Compounds Bonding Chapters 6 7 Valence Electrons Electrons

Compounds & Bonding Chapters 6 & 7

Valence Electrons • Electrons in the outer energy level are called valence electrons • It is these electrons that determine the formation of chemical bonds

Valence Electrons • Certain numbers of valance electrons are more stable than others. • Atoms will gain or lose electrons to become more stable.
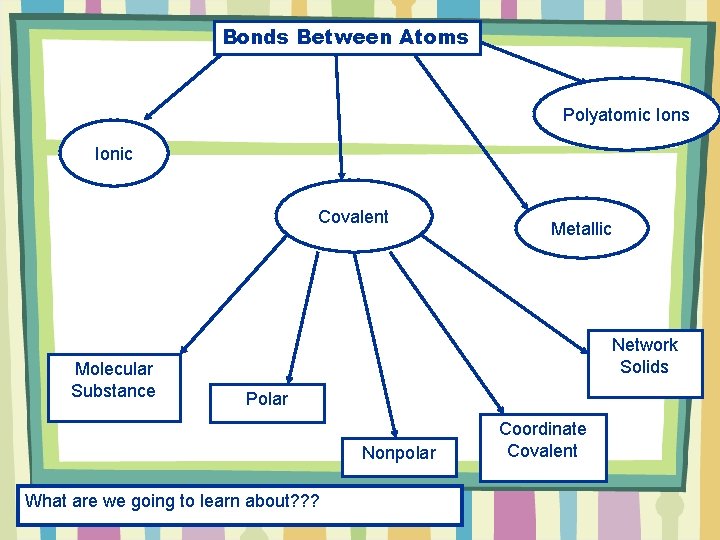
Bonds Between Atoms Polyatomic Ions Ionic Covalent Molecular Substance Metallic Network Solids Polar Nonpolar What are we going to learn about? ? ? Coordinate Covalent
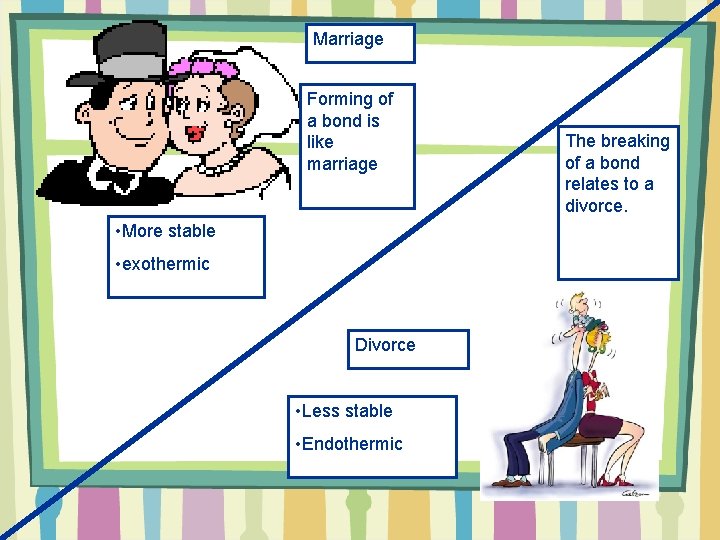
Marriage Forming of a bond is like marriage • More stable • exothermic Divorce • Less stable • Endothermic The breaking of a bond relates to a divorce.

Chemical Bond Types • Ionic • Covalent • Metallic
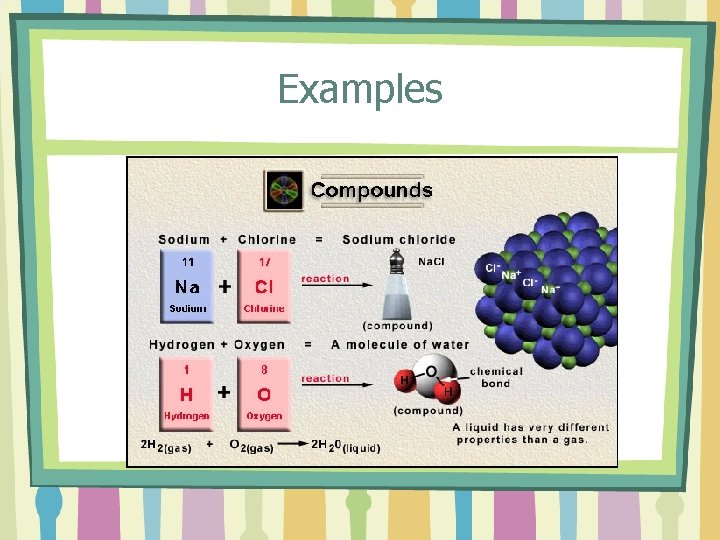
Examples

Types of Bonds • There are two types of bonds – ionic & covalent • Ionic bonds – a bond formed from a force between two opposite charges (+ and -). • Covalent bonds – a bond formed from sharing electrons between two atoms. – Single bonds = one bond between only two atoms – Multiple bonds = more than one bond between atoms

What are some characteristics of an ionic bond? 1. Crystalline (salt crystals) at room temperatures 2. Have higher melting points and boiling points compared to covalent compounds 3. Conduct electrical current in molten or solution state but not in the solid state 4. Polar bonds

What are some characteristics of a covalent bond? 1. Covalent bonds have definite and predictable shapes. 2. Very strong 3. Low melting and boiling points

Types of Bonds • Ionic bonds are made from metal and a nonmetal. • Covalent bonds are made from two nonmetals. – Polar molecules have a (+) and a (–) end to them (like ionic but weaker). – Nonpolar molecules have their electrons shared evenly so that there are no ends like a magnet.
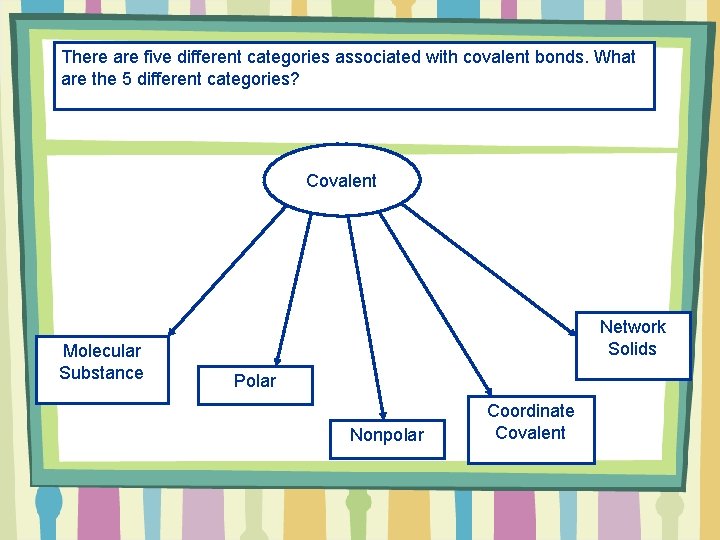
There are five different categories associated with covalent bonds. What are the 5 different categories? Covalent Molecular Substance Network Solids Polar Nonpolar Coordinate Covalent
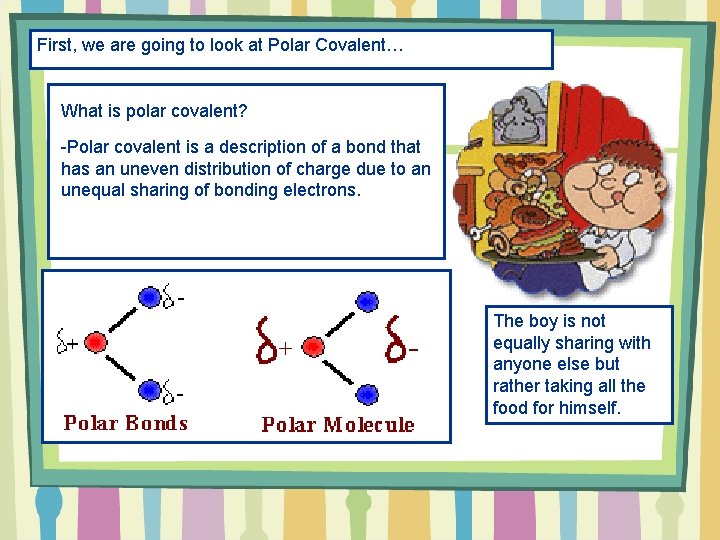
First, we are going to look at Polar Covalent… What is polar covalent? -Polar covalent is a description of a bond that has an uneven distribution c of charge due to an unequal sharing of bonding electrons. The boy is not equally sharing with anyone else but rather taking all the food for himself.

Next, we are going to look at Non-Polar Covalent… What is non-polar covalent? -Non polar covalent is a covalent bond that has an even distribution of charge due to an equal sharing of bonding electrons. This couple is non- polar because they are sharing the drink equally between them.
Next, we are going to look at Molecular Substances… What is a molecular substance? -A molecular substance is a substance that has atoms held together by covalent bonds. Name 2 Characteristics of a Molecular Substance. 1. Weak 2. Low melting and boiling points

Next, we are going to look at Coordinate Covalent… What is a Coordinate Covalent Bond? -A coordinate covalent bond is a bond formed when one atom donates both electrons that are shared. People donate their blood to help others just like atoms “donate” electrons to form stable octets.
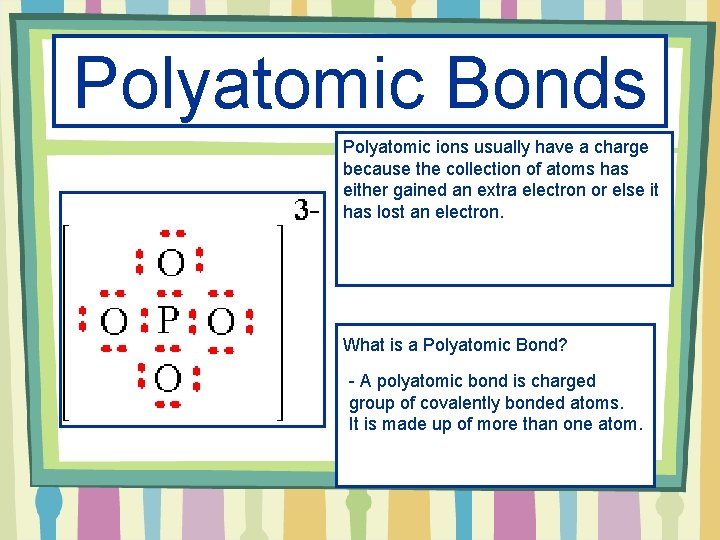
Polyatomic Bonds Polyatomic ions usually have a charge because the collection of atoms has either gained an extra electron or else it has lost an electron. What is a Polyatomic Bond? - A polyatomic bond is charged group of covalently bonded atoms. It is made up of more than one atom.
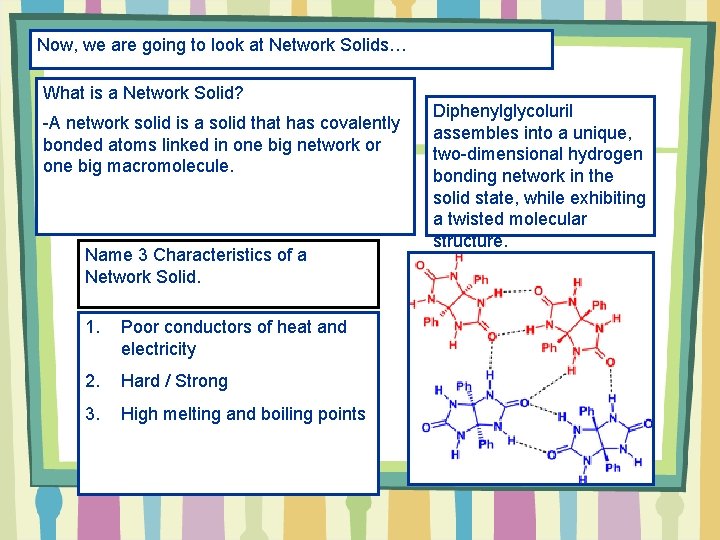
Now, we are going to look at Network Solids… What is a Network Solid? -A network solid is a solid that has covalently bonded atoms linked in one big network or one big macromolecule. Name 3 Characteristics of a Network Solid. 1. Poor conductors of heat and electricity 2. Hard / Strong 3. High melting and boiling points Diphenylglycoluril assembles into a unique, two-dimensional hydrogen bonding network in the solid state, while exhibiting a twisted molecular structure.
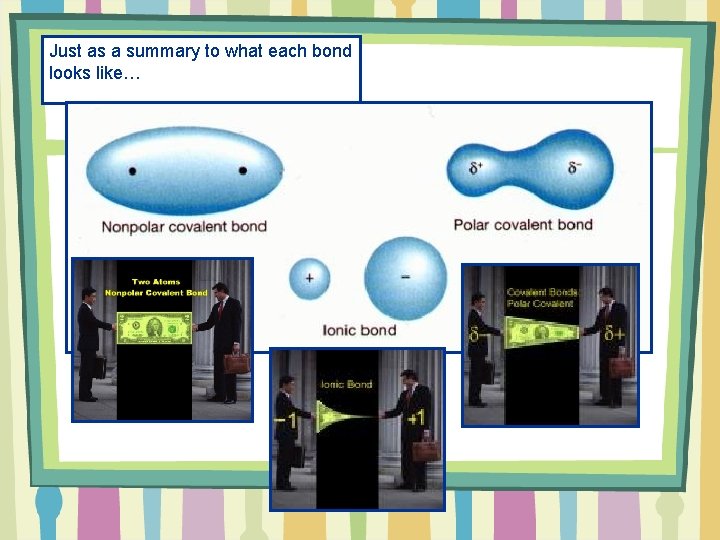
Just as a summary to what each bond looks like…

Predicting Bond Types • Will these pairs form ionic or covalent bonds? sodium & – calcium & – carbon & – aluminum – chlorine oxygen & sulfur

Writing Chemical Formulas • Binary Ionic Compounds – “bi” means two, compounds made of two elements – Example: potassium + iodine potassium iodide K+ + I- KI Oxidation number means charges on the “ions” (which means charges). Let’s practice!! beryllium + selenium beryllium selenide

Writing Chemical Formulas • Polyatomic Ions – “poly” means many, compounds made with two or more elements • Ex: ammonium + chlorine ammonium chloride NH 4+ + Cl NH 4 Cl • (DO NOT BREAK APART POLYATOMIC ELEMENTS – THEY TRAVEL TOGETHER!!) • Let’s practice!! – carbonate + magnesium carbonate
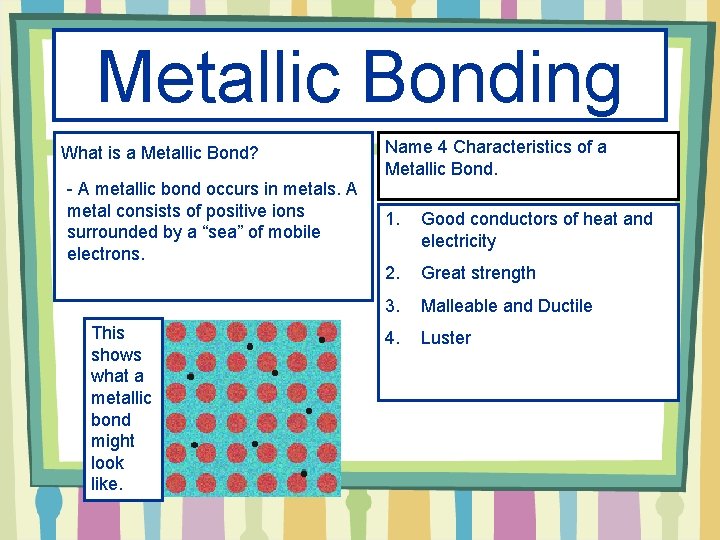
Metallic Bonding What is a Metallic Bond? - A metallic bond occurs in metals. A metal consists of positive ions surrounded by a “sea” of mobile electrons. This shows what a metallic bond might look like. Name 4 Characteristics of a Metallic Bond. 1. Good conductors of heat and electricity 2. Great strength 3. Malleable and Ductile 4. Luster

Naming Ionic Compounds • Use the whole first name (+ element) and the root of the second name (- element). Change the ending of the second element to -ide. • If there is a polyatomic, the name DOES NOT change!!! • Let’s practice!! Ca. Cl 2 Al. N BPO 4

Covalent Nomenclature Rules • Prefix System (binary compounds) 1. Less e-neg atom comes first. 2. Add prefixes to indicate # of atoms. Omit monoprefix on first element. 3. Change the ending of the second element to -ide.

Naming Covalent Molecules • Use prefixes to tell how many atoms of each element in the compound • Ex: CO 2, N 2 O 4, CCl 4 • Write the formula of the following from the name: sulfur dichloride, phosphorus hexachloride, dihydrogen monoxide # of atoms Prefixes 1 mono 2 di 3 tri 4 tetra 5 penta 6 hexa 7 hepta 8 octa 9 nona 10 deca

Naming Covalent Molecules ØNCl 4 nitrogen tetrachloride Ø N 2 O dinitrogen monoxide ØSF 6 sulfur hexafluoride

Writing Molecular Structures Øarsenic trichloride As. Cl 3 Ødinitrogen pentoxide N 2 O 5 Øtetraphosphorus decoxide P 4 O 10
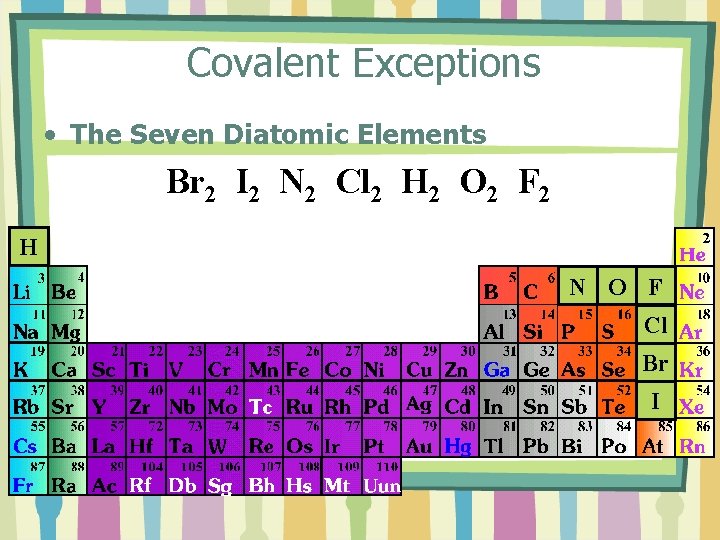
Covalent Exceptions • The Seven Diatomic Elements Br 2 I 2 N 2 Cl 2 H 2 O 2 F 2 H N O F Cl Br I
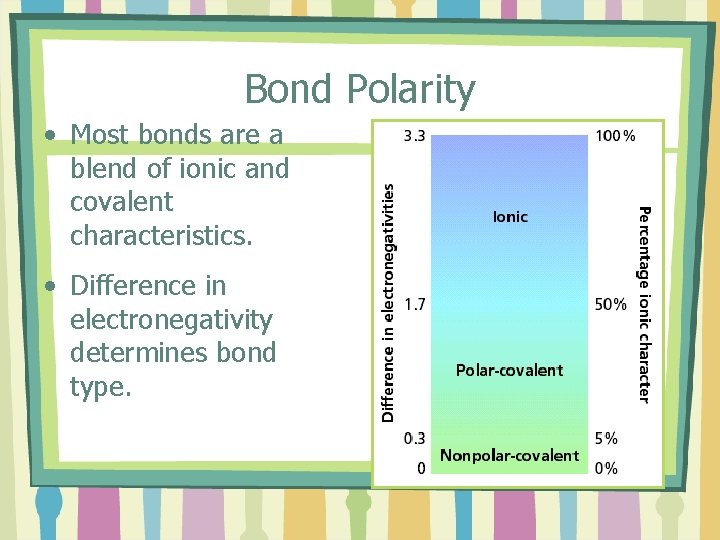
Bond Polarity • Most bonds are a blend of ionic and covalent characteristics. • Difference in electronegativity determines bond type.
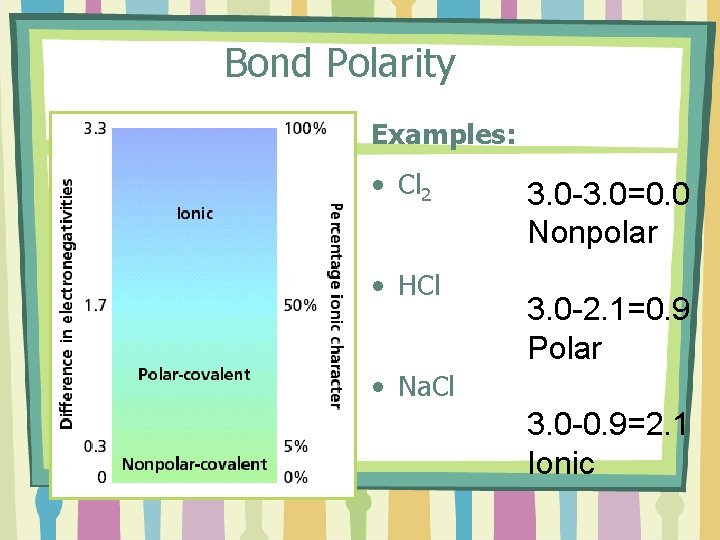
Bond Polarity Examples: • Cl 2 • HCl 3. 0 -3. 0=0. 0 Nonpolar 3. 0 -2. 1=0. 9 Polar • Na. Cl 3. 0 -0. 9=2. 1 Ionic

REMEMBER…. . • Electronegativity Trend (p. 161) – Increases up and to the right. – There are values to each participating element

Chemical Changes • Chemical reaction – a change in which one or more substances are converted into new substances Chemical equation reactants products Review symbols • Conservation of mass – starting mass of the reactants must equal the final mass of the products -Ex: Hg. O Hg + O 10. 0 g = 9. 3 g + ? g

Balancing Chemical Equations • Conservation of mass states that the reactants and the products must equal. • Balancing charges on ions sometimes makes the products equal the reactants. • Coefficients (big numbers) are used to multiply the compounds and balance the mass on each side of the chemical equation.

Balancing Chemical Equations 1. __ Al + __O 2 __Al 2 O 3 2. __ H 2 + __ O 2 __H 2 O 3. __ S + __ O 2 __SO 3

Classifying Chemical Reactions • • Synthesis Decomposition Single displacement Double displacement A + B AB AB A + BC A C + B A B + C D A D + BC

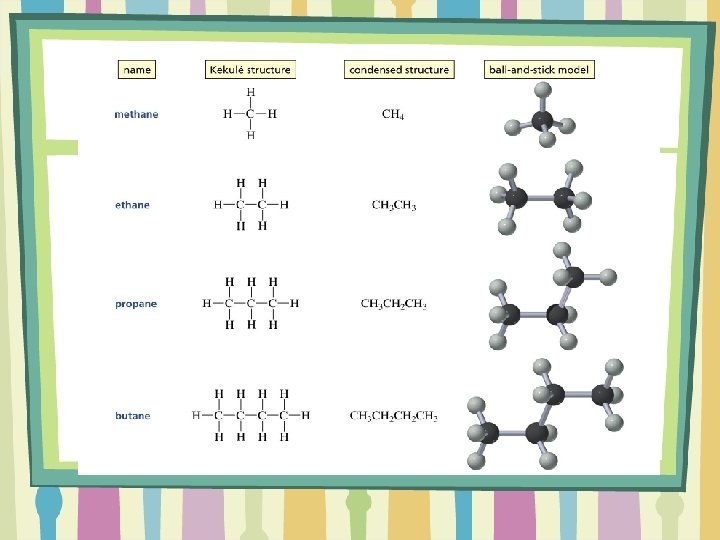
Organic Compounds • Organic compounds are # of made of mostly carbon and hydrogen atoms • Naming these 1 compounds starts with the # of carbon atoms 2 present using a 3 particular prefix. 4 • Then the ending depends on the type of 5 bonds made. Prefix for name Type Suffix of Bond single -ane methethdouble -ene proptriple -yne butpent-
- Slides: 38