Comprehensive Training to support the National Integrated TBHIV

Comprehensive Training to support the National Integrated TB/HIV Information System Implementation Current guidance and tools for DDU

Context (1) • The whole concept of DDU is not entirely new, clearly addressed in the current DOH policies and SOP • Guidance is provided on – what is meant by data quality – how information should be retrieved and used – how frequently the information should be used – timelines and data flow of data from facility to national

Context (2) • Guidance is not always adhered to • During facility engagements, couple of issues identified: e. g. – Non-standard data collection tools in some facilities – Non-usage of TIER. Net line lists – Data not flowing from one level to the other timeously – Outliers and missing data identified at higher levels instead of previous levels – Etc.

Governance documents • Key SOPs and Policies – Integrated TB/HIV Data Management SOP • Key role players • Data sources • THIS line lists • Data flow – DHMIS Policy • Data quality dimensions – Facility-level DHMIS SOP • Principles for implementing the SOP • Key HIS maintenance practices at sub-national levels

Governance documents

Integrated TB/HIV DM SOP – key role players • Facility SOP: – Admin Clerk (AC) – Facility and/or Operational Manager (FM/OM) – Facility/Health Information Officer (F/HIO) – Clinician – Club Manager – Mobile Clinic Nurse • Sub(district) and higher SOP: – Sub(district) managers (SDM/DM) • Information Management (IM) • HAST managers (HM) • DITs, TKIs, – Provincial managers (PM) • Information Management • PIT leads • HAST managers – National Managers 6

Integrated TB/HIV DM SOP – data sources • Data sources for capture in the TB/HIV information system – It is imperative that the prescribed standard data sources are used for digitising of information – Multiple non-standard data sources would affect quality of clinical governance – E. g. HIV clinical stationery, TB clinical stationery, HTS register, TB case ID register etc. • In line with Ideal Clinic prescript - ‘one patient, one folder number’ • Where available, Health Patient Record Number (HPRN) must be used as the main folder number

Integrated TB/HIV DM SOP - lists and reports • Line lists and reports – Important for line lists and reports to be generated at stipulated timeframes – Admin clerk is responsible for generating the line lists and reports – Facility Manager is responsible for ensuring appropriate action is taken
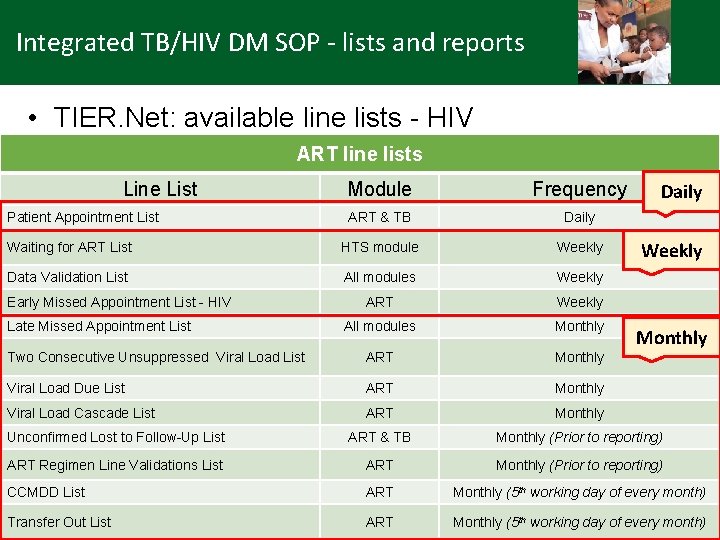
Integrated TB/HIV DM SOP - lists and reports • TIER. Net: available line lists - HIV ART line lists Line List Module Frequency ART & TB Daily Waiting for ART List HTS module Weekly Data Validation List All modules Weekly ART Weekly All modules Monthly Two Consecutive Unsuppressed Viral Load List ART Monthly Viral Load Due List ART Monthly Viral Load Cascade List ART Monthly Unconfirmed Lost to Follow-Up List ART & TB Monthly (Prior to reporting) ART Regimen Line Validations List ART Monthly (Prior to reporting) CCMDD List ART Monthly (5 th working day of every month) Transfer Out List ART Monthly (5 th working day of every month) Patient Appointment List Early Missed Appointment List - HIV Late Missed Appointment List Daily Weekly Monthly
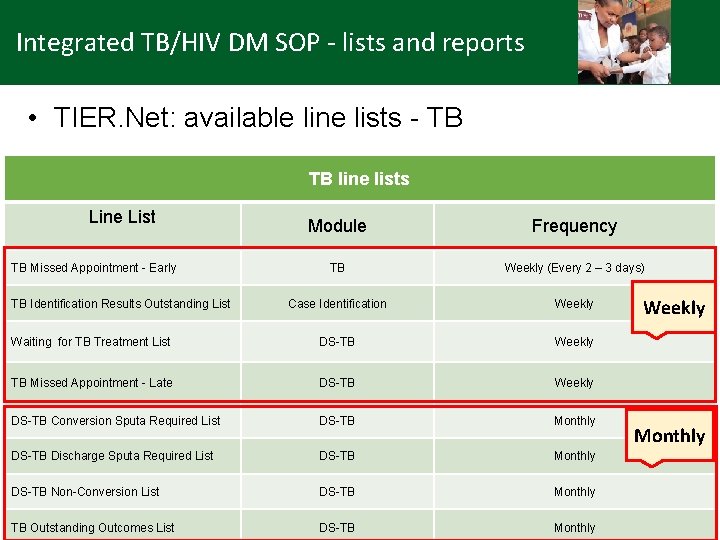
Integrated TB/HIV DM SOP - lists and reports • TIER. Net: available line lists - TB TB line lists Line List Module Frequency TB Weekly (Every 2 – 3 days) Case Identification Weekly Waiting for TB Treatment List DS-TB Weekly TB Missed Appointment - Late DS-TB Weekly DS-TB Conversion Sputa Required List DS-TB Monthly DS-TB Discharge Sputa Required List DS-TB Monthly DS-TB Non-Conversion List DS-TB Monthly TB Outstanding Outcomes List DS-TB Monthly TB Missed Appointment - Early TB Identification Results Outstanding List Weekly Monthly
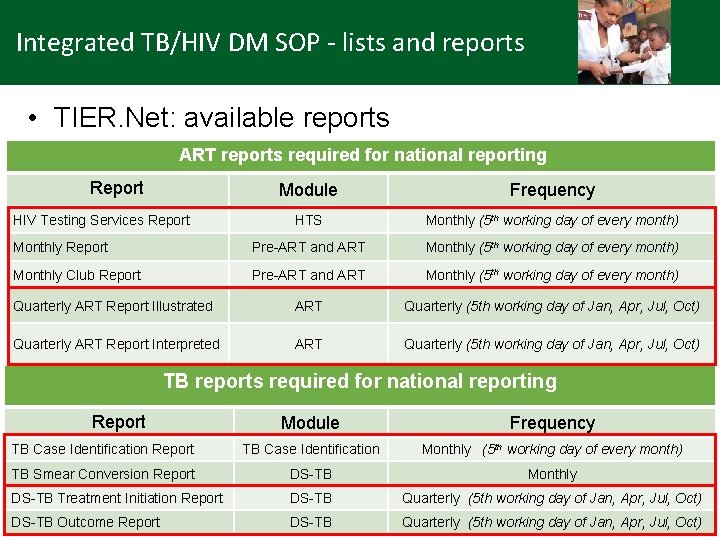
Integrated TB/HIV DM SOP - lists and reports • TIER. Net: available reports ART reports required for national reporting Report Module Frequency HTS Monthly (5 th working day of every month) Monthly Report Pre-ART and ART Monthly (5 th working day of every month) Monthly Club Report Pre-ART and ART Monthly (5 th working day of every month) Quarterly ART Report Illustrated ART Quarterly (5 th working day of Jan, Apr, Jul, Oct) Quarterly ART Report Interpreted ART Quarterly (5 th working day of Jan, Apr, Jul, Oct) HIV Testing Services Report TB reports required for national reporting Report Module Frequency TB Case Identification Report TB Case Identification Monthly (5 th working day of every month) TB Smear Conversion Report DS-TB Monthly DS-TB Treatment Initiation Report DS-TB Quarterly (5 th working day of Jan, Apr, Jul, Oct) DS-TB Outcome Report DS-TB Quarterly (5 th working day of Jan, Apr, Jul, Oct)
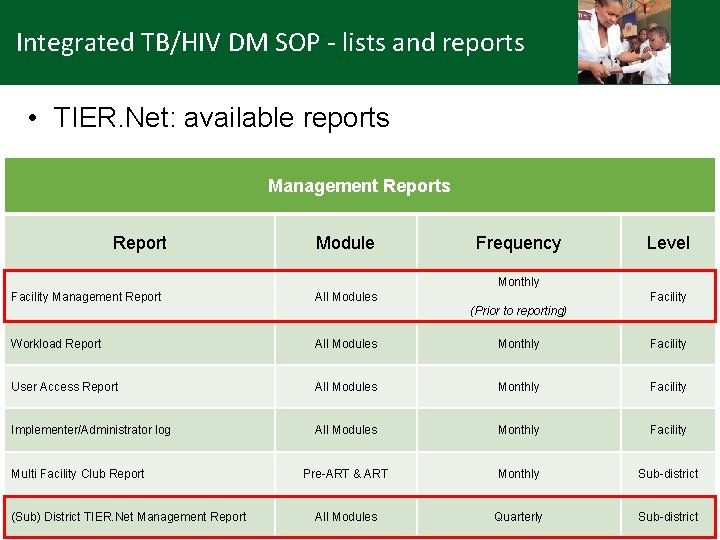
Integrated TB/HIV DM SOP - lists and reports • TIER. Net: available reports Management Reports Report Module Frequency Level Monthly Facility Management Report All Modules Facility (Prior to reporting) Workload Report All Modules Monthly Facility User Access Report All Modules Monthly Facility Implementer/Administrator log All Modules Monthly Facility Pre-ART & ART Monthly Sub-district All Modules Quarterly Sub-district Multi Facility Club Report (Sub) District TIER. Net Management Report

Integrated TB/HIV DM SOP - lists and reports • TIER. Net: available reports e. g. Facility management report Report and line lists Explanation of the alert Alert
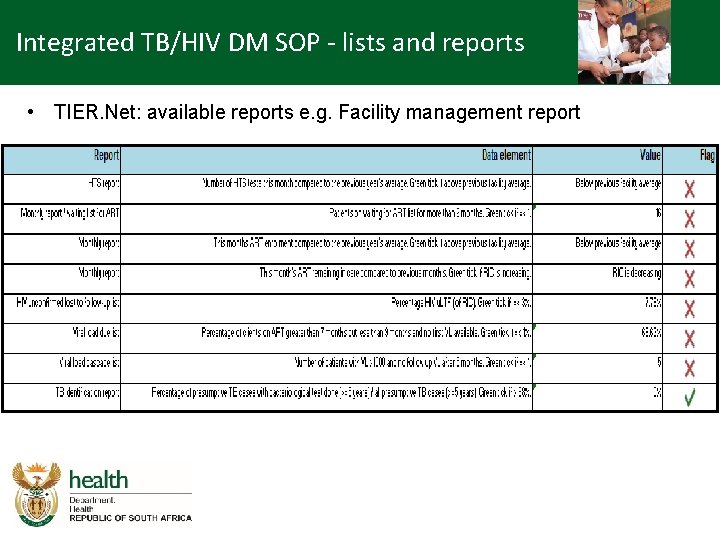
Integrated TB/HIV DM SOP - lists and reports • TIER. Net: available reports e. g. Facility management report
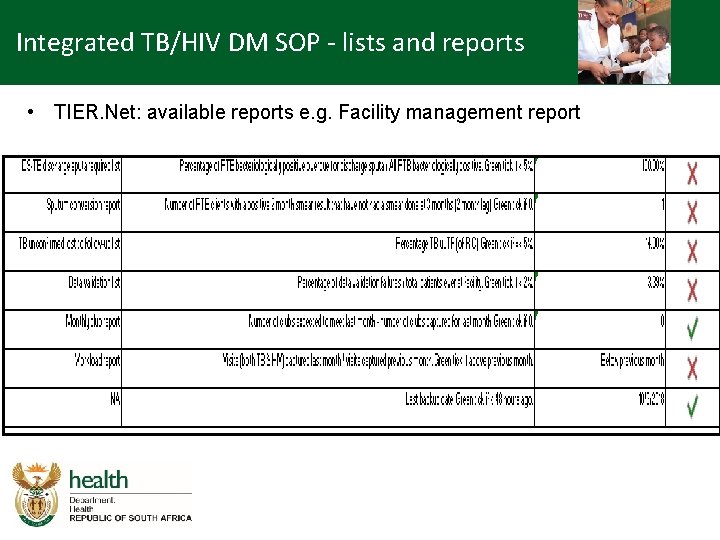
Integrated TB/HIV DM SOP - lists and reports • TIER. Net: available reports e. g. Facility management report
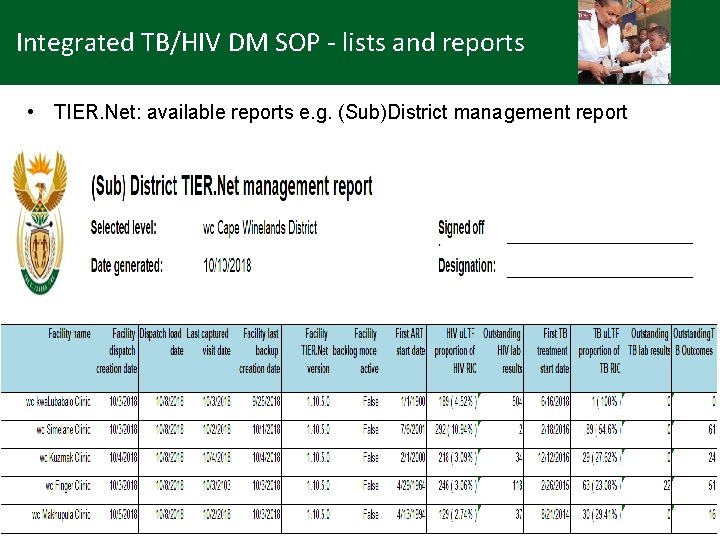
Integrated TB/HIV DM SOP - lists and reports • TIER. Net: available reports e. g. (Sub)District management report
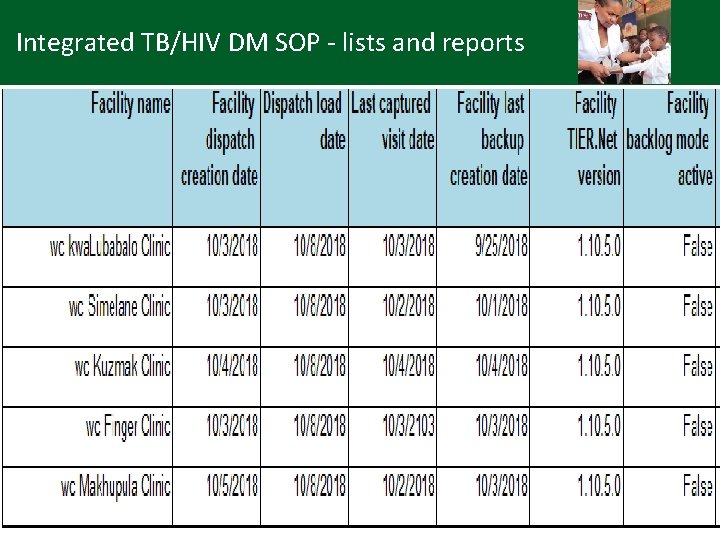
Integrated TB/HIV DM SOP - lists and reports • TIER. Net: available reports e. g. (Sub)District management report
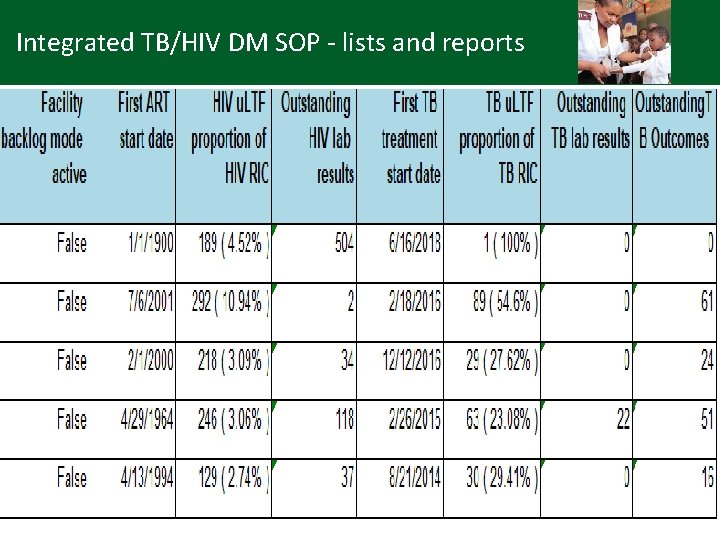
Integrated TB/HIV DM SOP - lists and reports • TIER. Net: available reports e. g. (Sub)District management report
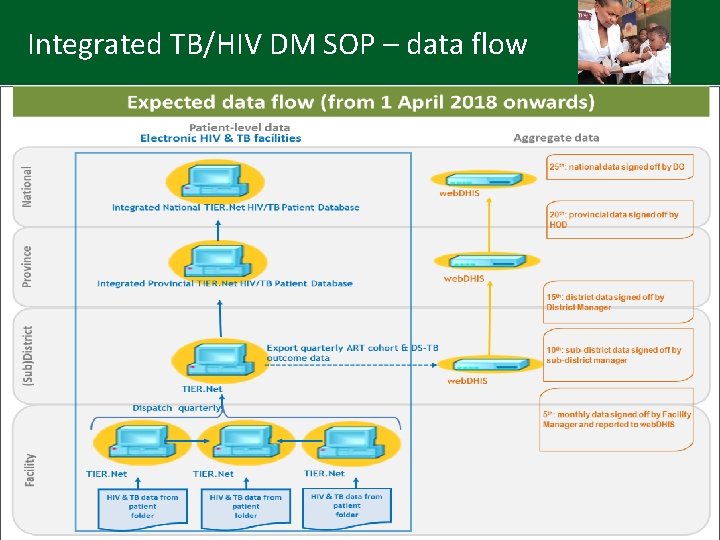
Integrated TB/HIV DM SOP – data flow

DHMIS Policy – data quality • Data quality improvement initiatives: – Data is generally considered to be of high quality if it is "fit for [its] intended uses in operations, decision making and planning“ • Who routinely interrogates data quality, and how do they do this? • What are the typical feedback mechanisms in place to address issues identified in those reviews? • External stakeholder’s role in this process (i. e. DSPs)? • WHO data quality tool?

DHMIS Policy – 8 data quality dimensions Relevance Coherence and comparability Accuracy Integrity Data Quality Timeliness Accessibility Completeness Reliability

DHMIS Policy – 8 data quality dimensions 1. Relevance – To ensure relevance of all indicators and data elements, NDo. H engages a broad range of stakeholders at all levels of health system 2. Integrity – values and related practices that maintain the confidence users have in the information itself 3. Timeliness – delays in data collection, conversion into information or data submission compromise the value of the data

DHMIS Policy – 8 data quality dimensions 4. Accessibility – ease with which users can obtain the information 5. Reliability – data should be analysed at all levels with purpose of identifying establishments with unstable data, and thus unstable service delivery 6. Completeness – All levels of health system should implement mechanisms for verifying the completeness and consistency of data

DHMIS Policy – 8 data quality dimensions 7. Accuracy – All facility managers shall be responsible for ensuring that data accuracy assessments are conducted in each facility, comparing tally sheets, registers and/or patient folders with the summary data and applying data validation rules before data is submitted for capturing 8. Coherence and comparability – Coherence = degree to which data can be successfully brought together with other similar information from different sources within a broad analytical framework overtime – Comparability = ability to compare data on the same characteristics between different points in time and geographical areas

DHMIS facility level SOP - Introduction • The DHMIS facility level SOP (2016) is an updated version of the 2013 SOPs • Clarifies responsibilities and procedures for effective management of aggregated routine health service data • The SOP is mandatory for all health information related activities for NDo. H • Facility Manager must ensure team members understand the SOPs objectives • Team members sign that they have read and understand the SOPs

DHMIS facility level SOP - Principles • Principles for implementing SOP: – Standardised set of rationalised registers – Service points allocated appropriate organisational units – Admin clerks enter data into various systems – Each patient should have a facility retained clinical record in which all services are recorded – All clinical records and data collection tools must be stored safely and access to them controlled

DHMIS facility-level SOP • Key HIS sub-national level maintenance practices: – All policies and SOPs must be available and implemented – Staff must be trained on all policies and SOPs – Each patient must have one patient folder/clinical record and file is always available in facility – Information recorded on data collection tool are consistent with patient folder and supporting documentation – All applicable patient records are captured on electronic databases, e. g. ETR. Net, TIER. Net etc. – Information captured in DHIS is consistent with data input forms

DHMIS facility-level SOP – All data collection tools used for collection of must be reviewed for quality and have been signed off – All registers reviewed for quality and have been signed-off by facility manager – All validation errors corrected or explained – All outliers explained – Processing of data updates has been completed correctly – Sign off forms are properly completed and signed by the facility manager

Thank you
- Slides: 29