Compounds Molecules Molecule The smallest identifiable unit that
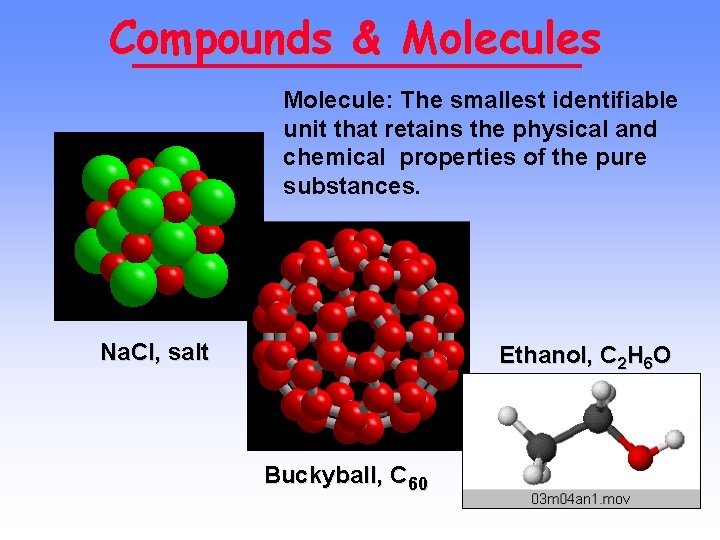
Compounds & Molecules Molecule: The smallest identifiable unit that retains the physical and chemical properties of the pure substances. Na. Cl, salt Ethanol, C 2 H 6 O Buckyball, C 60

Compounds & Molecules • COMPOUND is a combination of 2 or more elements in definite ratios by mass. • The character of each element is lost when forming a compound (e. g. , think of Na. Cl). • MOLECULES are the smallest units of a compound that retains the characteristics of the compound.

MOLECULAR FORMULAS • Formula for glycine is C 2 H 5 NO 2 (description of the composition) • In one molecule there are – 2 C atoms – 5 H atoms – 1 N atom – 2 O atoms

CONDENSED FORMULAS • Formula for glycine is NH 2 CO 2 H (composition and functional groups) • In one molecule there are – 1 NH 2 (amine group) – 1 CH 2 group – 1 CO 2 H group
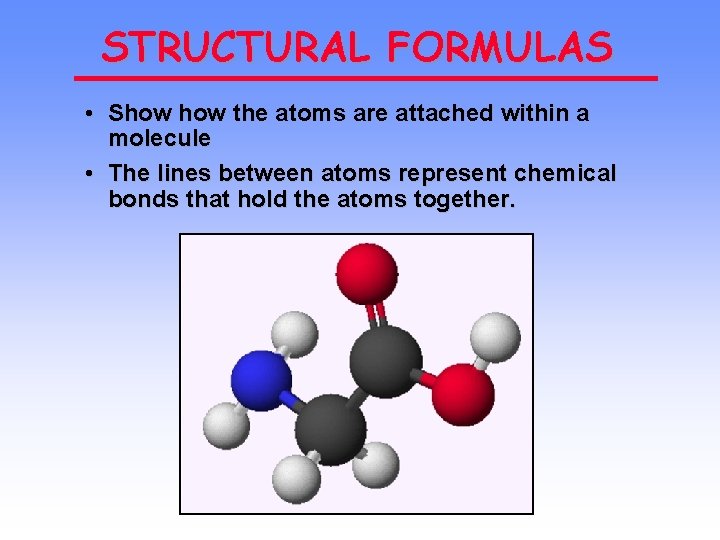
STRUCTURAL FORMULAS • Show the atoms are attached within a molecule • The lines between atoms represent chemical bonds that hold the atoms together.

WRITING FORMULAS • Can also write glycine as the condensed formula H 2 NCH 2 COOH showing functional groups (atom ordering and connectivity) • or in the form of a structural formula showing how atoms are attached to each other (bond orders)
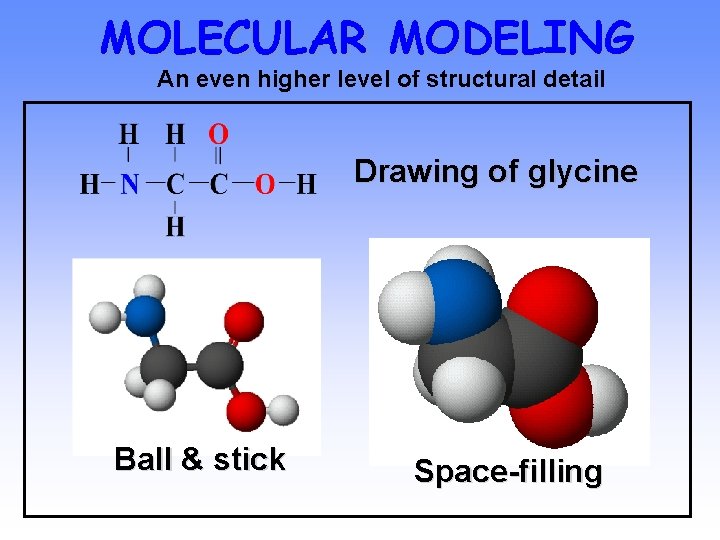
MOLECULAR MODELING An even higher level of structural detail Drawing of glycine Ball & stick Space-filling

Resources for Molecular Modeling • Modeling software –CAChe (General Chemistry Interactive CD -ROM) –Rasmol –Molden –Gaussview –Maestro

MOLECULAR WEIGHT AND MOLAR MASS Molecular weight = sum of the atomic weights of all atoms in the molecule. Molar mass = molecular weight in grams per mol.
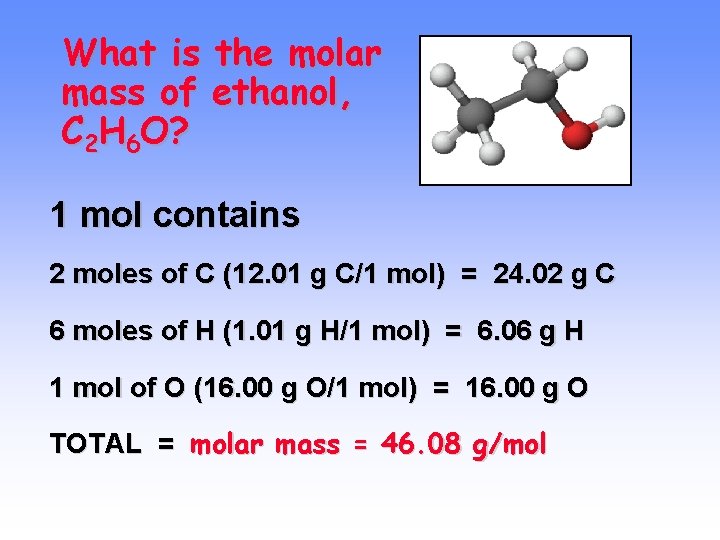
What is the molar mass of ethanol, C 2 H 6 O? 1 mol contains 2 moles of C (12. 01 g C/1 mol) = 24. 02 g C 6 moles of H (1. 01 g H/1 mol) = 6. 06 g H 1 mol of O (16. 00 g O/1 mol) = 16. 00 g O TOTAL = molar mass = 46. 08 g/mol
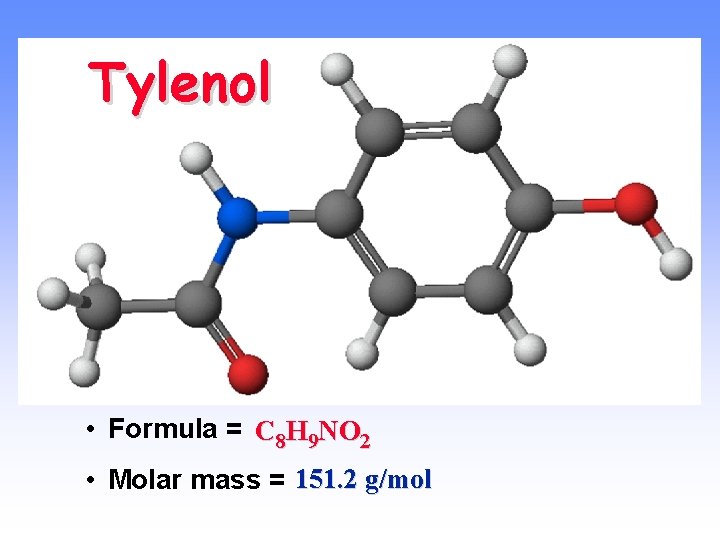
Tylenol • Formula = C 8 H 9 NO 2 • Molar mass = 151. 2 g/mol

Molar Mass

How many moles of alcohol (C 2 H 6 O) are there in a “standard” can of beer if there are 21. 3 g of C 2 H 6 O? (a) Molar mass of C 2 H 6 O = 46. 08 g/mol (b) Calc. moles of alcohol

How many molecules of alcohol are there in a “standard” can of beer if there are 21. 3 g of C 2 H 6 O? We know there are 0. 462 mol of C 2 H 6 O. = 2. 78 x 1023 molecules

How many atoms of C are there in a “standard” can of beer if there are 21. 3 g of C 2 H 6 O? There are 2. 78 x 1023 molecules. Each molecule contains 2 C atoms. Therefore, the number of C atoms is = 5. 57 x 1023 C atoms
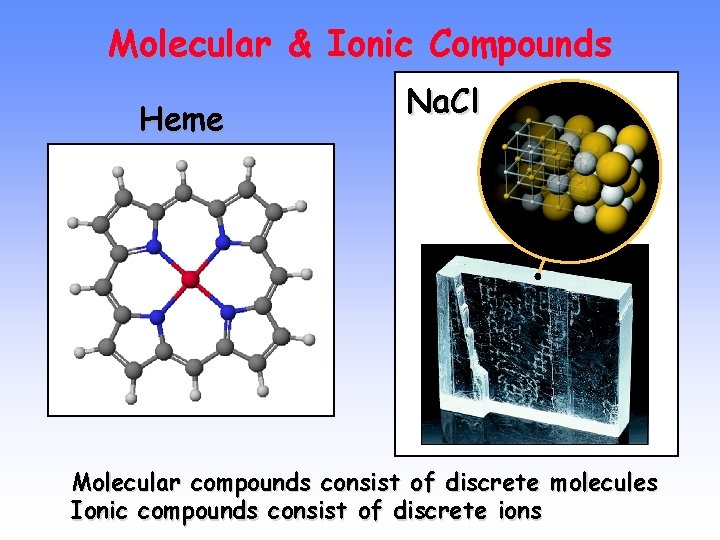
Molecular & Ionic Compounds Heme Na. Cl Molecular compounds consist of discrete molecules Ionic compounds consist of discrete ions

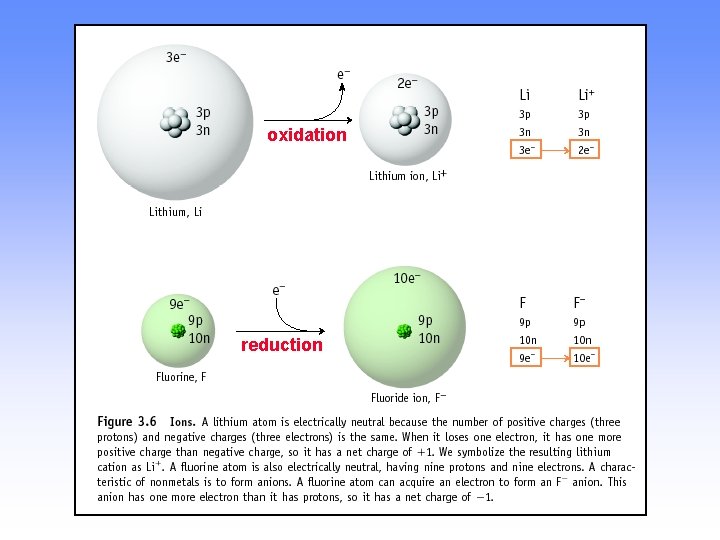
IONS AND IONIC COMPOUNDS • IONS are atoms or groups of atoms with a positive or negative charge. • Taking away an electron from an atom gives a CATION with a positive charge • Adding an electron to an atom gives an ANION with a negative charge.

Forming Cations & Anions A CATION forms when an atom loses one or more electrons. An ANION forms when an atom gains one or more electrons Mg 2+ F + e- --> F- Mg --> oxidation + 2 e- reduction
oxidation reduction

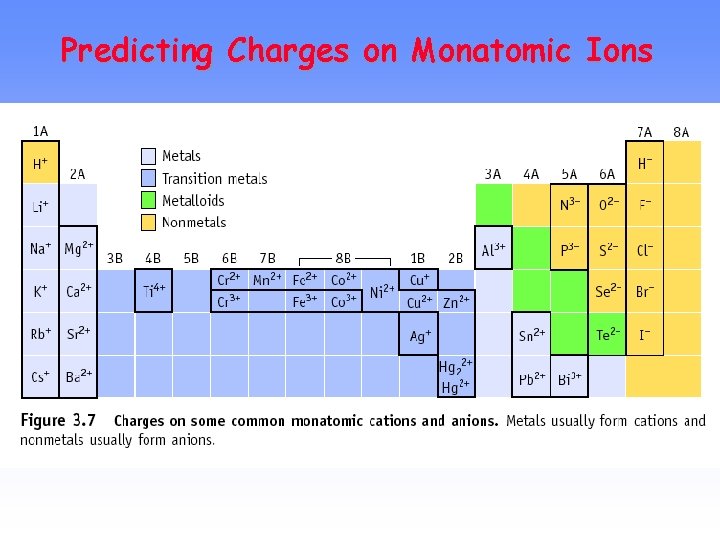
PREDICTING ION CHARGES In general • metals (Mg) lose electrons ---> cations • nonmetals (F) gain electrons ---> anions

Charges on Common Ions Cation charge=group # +1 +2 Anion charge=group #-8 -4 -3 -2 -1 +3 By losing or gaining e-, atom has same number of electrons as nearest Group 8 A atom.
Predicting Charges on Monatomic Ions

METALS M ---> n e- + Mn+ where n = periodic group Na+ sodium ion Mg 2+ magnesium ion Al 3+ aluminum ion Transition metals --> M 2+ or M 3+ are common Fe 2+ iron(II) ion Fe 3+ iron(III) ion
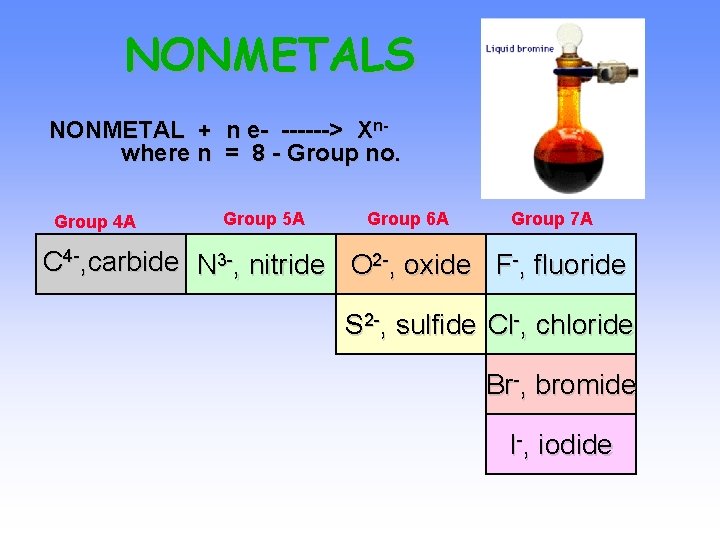
NONMETALS NONMETAL + n e- ------> Xnwhere n = 8 - Group no. Group 4 A Group 5 A Group 6 A Group 7 A C 4 -, carbide N 3 -, nitride O 2 -, oxide F-, fluoride S 2 -, sulfide Cl-, chloride Br-, bromide I-, iodide

Ion Formation Reaction of aluminum and bromine

POLYATOMIC IONS CD Screen 3. 6 Groups of atoms with a charge. MEMORIZE the names and formulas of common polyatomic ions listed in Table 3. 1, page 107 (next slide)

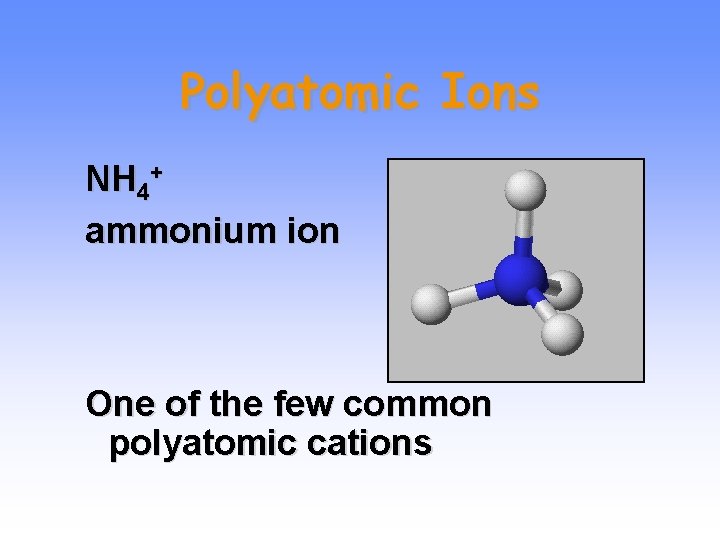
Polyatomic Ions NH 4+ ammonium ion One of the few common polyatomic cations
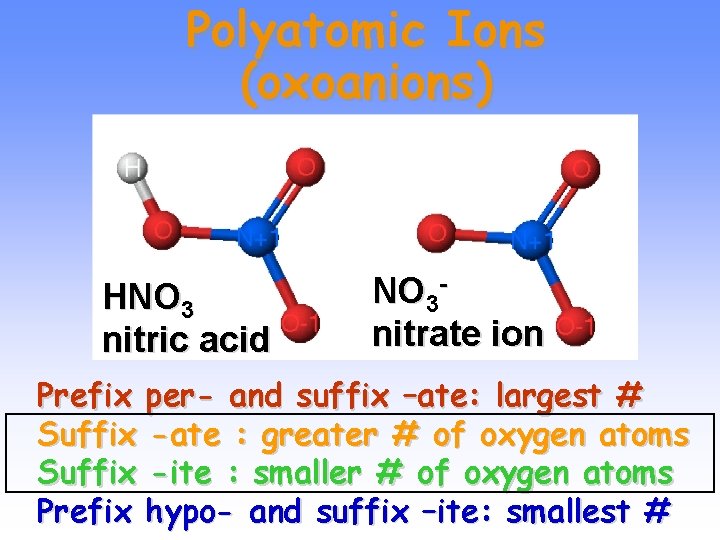
Polyatomic Ions (oxoanions) HNO 3 nitric acid NO 3 nitrate ion Prefix per- and suffix –ate: largest # Suffix -ate : greater # of oxygen atoms Suffix -ite : smaller # of oxygen atoms Prefix hypo- and suffix –ite: smallest #
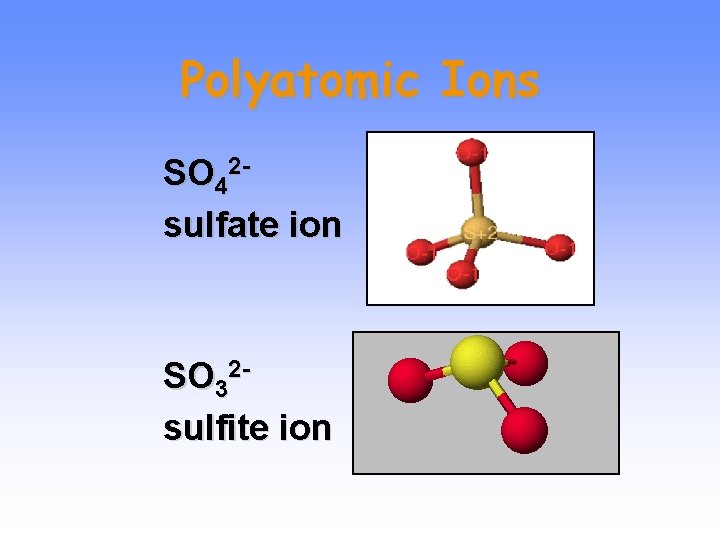
Polyatomic Ions SO 42 sulfate ion SO 32 sulfite ion
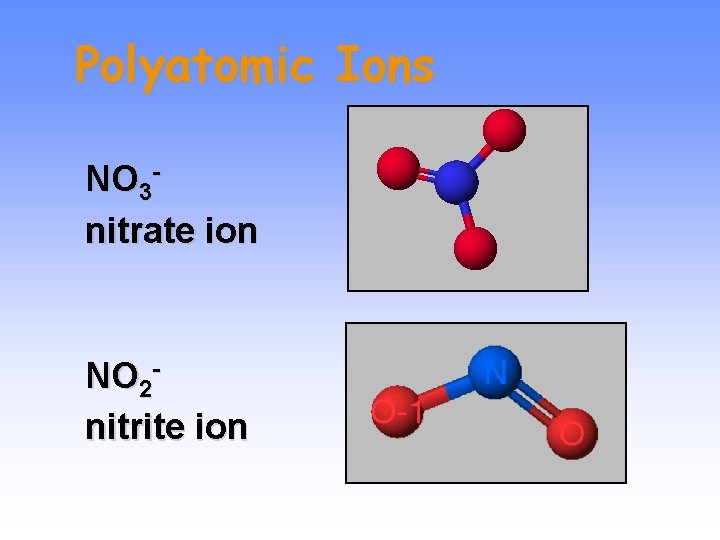
Polyatomic Ions NO 3 nitrate ion NO 2 nitrite ion
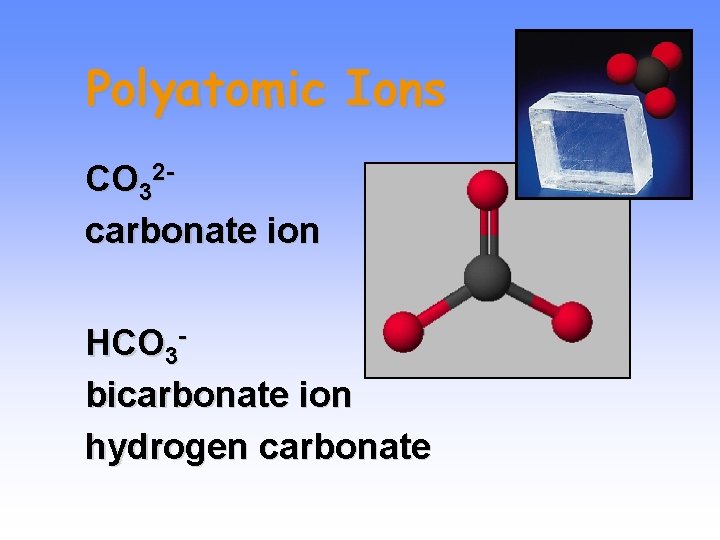
Polyatomic Ions CO 32 carbonate ion HCO 3 bicarbonate ion hydrogen carbonate
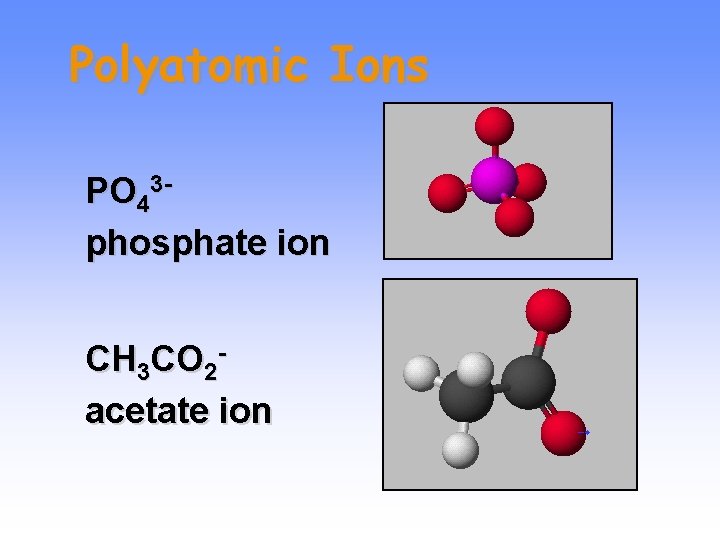
Polyatomic Ions PO 43 phosphate ion CH 3 CO 2 acetate ion

COMPOUNDS FORMED FROM IONS CATION + ANION ---> COMPOUND Na+ + Cl- --> Na. Cl A neutral compd. requires equal number of + and - charges.
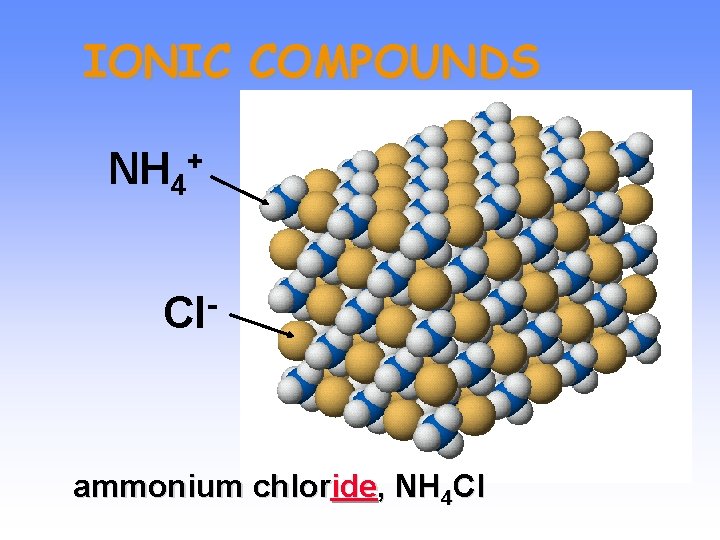
IONIC COMPOUNDS NH 4 + Cl ammonium chloride, NH 4 Cl
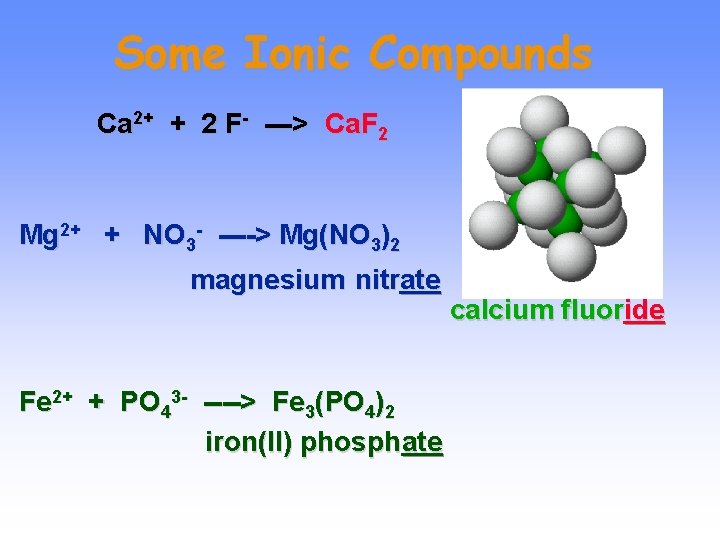
Some Ionic Compounds Ca 2+ + 2 F- ---> Ca. F 2 Mg 2+ + NO 3 - ----> Mg(NO 3)2 magnesium nitrate Fe 2+ + PO 43 - ----> Fe 3(PO 4)2 iron(II) phosphate calcium fluoride

Properties of Ionic Compounds Forming Na. Cl from Na and Cl 2 • A metal atom can transfer an electron to a nonmetal. • The resulting cation and anion are attracted to each other by electrostatic forces.

Electrostatic Forces The oppositely charged ions in ionic compounds are attracted to one another by ELECTROSTATIC FORCES. These forces are governed by COULOMB’S LAW.

Electrostatic Forces COULOMB’S LAW As ion charge increases, the attractive force ________. As the distance between ions increases, the attractive force ________. This idea is important and will come up many times in future discussions!
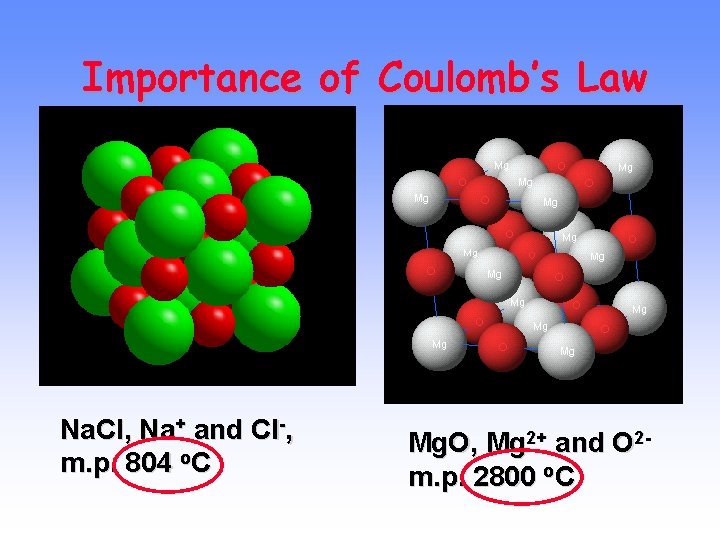
Importance of Coulomb’s Law Na. Cl, Na+ and Cl-, m. p. 804 o. C Mg. O, Mg 2+ and O 2 m. p. 2800 o. C

ELEMENTS THAT EXIST AS MOLECULES Allotropes of C See SCREEN 3. 2 on the CD-ROM

Screen 3. 2
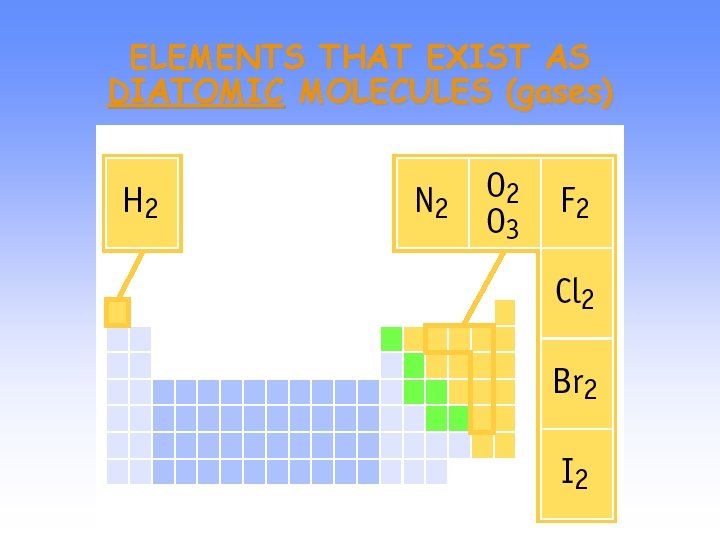
ELEMENTS THAT EXIST AS DIATOMIC MOLECULES (gases)

ELEMENTS THAT EXIST AS POLYATOMIC MOLECULES S 8 sulfur molecules White P 4 and polymeric red phosphorus
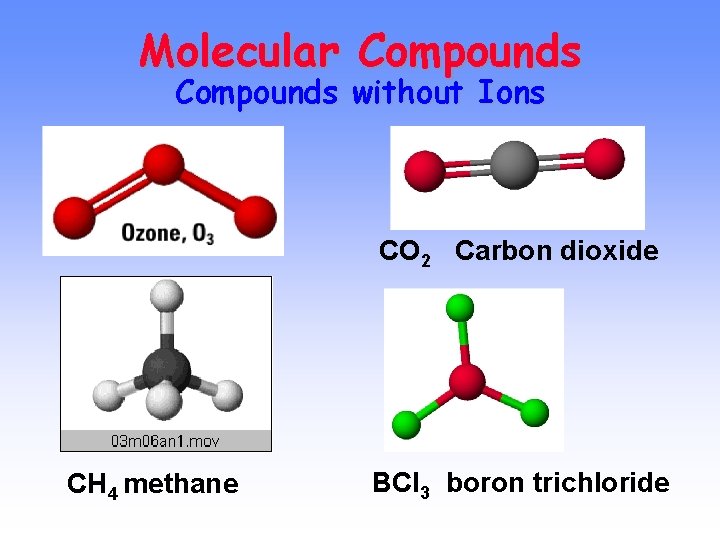
Molecular Compounds without Ions CO 2 Carbon dioxide CH 4 methane BCl 3 boron trichloride
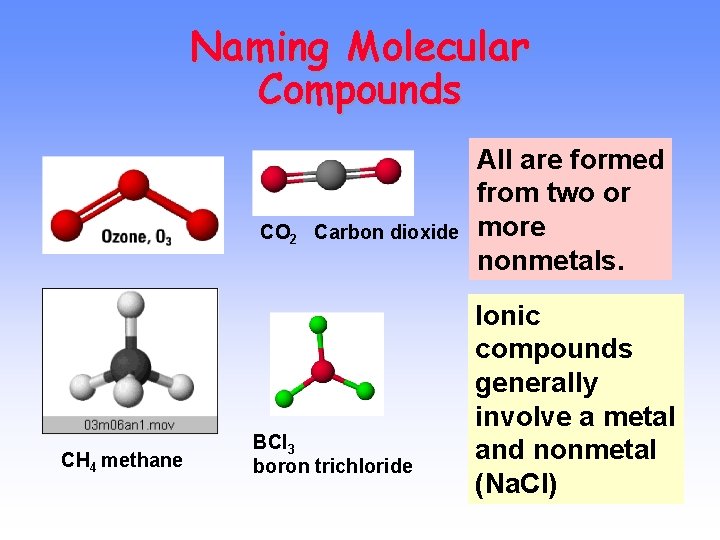
Naming Molecular Compounds CO 2 Carbon dioxide CH 4 methane BCl 3 boron trichloride All are formed from two or more nonmetals. Ionic compounds generally involve a metal and nonmetal (Na. Cl)
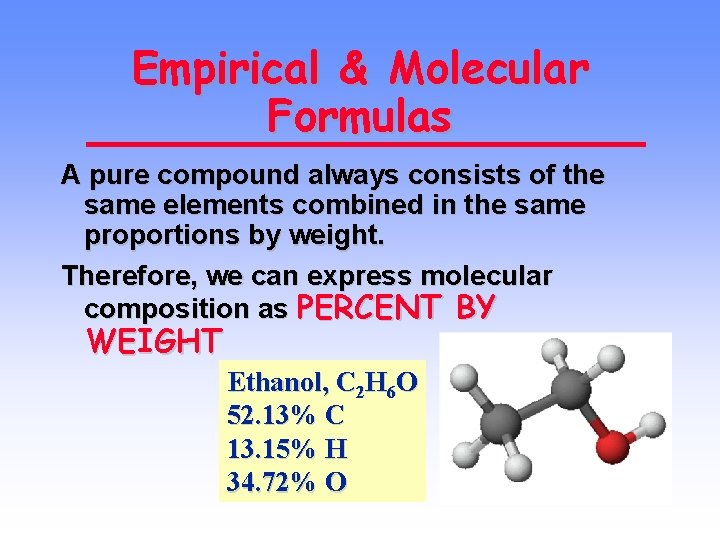
Empirical & Molecular Formulas A pure compound always consists of the same elements combined in the same proportions by weight. Therefore, we can express molecular composition as PERCENT BY WEIGHT Ethanol, C 2 H 6 O 52. 13% C 13. 15% H 34. 72% O

Percent Composition Consider some of the family of nitrogenoxygen compounds: NO 2, nitrogen dioxide and closely related, NO, nitrogen monoxide (or nitric oxide) Chemistry of NO, nitrogen monoxide Structure of NO 2

Percent Composition Consider NO 2, Molar mass = ? What is the weight percent of N and of O? What are the weight percentages of N and O in NO?

Determining Formulas In chemical analysis we determine the % by weight of each element in a given amount of pure compound and derive the EMPIRICAL or SIMPLEST formula. PROBLEM: A compound of B and H is 81. 10% B. What is its empirical formula?

A compound of B and H is 81. 10% B. What is its empirical formula? • Because it contains only B and H, it must contain 18. 90% H. • In 100. 0 g of the compound there are 81. 10 g of B and 18. 90 g of H. • Calculate the number of moles of each constitutent.

A compound of B and H is 81. 10% B. What is its empirical formula? Calculate the number of moles of each element in 100. 0 g of sample.

A compound of B and H is 81. 10% B. What is its empirical formula? Now, recognize that atoms combine in the ratio of small whole numbers Find the ratio of moles of elements in the compound.

A compound of B and H is 81. 10% B. What is its empirical formula? Take the ratio of moles of B and H. Always divide by the smaller number. But we need a whole number ratio. 2. 5 mol H/1. 0 mol B = 5 mol H to 2 mol B EMPIRICAL FORMULA = B 2 H 5
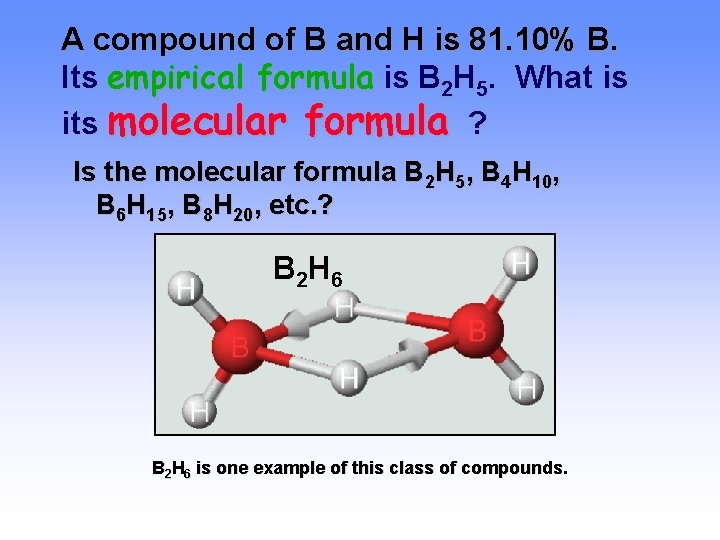
A compound of B and H is 81. 10% B. Its empirical formula is B 2 H 5. What is its molecular formula ? Is the molecular formula B 2 H 5, B 4 H 10, B 6 H 15, B 8 H 20, etc. ? B 2 H 6 is one example of this class of compounds.

A compound of B and H is 81. 10% B. Its empirical formula is B 2 H 5. What is its molecular formula? We need to do an EXPERIMENT to find the MOLAR MASS. Here experiment gives 53. 3 g/mol Compare with the mass of B 2 H 5 = 26. 66 g/unit Find the ratio of these masses. Molecular formula = B 4 H 10

How to Determine the molar mass? Mass spectrometer
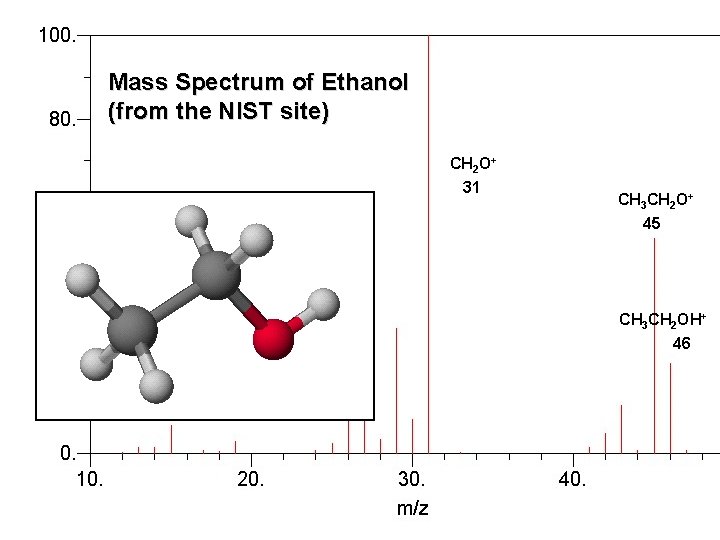
Mass Spectrum of Ethanol (from the NIST site) CH 2 O+ 31 CH 3 CH 2 O+ 45 CH 3 CH 2 OH+ 46

Determine the formula of a compound of Sn and I using the following data. • • Reaction of Sn and I 2 is done using excess Sn. Mass of Sn in the beginning = 1. 056 g Mass of iodine (I 2) used = 1. 947 g Mass of Sn remaining = 0. 601 g

Tin and Iodine Compound Find the mass of Sn that combined with 1. 947 g I 2. Mass of Sn initially = 1. 056 g Mass of Sn recovered = 0. 601 g Mass of Sn used = 0. 455 g Find moles of Sn used:

Tin and Iodine Compound Now find the number of moles of I 2 that combined with 3. 83 x 10 -3 mol Sn. Mass of I 2 used was 1. 947 g. How many moles of iodine atoms? = 1. 534 x 10 -2 mol I atoms

Tin and Iodine Compound Now find the ratio of number of moles of I and Sn that combined. Empirical formula is Sn. I 4
- Slides: 62