Compounds Ionic compounds Compound between Nonmetal and metal

Compounds

Ionic compounds • Compound between Non-metal and metal • Ionic bond • Large difference in electronegativity – (how much the atom wants an electron) • Found in solid form at SATP • Conducts electricity in aqueous solution • When combined the cation is used followed by the anion, the cation is used in full and the anion has a –ide suffix: oxide, fluoride

Cont’d • Multi valent metals – Different possibilities for ion charge • Iron, Copper • Check period table for common ion charge
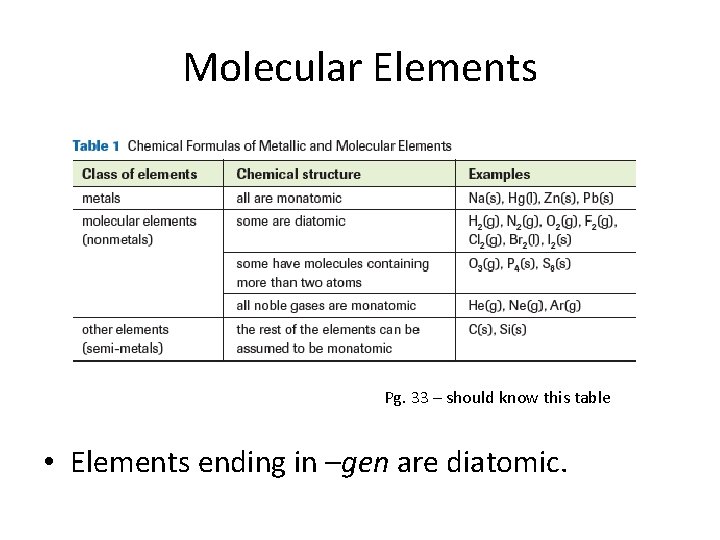
Molecular Elements Pg. 33 – should know this table • Elements ending in –gen are diatomic.

Molecular Compounds • • • Compounds of non-metals Covalent bond Small difference in electronegativity Found in all three states of matter at SATP Does not conduce electricity

Acid and Base Compounds • Identified by the H- prefix and occasionally the –COOH on the end of the compound • Found in all three states of matter at SATP • Conduce electricity in aqueous solution • Made of non-metals – assumed to be ionic • Acid p. H below 7 – aqueous hydrogen • Base p. H above 7 – aqueous hydroxides

Acid naming • Classical naming (for information) – hydrogen _____ide -> hydro_____ic acid – hydrogen _____ate -> ______ic acid – hydrogen _____ite -> ____ous acid – hydrogen hypo_____-> hypo_____ous • IPUAC naming (good to know basic) – hydrogen ______

States of matter in a compound • The state of matter is also given in the compound • i. e. H 20(s), H 20(l), H 20(g) • Na. Cl(s) or Na. Cl(aq) • Aqueous solution – the solvent is water (dissolved in water) - homogenous mixture in one phase • Solvent – a substance that dissolves a solute • Solute – substance that is dissolved into a solution
- Slides: 8