Data Management Standard Operating Procedure

Background • ART M&E SOP created to support ART data reporting (2011) • Historically weak compliance with the ART M&E SOP • TB/HIV integration required revision of SOP • Revised SOP presented at 2017 TWG • Discussion on roles & responsibilities, NHC consultation • Provincial feedback incorporated, continual revision • To facilitate utilisation, SOP split into facility-level and above 2

Introducing… • Add pictures 2 1 20 3

Why is an updated SOP required? • New SOP incorporates TB/HIV integration maintenance • New SOP follows revised DHMIS reporting timelines • Previous SOP only included ART information

What are the key components of the SOP? • Guiding principles and key messaging • Guidance for facility level data management • Roles, responsibilities and accountability • Timelines for when to generate line lists and reports

Purpose • Provides guidance and direction on data management principles for managing TB/HIV programme – Standardises and builds efficiencies 6

Guiding Principles of the SOP • Enforces and supports accountability and quality • Guides TB/HIV data flow • Gold standard for TB/HIV data management • Directs facility-level data management activities – Sub(District) and above SOP to follow • Patient confidentiality is protected under applicable legislation and policies • National, provincial, sub(district), facility staff have an integral role in the institutionalisation of TB/HIV data management processes

Key Messages (1) • Replaces previous ART M&E SOP and TB M&E SOP • Needs to be disseminated, reviewed, and institutionalised by all staff who contribute to data management in the facility • Reports and line lists need to be routinely pulled according to timelines (weekly, monthly, and/or quarterly) – For time sensitive reports/line lists, the specific date of when to pull the reports/line lists is provided – For other reports/line lists, facility will need to choose (according to their operational schedule) when in the week/month to pull • Reports/line lists must be consistently pulled (i. e. each Monday, or each first week) 8

Key Messages (2) • Adhering to SOP will improve reporting and patient management • Aligns with Ideal Clinics prescript of one patient, one folder number • Supports TB/HIV integrated clinical management • Integrates TB/HIV data into single data flow process – Clinical recording, data capturing, reporting, and folder flow • Facilitates data use at facility and reporting of data from facility upward • Covers data management from public health facilities (including correctional centres) • All supporting information/documentation can be found on THIS portal: www. tbhivinfosys. org. za 9

Monitoring systems, tools, and supporting documents 1

Source documents Programme Component Source Documents for capturing into TB/HIV information system TB TB Case Identification TB Identification Register TB treatment and patient TB Treatment Record (blue folder) management Moving/transferring of TB patients Integrated TB/HIV Patient Summary HIV Laboratory tests conducted NHLS Laboratory Results HIV testing services HTS Register HIV/ART treatment and patient HIV/ART Patient and Visit Summary (Adult, management Paediatric) ART Adherence Club Adherence clubs registers/ART stationery Management Transferring of HIV/ART patients Integrated TB/HIV Patient Summary Laboratory tests conducted NHLS Laboratory Results
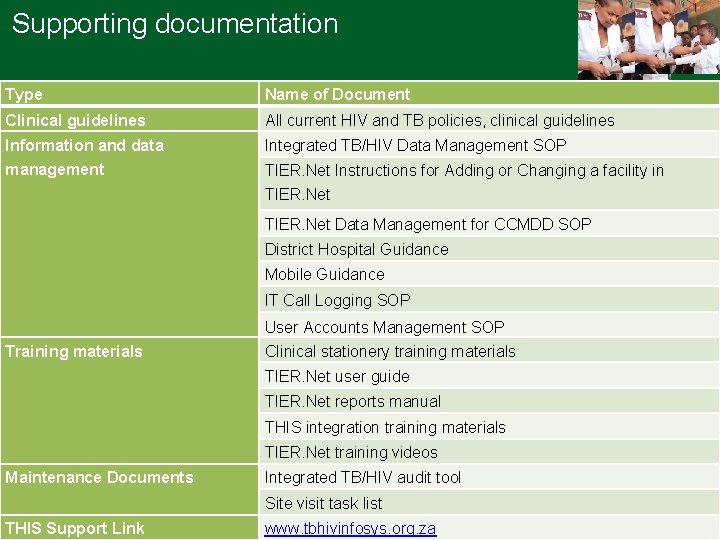
Supporting documentation Type Name of Document Clinical guidelines All current HIV and TB policies, clinical guidelines Information and data management Integrated TB/HIV Data Management SOP TIER. Net Instructions for Adding or Changing a facility in TIER. Net Data Management for CCMDD SOP District Hospital Guidance Mobile Guidance IT Call Logging SOP User Accounts Management SOP Training materials Clinical stationery training materials TIER. Net user guide TIER. Net reports manual THIS integration training materials TIER. Net training videos Maintenance Documents Integrated TB/HIV audit tool Site visit task list THIS Support Link www. tbhivinfosys. org. za

Timelines and data flow

TB Data Flow • Primary focus is on TIER. Net reporting • TB transition is not included in SOP, refer to THIS IG • Focus is on ensuring timeous and complete reporting for monthly and quarterly data • ETR. Net reporting hard stop March 2019 • TB ID register remains in use as it is source for presumptive TB data capture

HIV Data Flow • Primary focus is on TIER. Net reporting • Focus is on ensuring timeous and complete reporting for monthly and cohort data • TIER. Net HTS report can only be used if ALL HTS data captured • If community testing not captured per NDOH guidance, then add as aggregate to MDI form
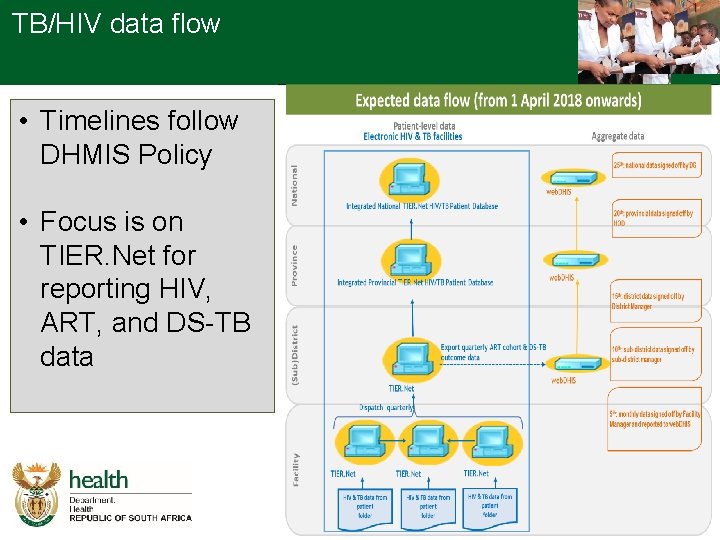
TB/HIV data flow • Timelines follow DHMIS Policy • Focus is on TIER. Net for reporting HIV, ART, and DS-TB data

Who is part of this Integrated TB/HIV Data Management SOP? • Facility SOP: – Admin Clerk (AC) – Facility and/or Operational Manager (FM/OM) – Facility/Health Information Officer (F/HIO) – Clinician – Club Manager – Mobile Clinic Nurse • Sub(district) and higher SOP: – Sub(district) managers (SDM/DM) • Information Management (IM) • HAST managers (HM) • DITs, TKIs, – Provincial managers (PM) • Information Management • PIT leads • HAST managers – National Managers 1
Who is an Admin Clerk (AC)?

Admin Clerk (AC) roles and responsibilities • ACs are responsible for administrative, reception and data management activities • Key activities include – Pulling, filing, archiving, ensuring file complete with stationery and overall folder hygiene – Capturing NHLS results that have been reviewed and signed-off by a clinician – Capturing into the monitoring system on a daily basis – Generating line lists and reports for routine data submission and reporting • Not responsible for ‘managing’ the broader THIS strategy

Facility/Health Information Officer (F/HIO) roles and responsibilities • Overseeing daily data management from all service points • Collating and verifying facility data • Providing reports to clinicians and FMs for review and discussion during weekly/monthly facility meetings • Submitting verified and approved data to sub(District)

Clinician roles and responsibilities • Recording of accurate and complete clinical visits into the standardised stationery • Participating in clinical and operational meetings • Demanding and reviewing data (line lists & reports) • Adhering to clinical governance prescripts • Sharing appropriate TB/HIV programmatic/clinical updates with administrative staff

Facility Manager (FM) roles and responsibilities • Responsible for ensuring the processes for all data produced at a health facility are completed • Overseeing staff responsible for those processes • Facilitating routine and consistent interrogation and utilisation of data available at the facility-level to improve patient management and outcomes • Providing overall management of – Data security – Audits – Stock control (related to data management)

Club Manager and Mobile Clinic Nurse Club Manager Mobile Clinic Nurse • Overseeing DM processes for Adherence Clubs • Overseeing DM processes for mobile clinics • Ensuring registers submitted to admin clerks for capturing into the monitoring system • Overseeing management of club data, including review, approval, and submission of club reports • Ensuring patient folders for MC patients are captured into monitoring system ART Adherence Guidelines • Liaising with ACs regarding filing of patient folders • Overseeing implementation of, and documentation of, recall processes for patients who have missed appointments Mobile Clinic Guidance

Sub(District), Provincial and National Integrated TB/HIV Data Management Standard Operating Procedure 2

Sub-District • Responsible for receiving, verifying, and approving facility data • Coordinating and driving data feedback loop with facilities • Reporting of facility data up along health system cascade • Supporting facilities within the sub-district with training, mentoring, and maintenance of THIS

District • Ensuring data for the districts (sub-districts, facilities) are received and interrogated according to timelines • Coordinating and driving data feedback loop with sub-district • Submitting verified and approved aggregated data to province • Ensure maintenance of THIS

Province • Overseeing THIS data management processes • Providing complete and accurate datasets to NDo. H • Coordinating and driving data feedback loop with districts • Ensuring that data resources are provided, data-related capacitation conducted/institutionalised, and adherence to NDOH data management guidance maintained • Overseeing governance of data management principles

National • Responsible for managing the National THIS strategy • Coordinating and driving feedback loop to provinces • Providing technical guidance and support to provincial teams • Custodians of National TB/HIV data set 2

District Support Partner (DSP) What is the role of a DSP What is NOT the DSP role • Supportive role • To ‘manage’ THIS strategy • Provide relevant and appropriate support as guided/requested by District(s) • Sole preoccupation cannot be data mining – i. e. staffing, equipment, fill gaps that DOH staff cannot reach Employees of district support partners require district manager approval to, in any way, hold, manipulate, or analyse patient-level data • Implement a non-NDOH endorsed agenda • Report data to funders that is not approved or in line with DHMIS

Summary of Facility Reports: Annexure B • User friendly chart/table summarising line lists and reports • Includes – Name of reports/line list – Date to generate – Description – Actions/interventions required and by whom • Broken down by timeframe – Daily, weekly, monthly, quarterly
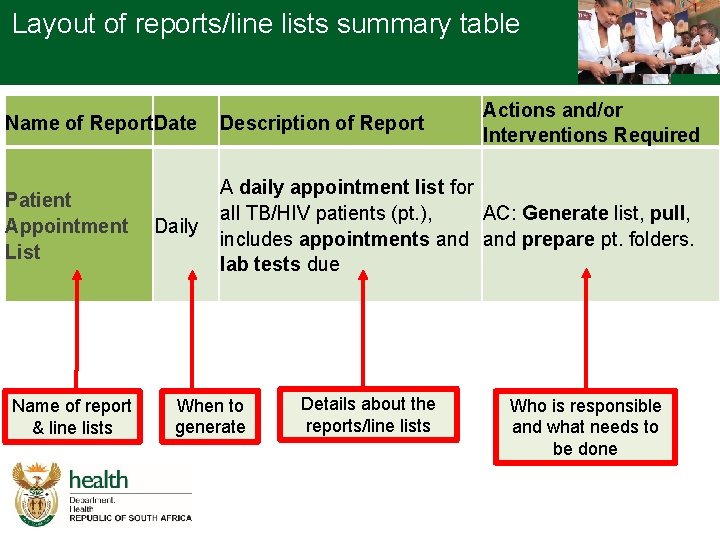
Layout of reports/line lists summary table Actions and/or Interventions Required Name of Report. Date Description of Report Patient Appointment List A daily appointment list for all TB/HIV patients (pt. ), AC: Generate list, pull, includes appointments and prepare pt. folders. lab tests due Name of report & line lists Daily When to generate Details about the reports/line lists 3 Who is responsible and what needs to be done
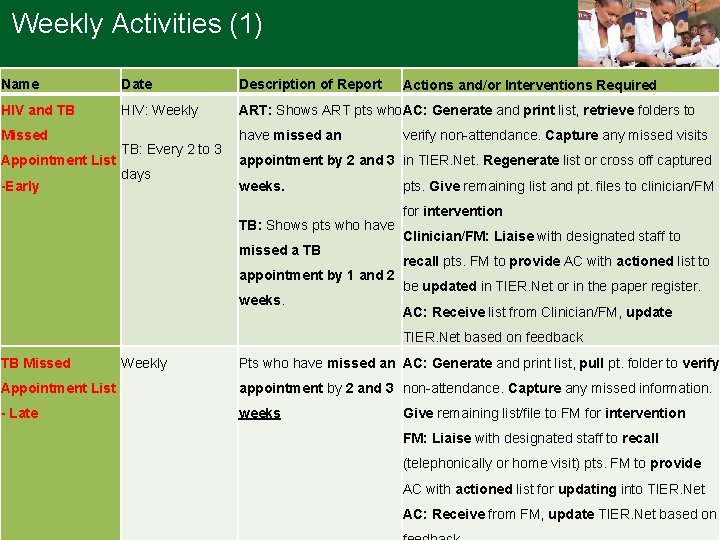
Weekly Activities (1) Name Date Description of Report HIV and TB HIV: Weekly ART: Shows ART pts who AC: Generate and print list, retrieve folders to Missed Appointment List -Early TB: Every 2 to 3 days have missed an Actions and/or Interventions Required verify non-attendance. Capture any missed visits appointment by 2 and 3 in TIER. Net. Regenerate list or cross off captured weeks. pts. Give remaining list and pt. files to clinician/FM TB: Shows pts who have missed a TB appointment by 1 and 2 weeks. for intervention Clinician/FM: Liaise with designated staff to recall pts. FM to provide AC with actioned list to be updated in TIER. Net or in the paper register. AC: Receive list from Clinician/FM, update TIER. Net based on feedback TB Missed Weekly Pts who have missed an AC: Generate and print list, pull pt. folder to verify Appointment List appointment by 2 and 3 non-attendance. Capture any missed information. - Late weeks Give remaining list/file to FM for intervention FM: Liaise with designated staff to recall (telephonically or home visit) pts. FM to provide AC with actioned list for updating into TIER. Net 3 AC: Receive from FM, update TIER. Net based on
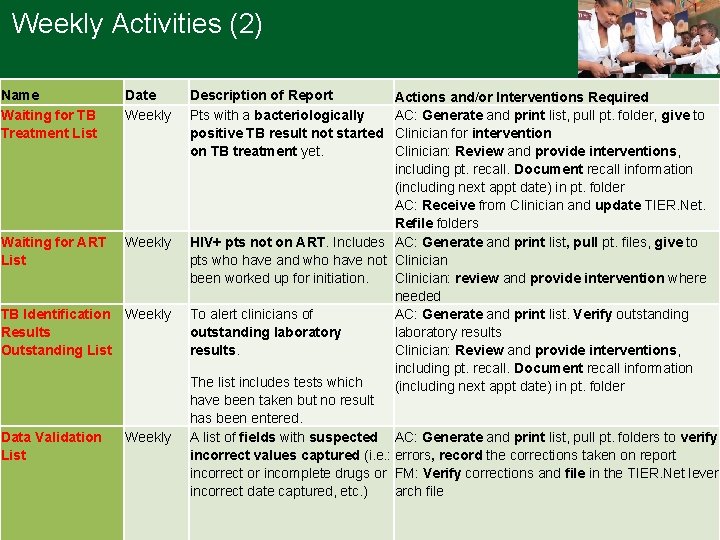
Weekly Activities (2) Name Waiting for TB Treatment List Date Weekly Waiting for ART List Weekly TB Identification Weekly Results Outstanding List Data Validation List Weekly Description of Report Pts with a bacteriologically positive TB result not started on TB treatment yet. Actions and/or Interventions Required AC: Generate and print list, pull pt. folder, give to Clinician for intervention Clinician: Review and provide interventions, including pt. recall. Document recall information (including next appt date) in pt. folder AC: Receive from Clinician and update TIER. Net. Refile folders HIV+ pts not on ART. Includes AC: Generate and print list, pull pt. files, give to pts who have and who have not Clinician been worked up for initiation. Clinician: review and provide intervention where needed To alert clinicians of AC: Generate and print list. Verify outstanding laboratory results. Clinician: Review and provide interventions, including pt. recall. Document recall information The list includes tests which (including next appt date) in pt. folder have been taken but no result has been entered. A list of fields with suspected AC: Generate and print list, pull pt. folders to verify incorrect values captured (i. e. : errors, record the corrections taken on report incorrect or incomplete drugs or FM: Verify corrections and file in the TIER. Net lever incorrect date captured, etc. ) arch file
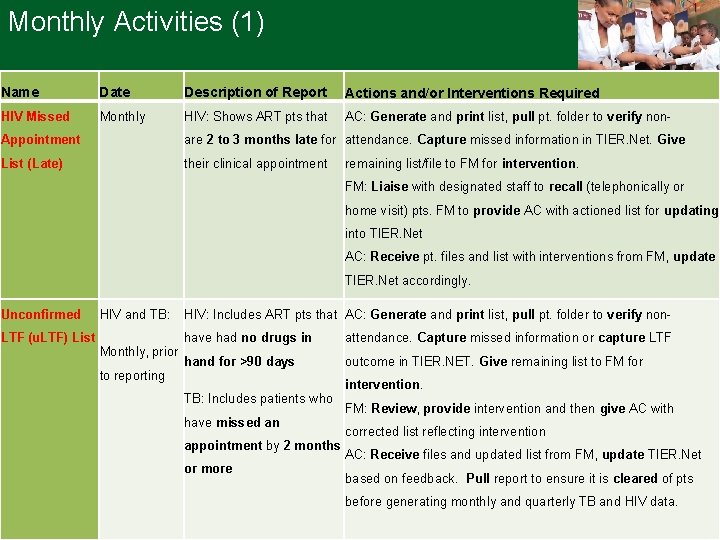
Monthly Activities (1) Name Date Description of Report Actions and/or Interventions Required HIV Missed Monthly HIV: Shows ART pts that AC: Generate and print list, pull pt. folder to verify non- Appointment are 2 to 3 months late for attendance. Capture missed information in TIER. Net. Give List (Late) their clinical appointment remaining list/file to FM for intervention. FM: Liaise with designated staff to recall (telephonically or home visit) pts. FM to provide AC with actioned list for updating into TIER. Net AC: Receive pt. files and list with interventions from FM, update TIER. Net accordingly. Unconfirmed LTF (u. LTF) List HIV and TB: Monthly, prior to reporting HIV: Includes ART pts that AC: Generate and print list, pull pt. folder to verify nonhave had no drugs in attendance. Capture missed information or capture LTF hand for >90 days outcome in TIER. NET. Give remaining list to FM for TB: Includes patients who have missed an appointment by 2 months or more intervention. FM: Review, provide intervention and then give AC with corrected list reflecting intervention AC: Receive files and updated list from FM, update TIER. Net based on feedback. Pull report to ensure it is cleared of pts before generating monthly and quarterly TB and HIV data. 3

Monthly Activities (2) - TB Name Date DS-TB Monthly Conversion Sputa Required List Description of Report A list of bacteriologically positive pts on regimens 1 or 3 with no conversion sputum result between 42 to 70 days after treatment start. Actions and/or Interventions Required AC: Generate and print. Give to clinician and FM. Clinician/FM: Review report and determine reason for there being no conversion result. Clinician to take and document intervention. TB Monthly Outstanding Outcomes List of pts who have been on TB treatment for longer than the expected duration & do not have an outcome. AC: Generate and print list, and pull pt. folders to verify missing outcomes. If outcomes missing, give list and folders to Clinician: Review and provide intervention. Once updated return list and folder to AC. AC: Update missing outcomes on TIER. Net, refile folders and file report in TIER. Net lever arch file. DS-TB Non- Monthly A list of DS-TB pts who were Conversion initiated onto TB treatment, List with a bacteriologically positive smear or culture result at 2 or 3 months after starting TB treatment AC: Generate and print. Give to clinician and FM for intervention. Clinician/FM: Review and verify report, provide intervention.

Monthly Activities (3) - TB Name Date DS-TB Monthly Discharge Sputa Required List Description of Report Shows all DS-TB pts requiring discharge sputa results, irrespective if diagnosis & conversion sputa. Actions and/or Interventions Required AC: Generate and print. Ensure all lab results are captured and filed in pt. blue cards. Give list to clinician and FM Clinician/FM: Review report and provide interventions. DS-TB Cases Monthly by HIV & ART status Lists TB registrations by HIV and ART status to support clinical management, as per TB/HIV guidelines Conversion report for all smear positive TB cases and includes outcomes prior to sputum conversion up to 2 and 3 months AC: Generate and print. Give to clinician and FM. Clinician/FM: Review report and provide interventions. DS-TB Smear Monthly Conversion Report AC: Generate and print report, give to Clinician: Provide clinical guidance regarding documentation at next clinical management meeting FM: Review, verify, and validate report; approve/sign-off report and file; discuss report with staff during review meetings.
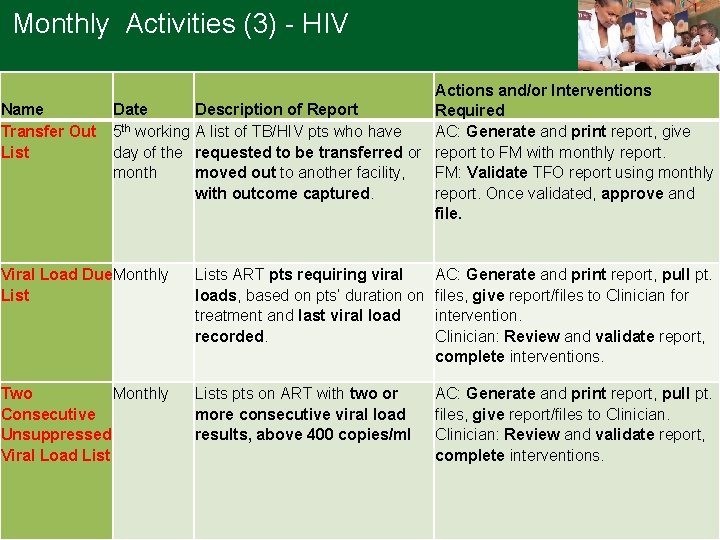
Monthly Activities (3) - HIV Name Transfer Out List Actions and/or Interventions Date Description of Report Required 5 th working A list of TB/HIV pts who have AC: Generate and print report, give day of the requested to be transferred or report to FM with monthly report. month moved out to another facility, FM: Validate TFO report using monthly with outcome captured. report. Once validated, approve and file. Viral Load Due Monthly Lists ART pts requiring viral loads, based on pts’ duration on treatment and last viral load recorded. AC: Generate and print report, pull pt. files, give report/files to Clinician for intervention. Clinician: Review and validate report, complete interventions. Two Monthly Consecutive Unsuppressed Viral Load Lists pts on ART with two or more consecutive viral load results, above 400 copies/ml AC: Generate and print report, pull pt. files, give report/files to Clinician: Review and validate report, complete interventions.
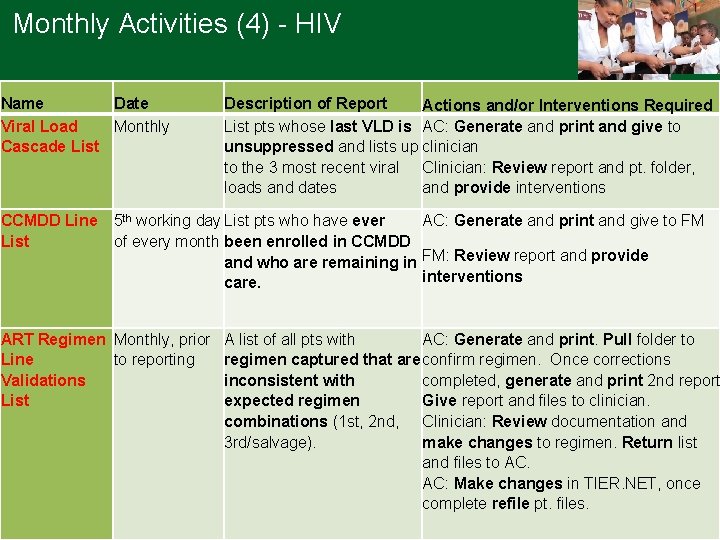
Monthly Activities (4) - HIV Name Date Viral Load Monthly Cascade List CCMDD Line List Description of Report Actions and/or Interventions Required List pts whose last VLD is AC: Generate and print and give to unsuppressed and lists up clinician to the 3 most recent viral Clinician: Review report and pt. folder, loads and dates and provide interventions 5 th working day List pts who have ever AC: Generate and print and give to FM of every month been enrolled in CCMDD and who are remaining in FM: Review report and provide interventions care. ART Regimen Monthly, prior Line to reporting Validations List A list of all pts with AC: Generate and print. Pull folder to regimen captured that are confirm regimen. Once corrections inconsistent with completed, generate and print 2 nd report expected regimen Give report and files to clinician. combinations (1 st, 2 nd, Clinician: Review documentation and 3 rd/salvage). make changes to regimen. Return list and files to AC. AC: Make changes in TIER. NET, once complete refile pt. files.

Monthly – Facility and system reports Name Facility Management Report Date Monthly, prior to reporting Description of Report Summary report listing critical information from each report in the Clinical Governance and Pt and Facility Management Reports Actions and/or Interventions Required AC: Generate and print report. If the indicator has a RED X, generate corresponding report relating to the potential issues. Give report to FM for review and intervention FM: Review report and provide interventions as required Workload Report Monthly Shows all visits captured, modified per user FIO: Generate and print report, give to FM. FM: Review report and provide interventions where needed. User Access Monthly Report Produces a list of user accounts FIO: Generate and print report, give to FM. activities, including the last log in FM: Review report and provide interventions date, activation status, and where needed. time/date for each log in/off. Implementer/ Monthly Administrator Log Report Logged activities by implementer/administrator FIO: Generate and print report, review with FM. FM: Review report and identify any implementers or administrators who have captured data. Engage with I/A to address identified issues where needed.
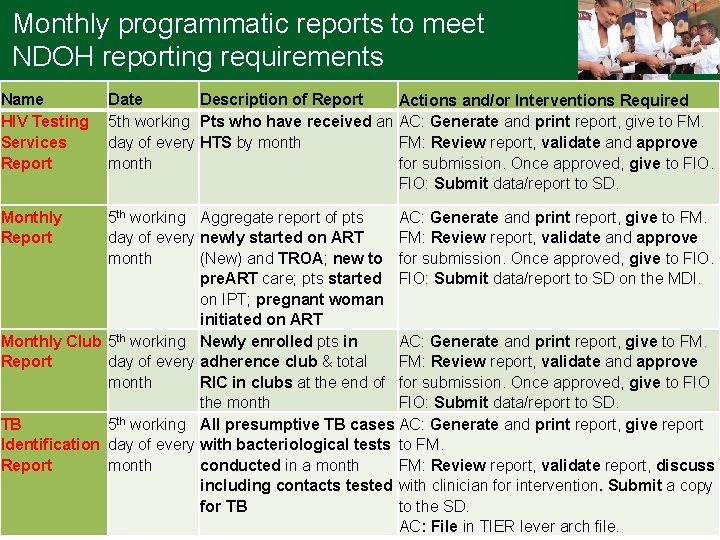
Monthly programmatic reports to meet NDOH reporting requirements Name HIV Testing Services Report Monthly Report Date Description of Report Actions and/or Interventions Required 5 th working Pts who have received an AC: Generate and print report, give to FM. day of every HTS by month FM: Review report, validate and approve month for submission. Once approved, give to FIO: Submit data/report to SD. 5 th working Aggregate report of pts AC: Generate and print report, give to FM. day of every newly started on ART FM: Review report, validate and approve month (New) and TROA; new to for submission. Once approved, give to FIO. pre. ART care; pts started FIO: Submit data/report to SD on the MDI. on IPT; pregnant woman initiated on ART Monthly Club 5 th working Newly enrolled pts in AC: Generate and print report, give to FM. Report day of every adherence club & total FM: Review report, validate and approve month RIC in clubs at the end of for submission. Once approved, give to FIO the month FIO: Submit data/report to SD. TB 5 th working All presumptive TB cases AC: Generate and print report, give report Identification day of every with bacteriological tests to FM. Report month conducted in a month FM: Review report, validate report, discuss including contacts tested with clinician for intervention. Submit a copy for TB to the SD. AC: File in TIER lever arch file.
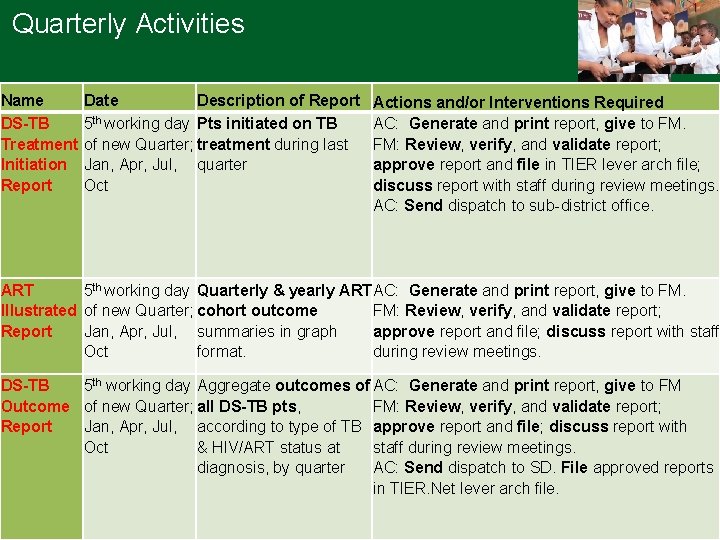
Quarterly Activities Name DS-TB Treatment Initiation Report Date Description of Report 5 th working day Pts initiated on TB of new Quarter; treatment during last Jan, Apr, Jul, quarter Oct Actions and/or Interventions Required AC: Generate and print report, give to FM. FM: Review, verify, and validate report; approve report and file in TIER lever arch file; discuss report with staff during review meetings. AC: Send dispatch to sub-district office. ART 5 th working day Quarterly & yearly ART AC: Generate and print report, give to FM. Illustrated of new Quarter; cohort outcome FM: Review, verify, and validate report; Report Jan, Apr, Jul, summaries in graph approve report and file; discuss report with staff Oct format. during review meetings. DS-TB 5 th working day Aggregate outcomes of AC: Generate and print report, give to FM Outcome of new Quarter; all DS-TB pts, FM: Review, verify, and validate report; Report Jan, Apr, Jul, according to type of TB approve report and file; discuss report with Oct & HIV/ART status at staff during review meetings. diagnosis, by quarter AC: Send dispatch to SD. File approved reports in TIER. Net lever arch file.

Annexure C: User Account Profiles • Provides summary of user account profiles available in TIER. Net. – Implementer, Administrator and User • Details available in ‘TIER. Net User Account Management: Guidelines for the creating, managing, and terminating user accounts in TIER. Net Guide’, – available on www. tbhivinfosys. org. za. 4

Thank you

THIS Maintenance Tools 44

THIS maintenance tools (1) • There are two standard tools that are routinely used when assessing TIER. Net management status at facility-level, namely - Integrated Audit Tool (IAT) - Site Visit Task List (SVTL) • The tools must be used by any relevant individuals (programme managers, supervisors, DSPs etc. ) during facility visits

THIS maintenance tools (2) • Changes to the tools are significant • Dissemination must be well thought-out • Accompanied with bespoke support to those who are meant to use the tools • Previous version focused more on technical and staffing • Focuses on TB/HIV integration – all modules • Supports discussion around transition from implementation to maintenance

THIS maintenance tools (3) • Supports the implementation of data use • Rigorous and lengthy engagement with tools to test veracity • Provincial counterparts were asked for feedback • Final tool incorporated feedback from various provinces

THIS maintenance tools (4) • The tools assess 1. Data Quality a) Clinical record keeping b) Data capturing c) Reporting 2. Facility adherence to SOPs and policies 3. Status of TIER. Net usage, in its totality, at facility level 4. Status of facility resources for TIER maintenance

Introduction – Integrated audit tool • Previous audit tool focused on completeness and accuracy of ART clinical stationery – aspects that continue to be critical to audit • With TB/HIV integration, need arose to expand audit tool alongside TIER. Net functionality expansion • Same principles apply – tool follows clinical stationery and TIER. Net patient record flow/structure • Robust testing of tool across country, continued revision • Sought feedback from provincial counterparts via the TWG • Incorporated provincial feedback and learnings from testing

Integrated audit tool (1) • Integrated audit tool has separate tabs that focuses on the following areas: - Clinical recording (ART & TB stationery) - Data capturing (ART & TB stationery) - HTS capturing - Policy & data quality o Availability of SOP o Policy adherence o Data quality (registers, input forms, web. DHIS) – takes a cue from RIPDA - Results appraisal and remedial actions

Integrated audit tool (3) • Findings from audit must be discussed with the FM in detail • Focus on the discussions that the tool prompts, and less on the specific • Tool should be standardised across facilities using TIER. Net • FOLLOWS THE LOGIC AND FLOW OF THE ART CLINICAL STATIONERY

Integrated audit tool – what's needed? • For auditing of ART and TB clinical record keeping – Total of 9 files should be drawn – 3 x ART, 3 x TB and 3 x TB/HIV co-infected – Therefore, 6 x ART and 6 x TB files for auditing • Same folders that are used to audit clinical record keeping should the used to audit the data capturing • HTS capturing should be done directly from the HTS register – • Auditing capturing of the HTS module against the HTS register Latest submitted monthly data (monthly input form and exact TIER. Net “Monthly report” that was used as the source document)
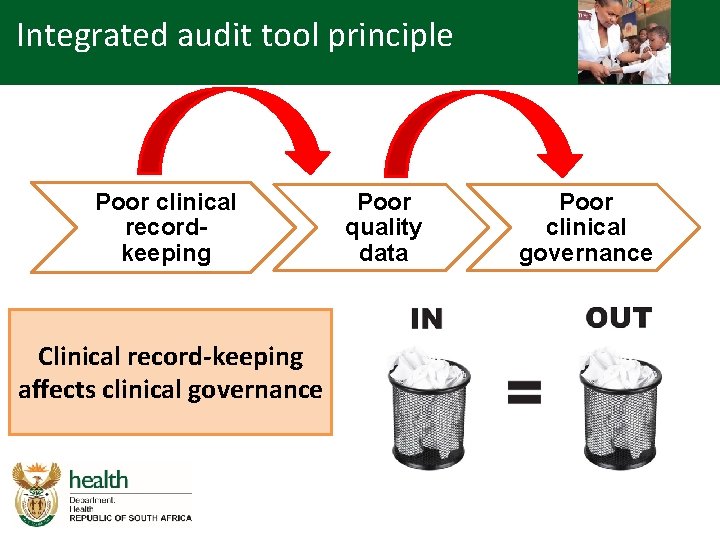
Integrated audit tool principle Poor clinical recordkeeping Clinical record-keeping affects clinical governance Poor quality data Poor clinical governance

Integrated Audit Tool Intro Page

Enter facility details here ///// Select which modules you are auditing here Indicate when the last back-up was conducted

Integrated Audit Tool HTS Capture
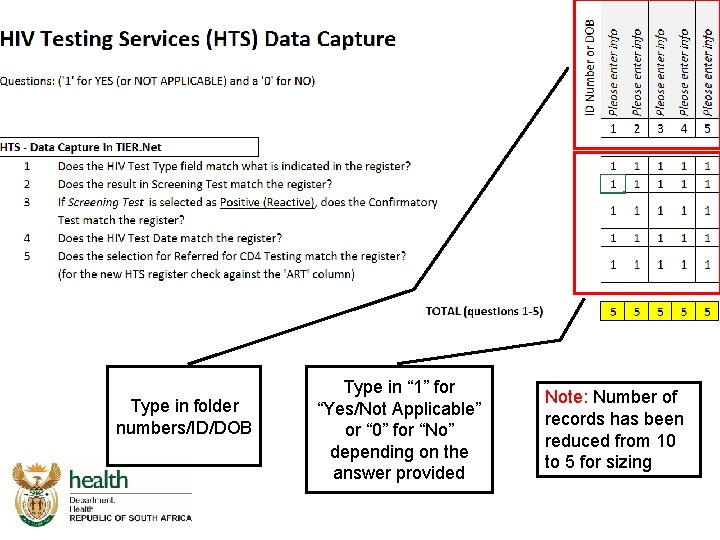
Type in folder numbers/ID/DOB Type in “ 1” for “Yes/Not Applicable” or “ 0” for “No” depending on the answer provided Note: Number of records has been reduced from 10 to 5 for sizing

Integrated Audit Tool ART Clinical Record Keeping

Integrated audit tool – ART clinical record keeping

This only accounts for the standardised ART clinical stationery Refer to “Long Term Record” on front page of clinical stationery Refer to “Patient details” section on front page of clinical stationery Type in folder number Note: Number of records has been reduced from 6 to 3 for sizing

Refer to “Long Term Record” on front page of clinical stationery Note: Number of records has been reduced from 6 to 3 for sizing

Integrated Audit Tool ART Data Capture

Integrated audit tool – ART data capture

Note: Number of records has been reduced from 6 to 3 for sizing Compare against front page of clinical stationery Folder number autopopulated from “clinical recordkeeping” section
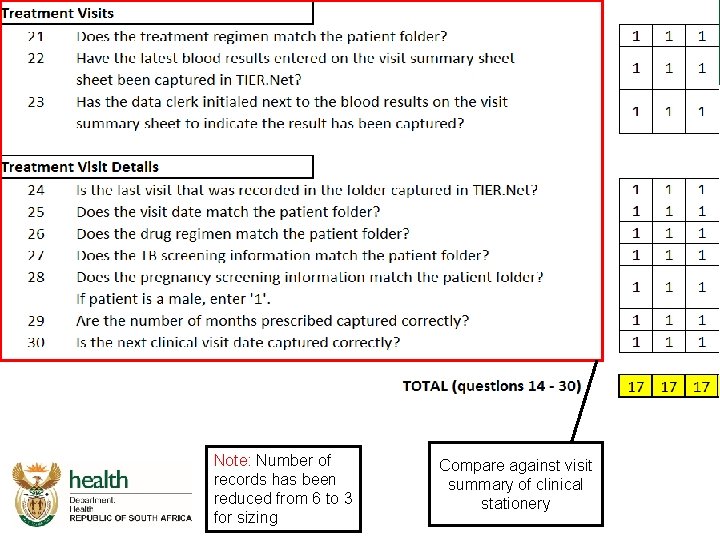
Note: Number of records has been reduced from 6 to 3 for sizing Compare against visit summary of clinical stationery

Integrated Audit Tool DS-TB Clinical Record Keeping

Integrated audit tool - TB record keeping
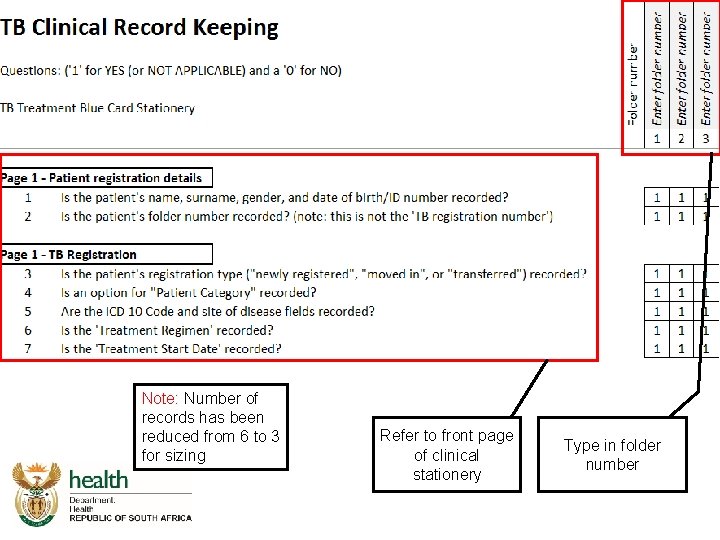
Note: Number of records has been reduced from 6 to 3 for sizing Refer to front page of clinical stationery Type in folder number

Note: Number of records has been reduced from 6 to 3 for sizing Refer to “Progress notes” section of clinical stationery Refer to page 2 of clinical stationery

Refer to “Progress notes” section of clinical stationery Note: Number of records has been reduced from 6 to 3 for sizing

Integrated Audit Tool DS-TB Data Capture

Integrated audit tool - TB data capture
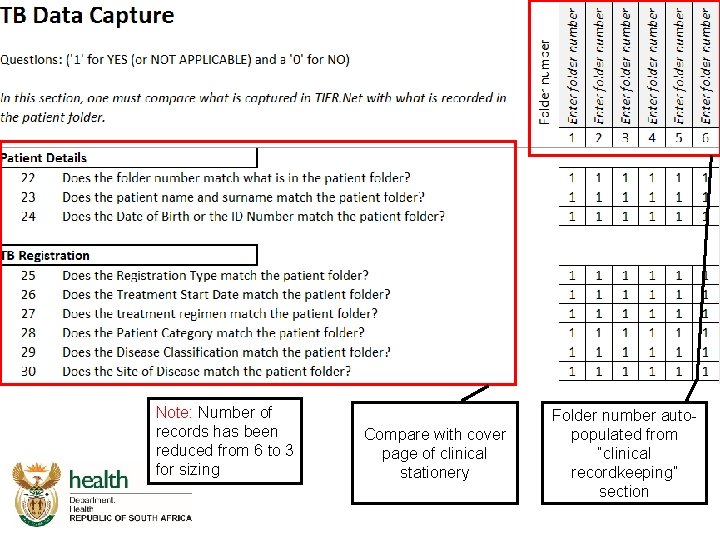
Note: Number of records has been reduced from 6 to 3 for sizing Compare with cover page of clinical stationery Folder number autopopulated from “clinical recordkeeping” section
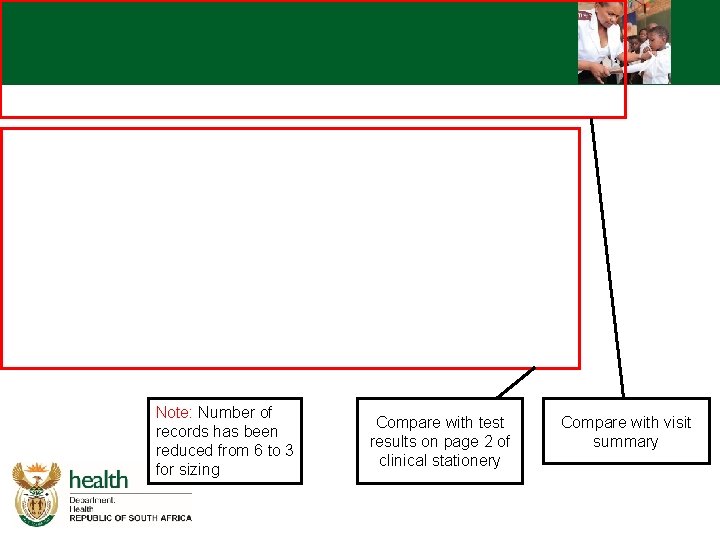
Note: Number of records has been reduced from 6 to 3 for sizing Compare with test results on page 2 of clinical stationery Compare with visit summary

Compare with “Treatment outcome” section Note: Number of records has been reduced from 6 to 3 for sizing

Integrated Audit Tool SOP/Policy Adherence Data Quality
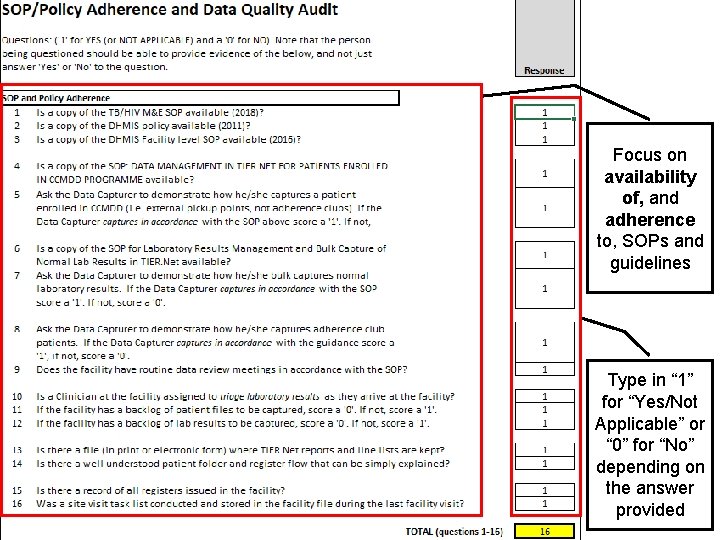
Integrated audit tool – SOP/Policy adherence Focus on availability of, and adherence to, SOPs and guidelines Type in “ 1” for “Yes/Not Applicable” or “ 0” for “No” depending on the answer provided

Integrated audit tool – Data quality

Integrated audit tool – Data quality 9 data elements to be assessed as part of the data quality audit
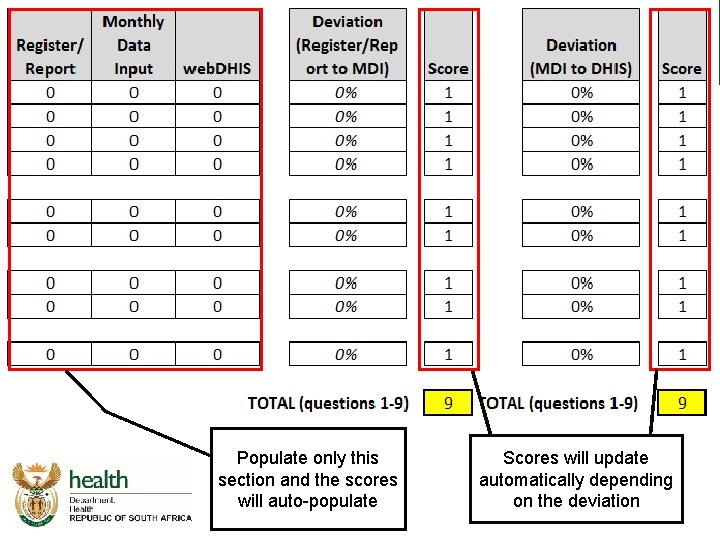
Populate only this section and the scores will auto-populate Scores will update automatically depending on the deviation

Integrated Audit Tool Scoring Summary Remedial Actions
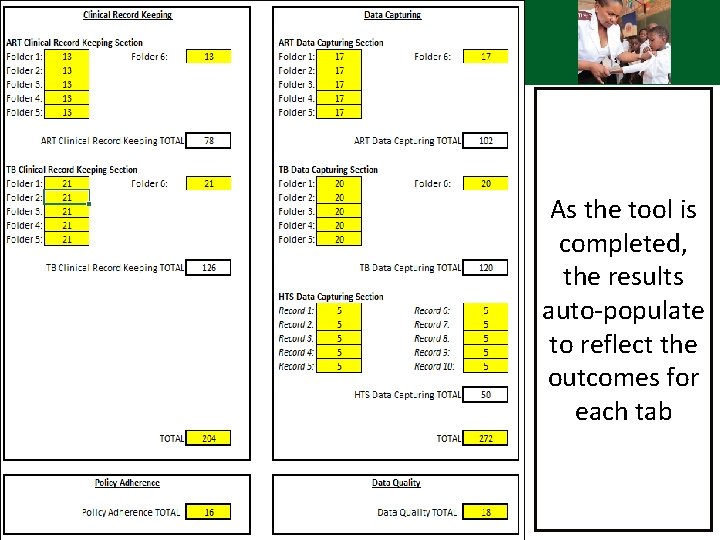
Integrated audit tool – Results As the tool is completed, the results auto-populate to reflect the outcomes for each tab

Integrated audit tool – Results The overall score will be reflected here Participants of the audit, date of audit and district representative Remedial actions based on what the findings of the audit were

Thank you

THIS Portal 85

THIS Portal • The portal will be a routine resource for those who regularly manage, utilise, and interact with the TB/HIV information system • THIS portal will replace VULA and all updates with regards to TIER and policies will be uploaded • All participants to be assigned usernames and passwords will be assigned after the trainings • Go onto the portal by following the link - http: //tbhivinfosys. org. za/ • Logins will be disseminated after the trainings

THIS Portal • Currently, portal has three primary functions: – FAQ • This section includes answers to frequently asked questions – Resources and tools • This section is where critical files, including guidance materials, training slide decks, TIER. Net installers, and other resources, can be found • Some files are available openly, and some are available only to users with login details – Contact us • This is where users can log/submit queries to the National Implementation Team (NIT)
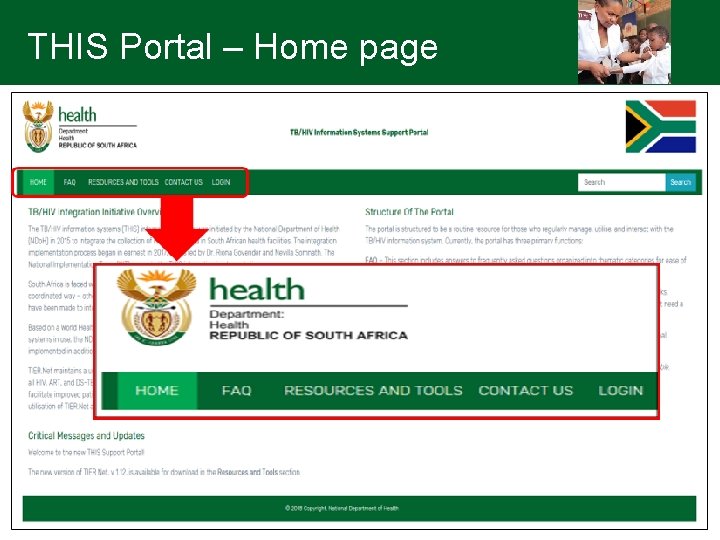
THIS Portal – Home page

Thank you

Site Visit Task List 90

Overview - SVTL (1) • Site visit task list covers the following areas: – Facility TIER. Net implementation status • Modules on TIER. Net • Technical (version, anti-virus updates etc. ) • Backlog – Previous audit – Usage of patient management lists • Establish the use of patient management lists and reports

Overview - SVTL (2) – Status of data capturing and patient flow • Establish the patient folder flow • Lab results management – Availability of resources (staffing, equipment) – Action Plan

Overview - SVTL (3) • Participants include different categories of staff – Facility personnel (FM, AC, etc. ) – (Sub)district managers • Items that require attention of the facility needs to be highlighted • Findings from site visit task list must be discussed with the FM in detail • Follow-up engagements need to be scheduled to ensure action items have been implemented
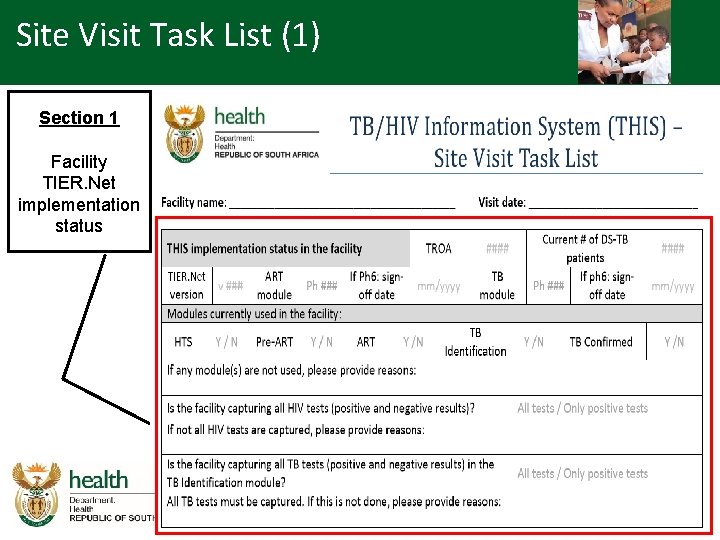
Site Visit Task List (1) Section 1 Facility TIER. Net implementation status
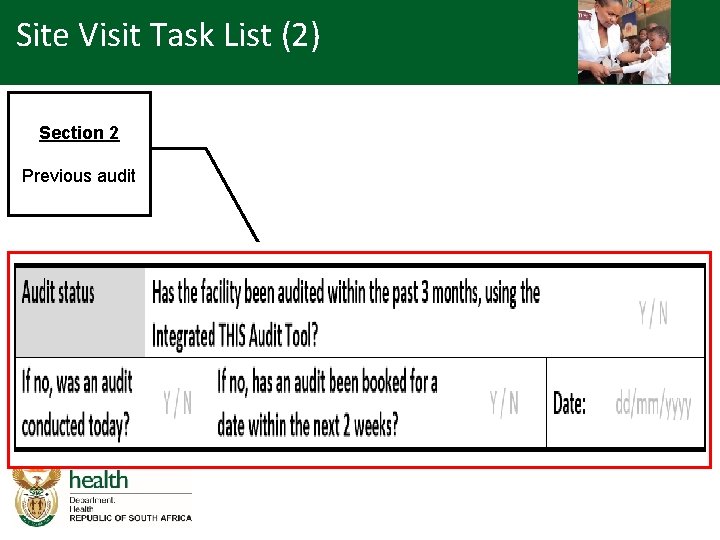
Site Visit Task List (2) Section 2 Previous audit
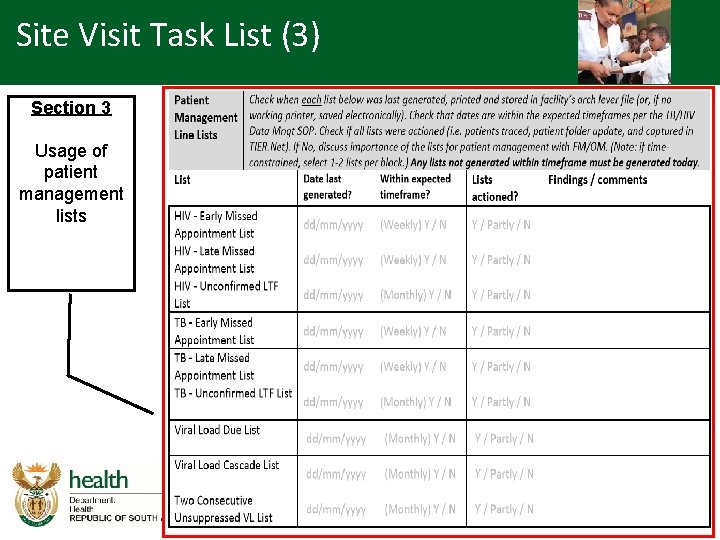
Site Visit Task List (3) Section 3 Usage of patient management lists
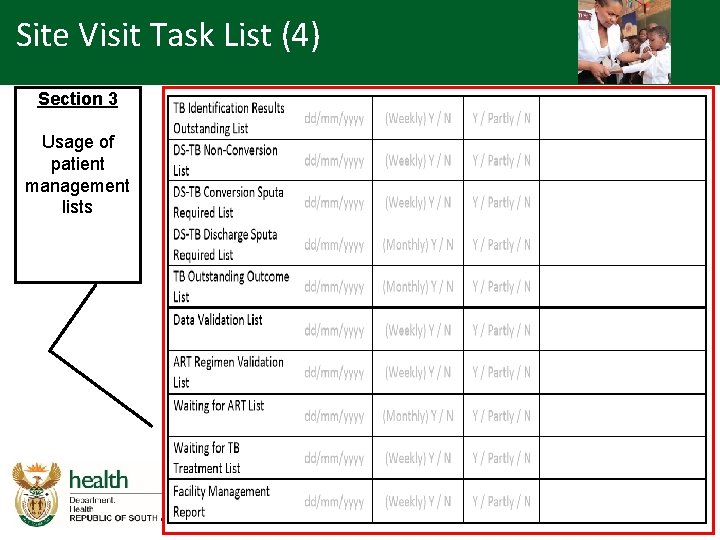
Site Visit Task List (4) Section 3 Usage of patient management lists
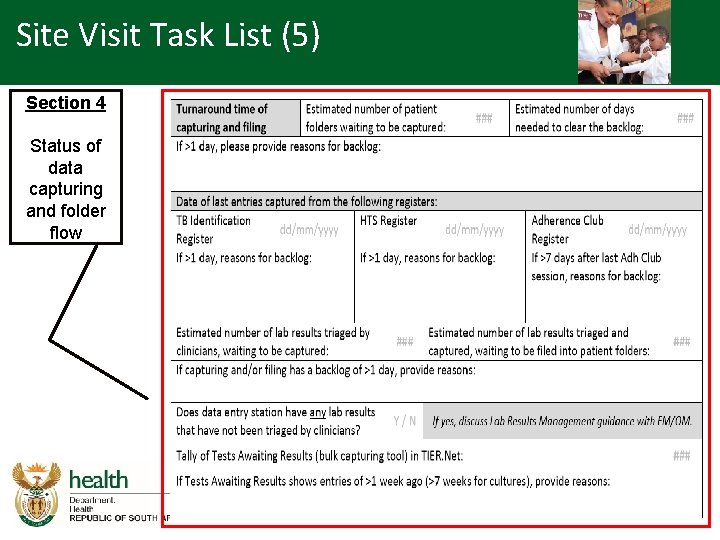
Site Visit Task List (5) Section 4 Status of data capturing and folder flow
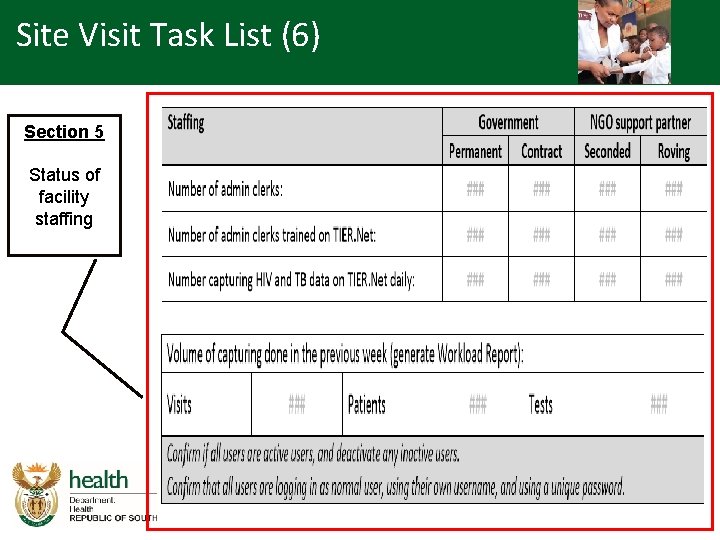
Site Visit Task List (6) Section 5 Status of facility staffing
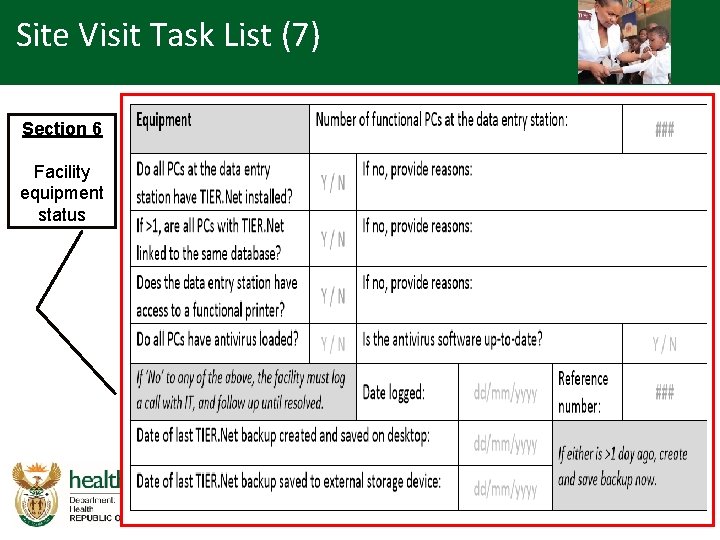
Site Visit Task List (7) Section 6 Facility equipment status

Site Visit Task List (8) Section 7 Action plan

Site Visit Task List (9) Section 8 Attendees

Thank you

Exercise

Scenario - SVTL • Participants to split into eight groups using the numbers provided during morning registration – 5 minutes • Scenario – You are a (sub)district manager and you have conducted a site visit to Hatfield CHC – During your visit you use the site visit task list to assess the maintenance of TIER. Net – During usage of the tool various issues arise (see provided attachment)

Exercise layout - SVTL • Each group to discuss the site visit task list and compile feedback – 30 minutes – Discuss what you observe from the handout copy of the site visit task list? – Discuss what are the key issues to take from the site visit task list – Develop remedial actions to address the issues • Groups must select a representative to report back on the findings and remedial actions • The feedback will be in the form of a group discussion – 30 minutes

Thank you NIT_Support@health. gov. za www. tbhivinfosys. org. za
- Slides: 107