COMPOUNDS Chapter Sixteen Compounds 16 1 Chemical Bonds

COMPOUNDS

Chapter Sixteen: Compounds Ø 16. 1 Chemical Bonds and Electrons Ø 16. 2 Chemical Formulas Ø 16. 3 Molecules and Carbon Compounds

Chapter 16. 1 Learning Goals ØInfer the relationship between the number of valence electrons and the behavior of atoms. ØCompare and contrast ionic and covalent bonding. ØDraw Lewis diagrams to represent the valence electrons of atoms.

Investigation 16 A Chemical Bonds ØKey Question: Why do atoms form chemical bonds?
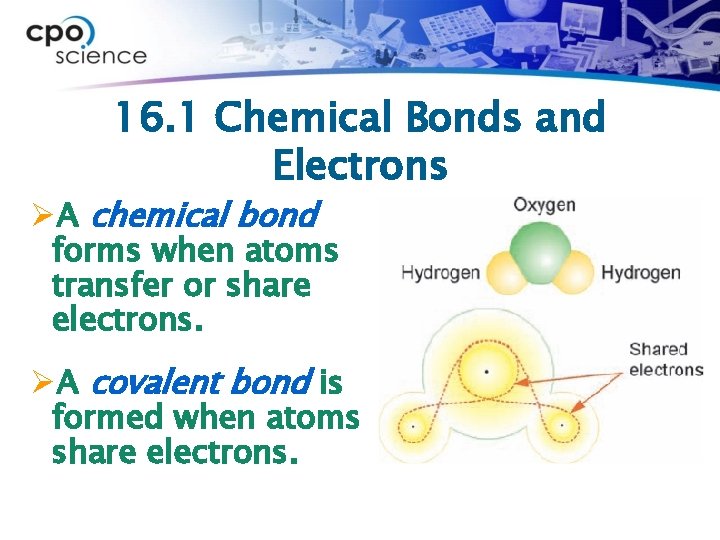
16. 1 Chemical Bonds and Electrons ØA chemical bond forms when atoms transfer or share electrons. ØA covalent bond is formed when atoms share electrons.

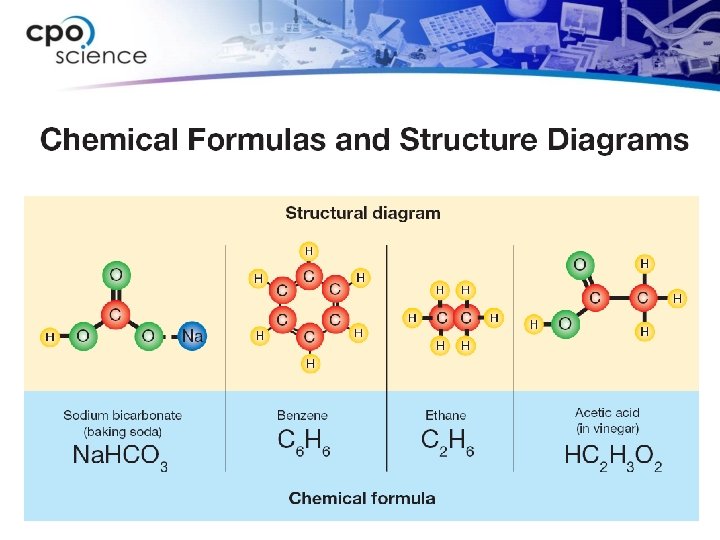
16. 1 Chemical formulas ØA molecule’s chemical formula tells you the ratio of atoms of each element in the compound.

16. 1 Ionic bonds ØNot all compounds are made of molecules. ØIonic bonds are bonds in which electrons are transferred from one atom to another. Sodium and chlorine form an ionic bond because the positive sodium ion is attracted to the negative chloride ion.

16. 1 Why chemical bonds form ØIt takes energy to separate atoms that are bonded together. ØThe same energy is released when chemical bonds form. ØAtoms form bonds to reach a lower energy state.

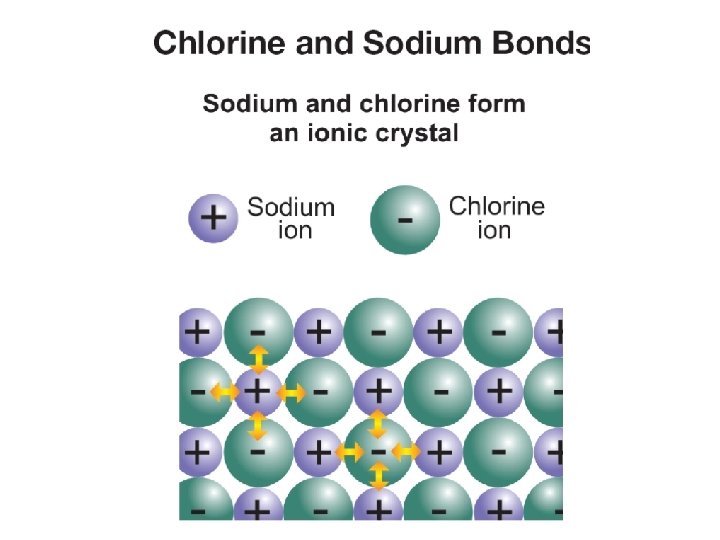
16. 1 Reactivity ØIn chemistry, reactive means an element readily forms chemical bonds, often releasing energy. ØSome elements are more reactive than others. ØThe closer an element is to having the same number of electrons as a noble gas, the more reactive the element is.


16. 1 Valence electrons ØChemical bonds are formed only between the electrons in the highest unfilled energy level. ØThese electrons are called valence electrons.
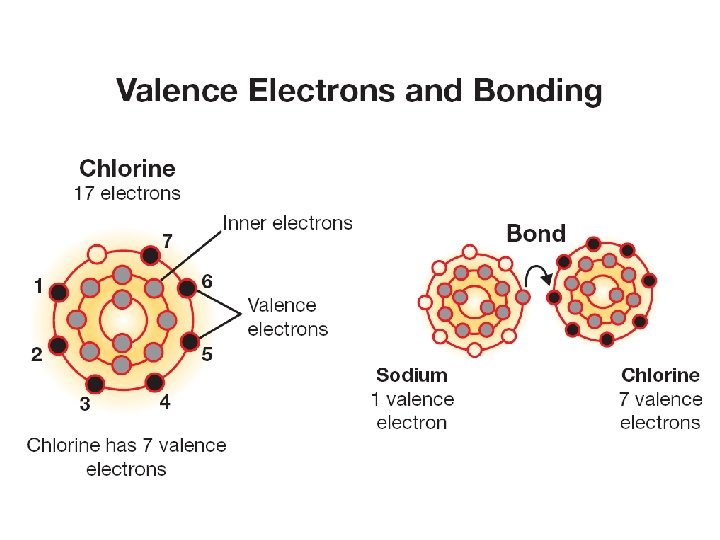

16. 1 Valence electrons and the periodic table ØGoing from left to right across a period each new element has one more valence electron than the one before it. How many valence electrons does nitrogen have?

16. 1 Valence electrons and the periodic table ØOxygen combines with one beryllium atom because beryllium can supply two valence electrons to give oxygen its preferred number of 8.

16. 1 Valence electrons and the periodic table ØCarbon has four valence electrons. ØTwo oxygen atoms can bond with a single carbon atom, each oxygen sharing two of carbon’s four valence electrons. ØThe bonds in carbon dioxide (CO 2) are double bonds because each bond involves 2 electrons.

16. 1 Lewis dot diagrams ØA clever way to keep track of valence electrons is to draw Lewis dot diagrams. ØA dot diagram shows the element symbol surrounded by one to eight dots representing the valence electrons. What is the dot structure for nitrogen?



Chapter Sixteen: Compounds Ø 16. 1 Chemical Bonds and Electrons Ø 16. 2 Chemical Formulas Ø 16. 3 Molecules and Carbon Compounds

Chapter 16. 2 Learning Goals ØUse the periodic table to make predictions about whether atoms will most likely form ionic or covalent bonds. ØDescribe how oxidations numbers can be used to write chemical formulas of compounds. ØCorrectly name chemical compounds.

Investigation 16 B Chemical Formulas ØKey Question: Why do atoms combine in certain ratios?

16. 2 Chemical Formulas and Oxidation Numbers ØAll compounds have an electrical charge of zero (they are neutral). ØAn oxidation number indicates the charge on the atom (or ion) when electrons are lost, gained, or shared in chemical bonds.

16. 2 Oxidation Numbers ØA sodium atom always ionizes to become Na+ (a charge of +1) when it combines with other atoms to make a compound. ØTherefore, we say that sodium has an oxidation number of 1+. What is chlorine’s oxidation number?
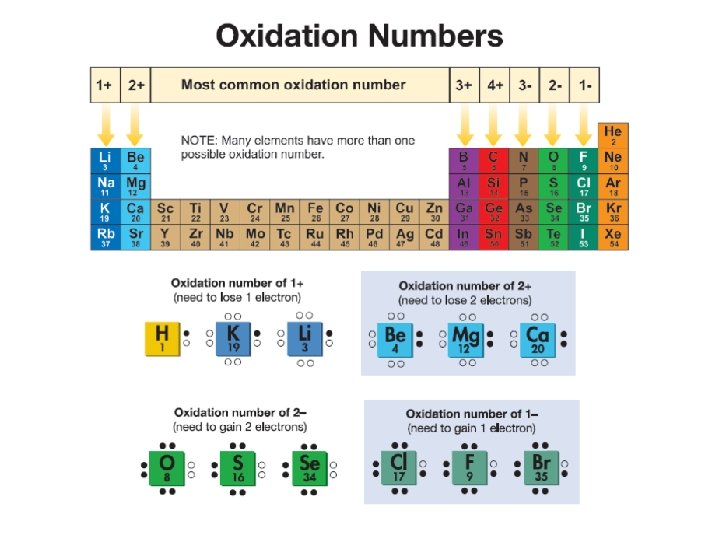

16. 2 Ionic bonds ØOn the periodic table, strong electron donors are the left side (alkali metals). ØStrong electron acceptors are on the right side (halogens). ØThe further apart two elements are on the periodic table, the more likely they are to form an ionic compound.


16. 2 Covalent bonds ØCovalent compounds form when elements have roughly equal tendency to accept electrons. ØElements that are both nonmetals and therefore close together on the periodic table tend to form covalent compounds.

16. 2 Oxidation numbers and chemical formulas ØRemember, the oxidation numbers for all the atoms in a compound must add up to zero.


16. 2 Oxidation numbers ØSome periodic tables list multiple oxidation numbers for most elements. ØThis is because more complex bonding is possible.


Solving Problems ØIron and oxygen combine to form a compound. Iron (Fe) has an oxidation number of 3+. Oxygen (O) has an oxidation number of 2–. ØPredict the chemical formula of this compound.

Solving Problems 1. Looking for: Ø …formula for a binary compound 2. Given Ø … Fe 3+ and O 2– 3. Relationships: Ø Write the subscripts so that the sum of the oxidation numbers equals zero. 4. Solution Ø Two iron atoms = 2 × (3+) = 6+ Ø Three oxygen atoms = 3 × (2–) = 6–

Solving Problems 2 - 3+ Fe 3 O x 2 = 6
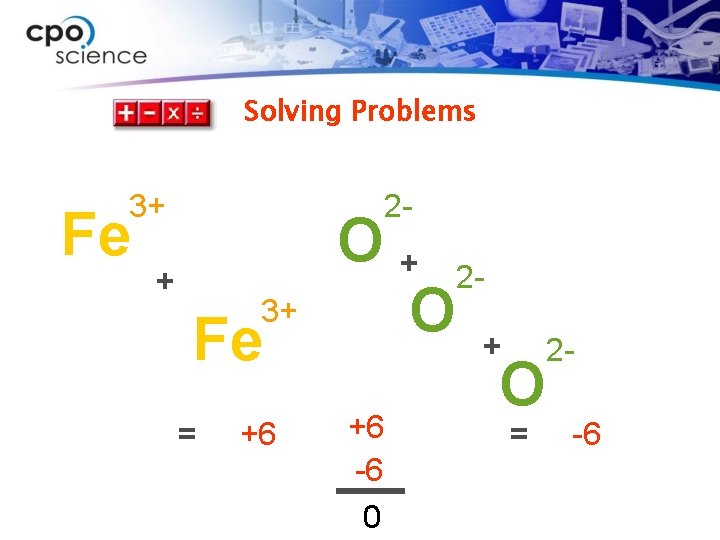
Solving Problems 3+ Fe 2 - + 3+ Fe = +6 O + 2 O +6 -6 0 = -6

Solving Problems 3+ 2 - Fe 2 O 3

16. 2 Polyatomic ions ØCompounds can contain more than two elements. ØSome of these types of compounds contain polyatomic ions. ØA polyatomic ion has more than one type of atom. ØThe prefix poly means “many. ”
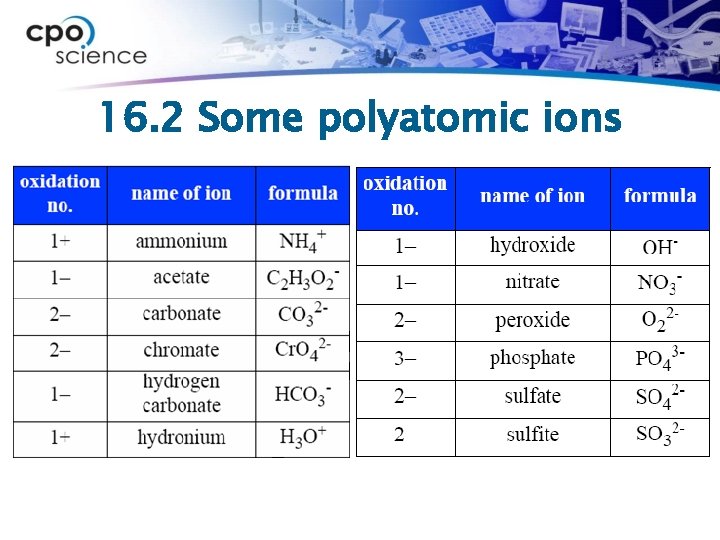
16. 2 Some polyatomic ions

Solving Problems ØAl 3+ combines with sulfate (SO 4)2– to make aluminum sulfate. ØWrite the chemical formula for aluminum sulfate.

Solving Problems 1. Looking for: Ø …formula for a ternary compound 2. Given Ø … Al 3+ and SO 42– 3. Relationships: Ø Write the subscripts so that the sum of the oxidation numbers equals zero. 4. Solution Ø Two aluminum ions = 2 × (3+) = 6+ Ø Three sulfate ions = 3 × (2–) = 6–

Solving Problems 3+ 2 - Al 2 (SO 4) 3

Chapter Sixteen: Compounds Ø 16. 1 Chemical Bonds and Electrons Ø 16. 2 Chemical Formulas Ø 16. 3 Molecules and Carbon Compounds

Chapter 16. 3 Learning Goals ØExplain the significance of carbon in the structure of many different molecules. ØDescribe the importance of carbon to living organisms. ØCompare and contrast the structure and function of carbohydrates, lipids, proteins, and nucleic acids.

Investigation 16 C Carbon and its Chemistry ØKey Question: What are some common molecules that contain carbon?

16. 3 Molecules and Carbon Compounds ØIn addition to the elements from which it is made, the shape of a molecule is also important to its function and properties. ØWe use structural diagrams to show the shape and arrangement of atoms in a molecule.
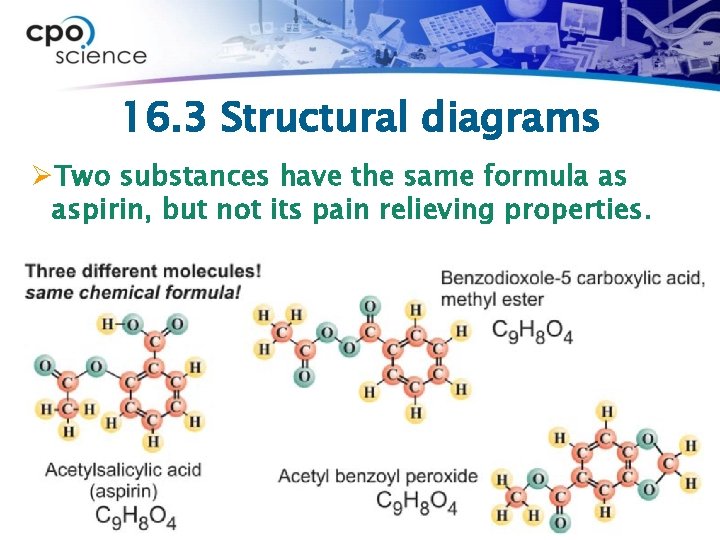
16. 3 Structural diagrams ØTwo substances have the same formula as aspirin, but not its pain relieving properties.

16. 3 The chemistry of carbon Ø Carbon molecules come in three basic forms: straight chains, branching chains, and rings. Ø All three forms are found in important biological molecules.
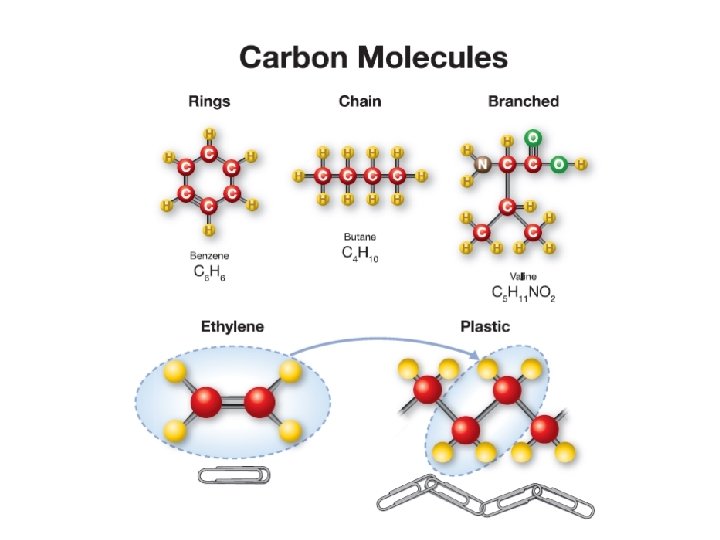

16. 3 Organic compounds ØOrganic chemistry is the branch of chemistry that specializes in carbon compounds, also known as organic molecules. ØPlastic, rubber, and gasoline are important carbon compounds. ØScientists classify the organic molecules in living things into four basic groups: carbohydrates, proteins, fats, and nucleic acids.

16. 3 Carbohydrates Ø Carbohydrates are energy-rich compounds made from carbon, hydrogen, and oxygen. Ø Carbohydrates are classified as either sugars or starches.

16. 3 Carbohydrates Ø Carbohydrates are mainly composed of carbon, hydrogen, and oxygen in a ratio of about 1: 2: 1. Ø Glucose, C 6 H 12 O 6, is a simple sugar. Ø Table sugar is a carbohydrate called sucrose.

16. 3 Carbohydrates Ø Starches are long chains of simple sugars joined together. Ø Cellulose is the primary molecule in plant fibers, including wood.

16. 3 Lipids Ø Like carbohydrates, lipids are energyrich compounds made from carbon, hydrogen, and oxygen whose ratio is much less than 1: 2: 1. Ø Lipids include fats, oils, and waxes.

16. 3 Lipids Ø A typical fat molecule has a two-part structure: Ø glycerol Ø fatty acid chains

16. 3 Saturated or unsaturated fat? ØIn a saturated fat, carbon atoms are surrounded by as many hydrogen atoms as possible. Ø An unsaturated fat has fewer hydrogen atoms than it could have.

16. 3 Proteins Ø Proteins are basic molecular building blocks of cells and all parts of animals. Ø Proteins are among the largest organic molecules. Why is the shape of a protein important?

16. 3 Enzymes ØEnzymes are proteins. ØAn enzyme is a type of protein that cells use to speed up chemical reactions in living things.
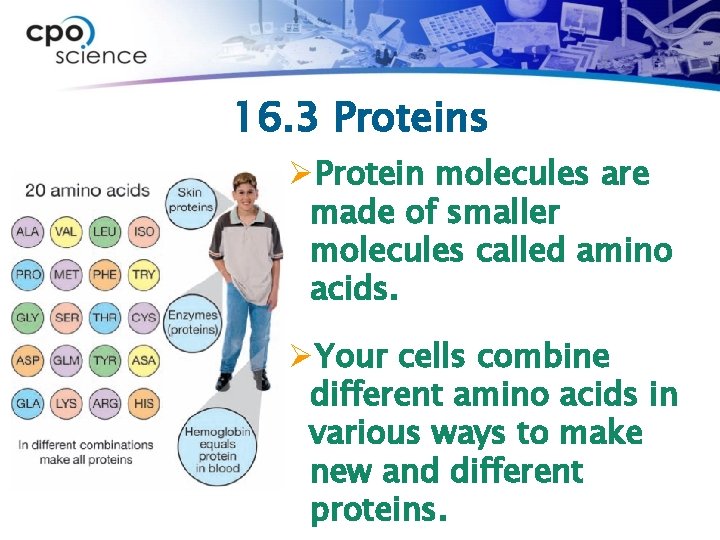
16. 3 Proteins ØProtein molecules are made of smaller molecules called amino acids. ØYour cells combine different amino acids in various ways to make new and different proteins.

16. 3 Nucleic Acids ØNucleic acids are compounds made of long, repeating chains called nucleotides. Each nucleotide contains: 1. 2. 3. a sugar molecule a phosphate molecule, and a base molecule.
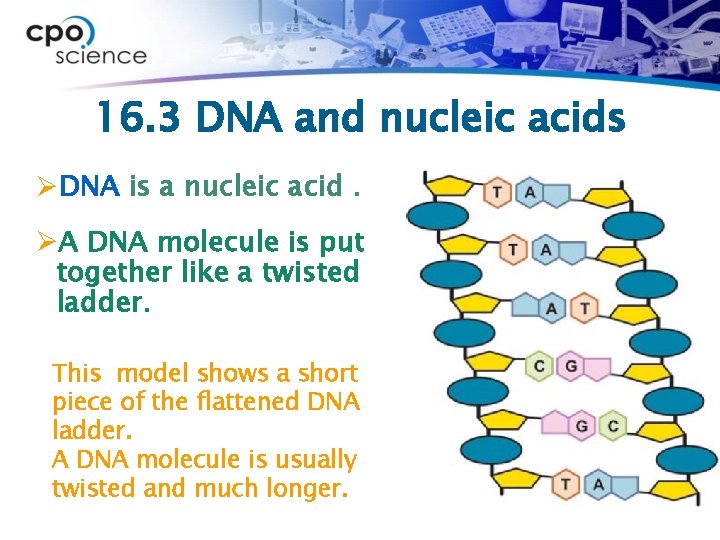
16. 3 DNA and nucleic acids ØDNA is a nucleic acid. ØA DNA molecule is put together like a twisted ladder. This model shows a short piece of the flattened DNA ladder. A DNA molecule is usually twisted and much longer.
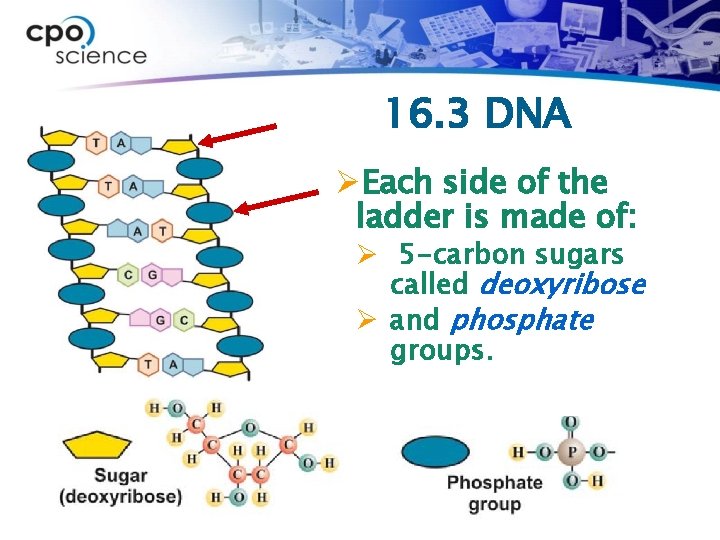
16. 3 DNA ØEach side of the ladder is made of: Ø 5 -carbon sugars called deoxyribose Ø and phosphate groups.
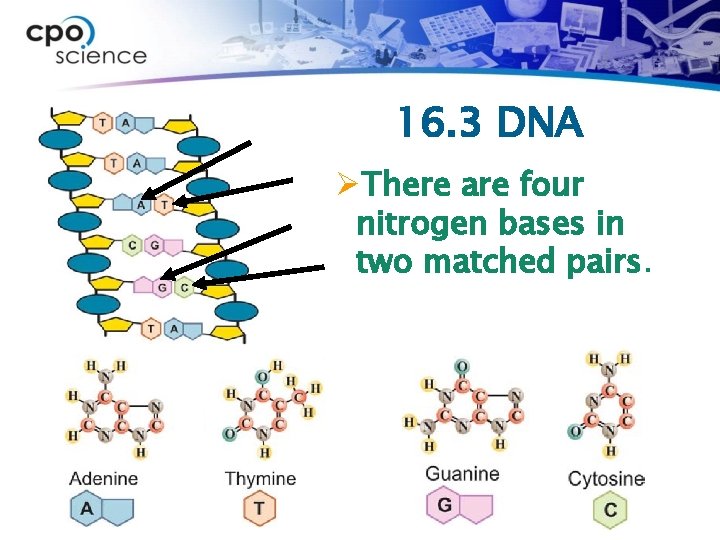
16. 3 DNA ØThere are four nitrogen bases in two matched pairs.


The Spin on Scrap Tires Ø As the number of cars on the road increases each year, so does the number of scrap tires. For many years, the only disposal options were to throw scrap tires into landfills or burn them, which caused air pollution. Today, scientists and engineers are coming up with innovative ways to put a new spin on discarding old tires.
- Slides: 67