Compounds ionic bonds covalent bonds and naming compounds

Compounds, ionic bonds, covalent bonds, and naming compounds THE STRUCTURE OF MATTER

What is a compound? �A compound is made of two or more elements that are chemically combined � The forces that hold the atoms together are called chemical bonds

Why do elements form compounds? � To make themselves stable - How do atoms make themselves stable? � By filling their outer most energy level (orbital) - 1 st energy level – 2 electrons to be filled -2 nd energy level – 8 electrons to be filled - 3 rd energy level – 8 electrons to be filled � By filling their outer shell, each atom will have a similar structure to a noble gas
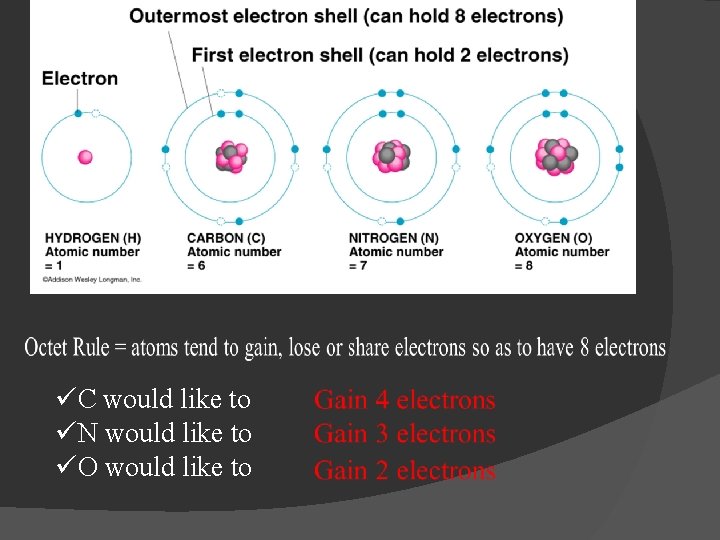
üC would like to üN would like to üO would like to

Chemical Bonds hold electrons together � There � Ionic are two types of chemical bonds: Bonds – strong bonds � Covalent Bonds – not as strong

Ionic Bonds � Bond formed by the Transfer of electrons from one atom to another � Occurs between a metal and a nonmetal � Metals lose electrons and are electron donors � Nonmetals gain an electron and are electron acceptors
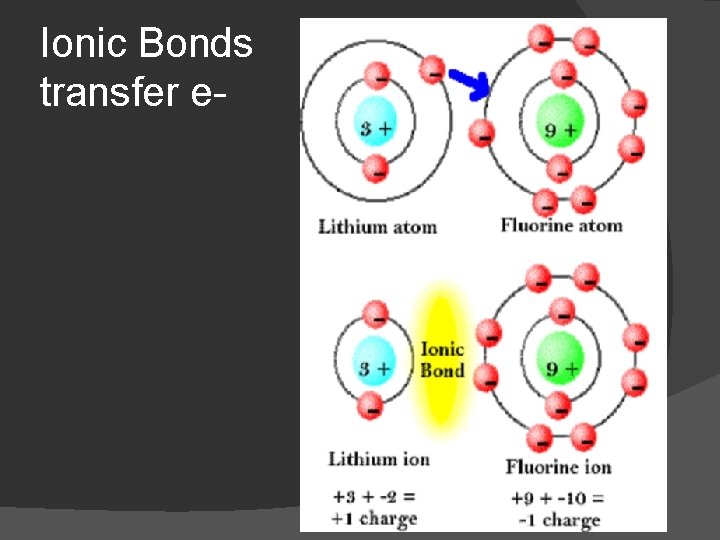
Ionic Bonds transfer e-

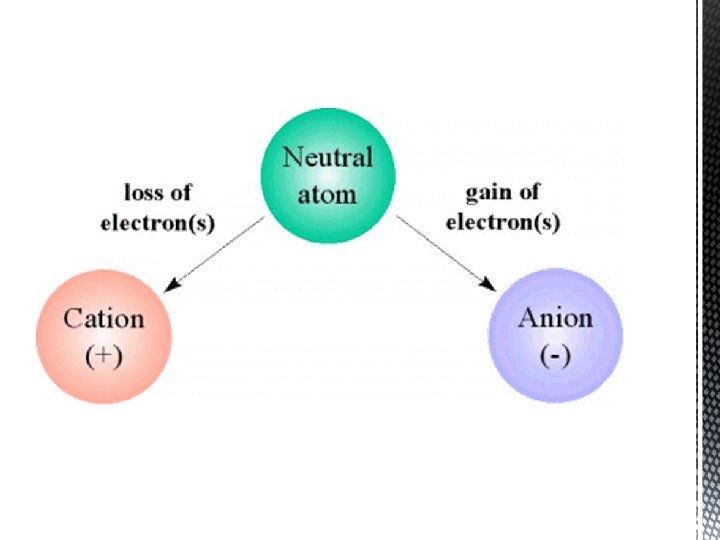
What is an Ion? � An Ion is an atom that has gained or lost an electron which gives the atom as a whole a positive or negative charge

Cation and Anion � When an atom gains or loses an electron when they bond we give it a new name Cations - are atoms that lose an election so they become more positive(+) - Metals � Anions- are atoms that gain an electron so they become more negative(-) - Non-meals �

Ionic Bonds
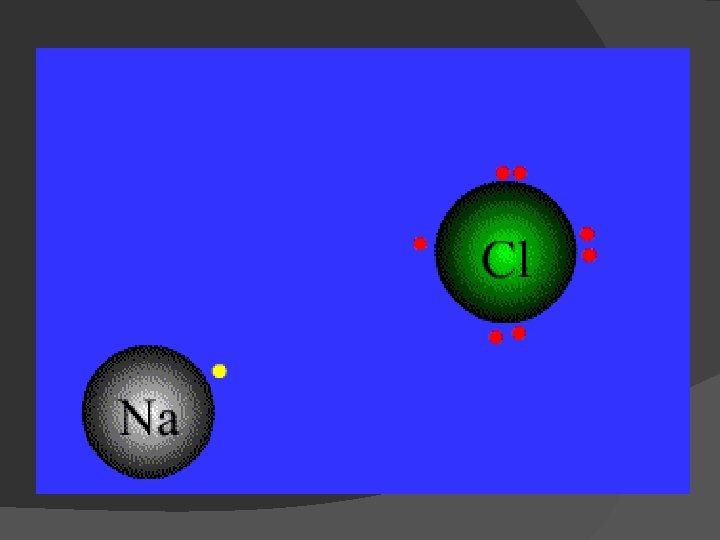
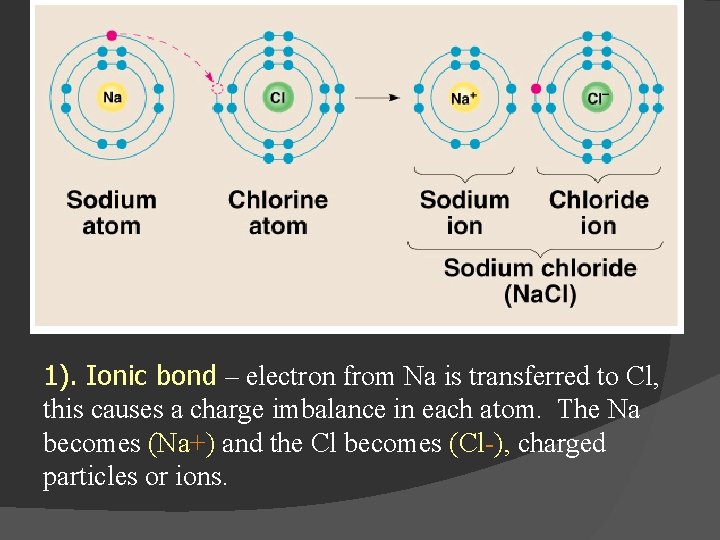
1). Ionic bond – electron from Na is transferred to Cl, this causes a charge imbalance in each atom. The Na becomes (Na+) and the Cl becomes (Cl-), charged particles or ions.

Covalent Bonds � Bond formed by the sharing of electrons � Occurs between nonmetals and other nonmentals � Not as strong of a bond as ionic bond
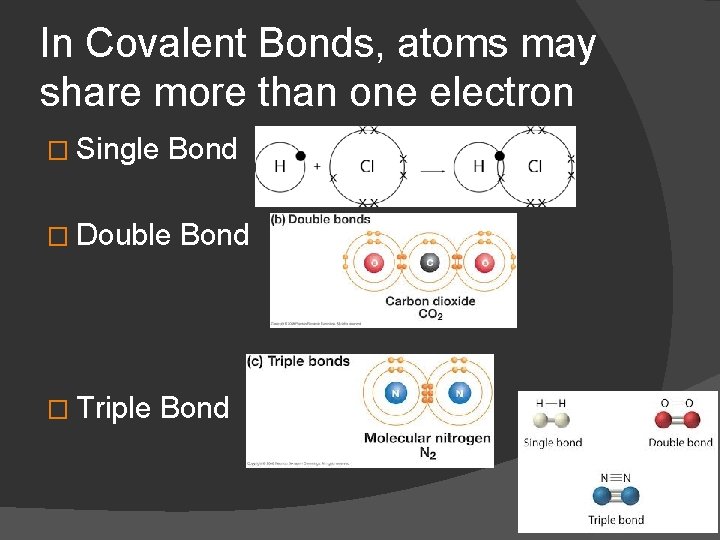
In Covalent Bonds, atoms may share more than one electron � Single Bond � Double � Triple Bond

Naming Ionic Compounds � Ionic Compounds – named for the cations(+ metal) and anion(- nonmetal)) they are made of � Cation comes first � Anion comes second + -ide on the end of the element

Steps to name ionic compounds Identify the cation (metal) 2. Identify the anion (nonmetal) 3. Change the anion (nonmetal) by adding –ide to the end 1. Ex: Na. Cl - Sodium Chloride KBr Ca. O

Writing the Formulas of Ionic Compounds � Ionic compounds must have a total charge of zero � Ex: Calcium has a +2 charge (Ca 2+) Fluoride has a – 1 charge (F 1 -) To make Calcium Fluoride equal 0 you need: 1 Calcium and 2 Fluorine atoms So the formula for calcium fluoride would be Ca. F 2

Steps to writing Ionic formulas Identify the charge of the cation (+1, +2, +3) 2. Identify the charge of the anion (-1, -2, -3) 3. Balance the charges so they equal zero 4. The charge of the cation and anion becomes a subscript (little number at the bottom) 1.

Steps to writing Ionic formulas Example: Zinc Chloride 1. Zinc +2 2. Chlorine -1 3. Balance: 1 Zinc and 2 Chlorine = 0 4. Zn. Cl 2

Steps to writing Ionic formulas � Example: Sodium Nitride 1. Na +1 2. N -3 3. Balance: Need 3 Na for every 1 N 4. Na 3 N
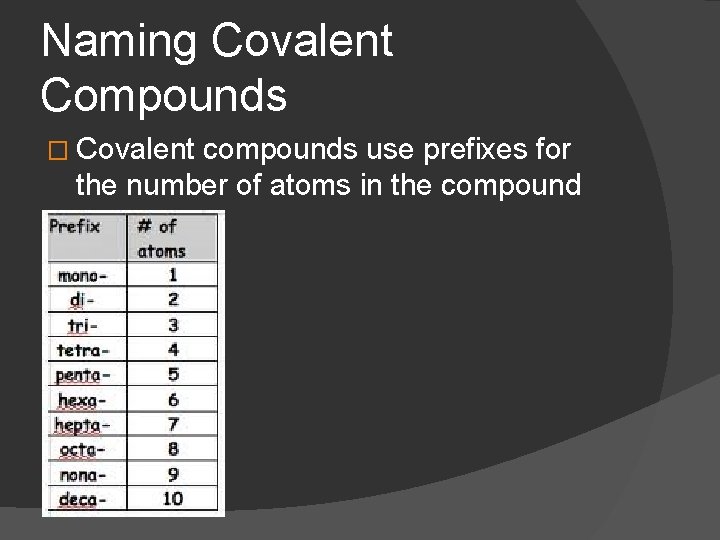
Naming Covalent Compounds � Covalent compounds use prefixes for the number of atoms in the compound

Steps to Naming Covalent Compounds Both are nonmetals Use prefixes to indicate how many atoms are present 3. Name the first element: prefix + full name 4. Name second element: prefix+ full name+ ide at the end 5. Don’t use “mono” with the first element Example: BF 3 – Boron Trifluoride SO 3 – Sulfur Trioxide 1. 2.

Steps to writing Covalent Compounds Identify the elements 2. Identify the prefixes of the element 3. Charges don’t matter for these formulas 1. Examples: Carbon dioxide = 1 carbon, 2 oxygen = CO 2 Hydrogen Monoxide =1 Hydrogen, 1 Oxygen = HO Dihydrogen monoxide = H 2 O Phosphorus Hexabromide = PBr 6
- Slides: 26