Compound Names and Formulas Section 4 3 Naming











- Slides: 16

Compound Names and Formulas Section 4. 3

Naming Compounds • Each compound has a unique name • Relatively easy rules for naming ionic and covalent compounds

Naming Ionic Compounds • Name of cation stays the same – Lithium atom lithium (ion) – Magnesium atom magnesium (ion) • Name of anion turns into an “ide” – Chlorine atom chloride (ion) – Iodine atom iodide (ion) • Name = cation + anion – Lithium + Chlorine Lithium Chloride

More examples of ionic compounds • • Barium Chloride Magnesium Bromide Sodium Sulfide Cesium Nitride Rubidium Oxide Lithium Fluoride Potassium Chloride

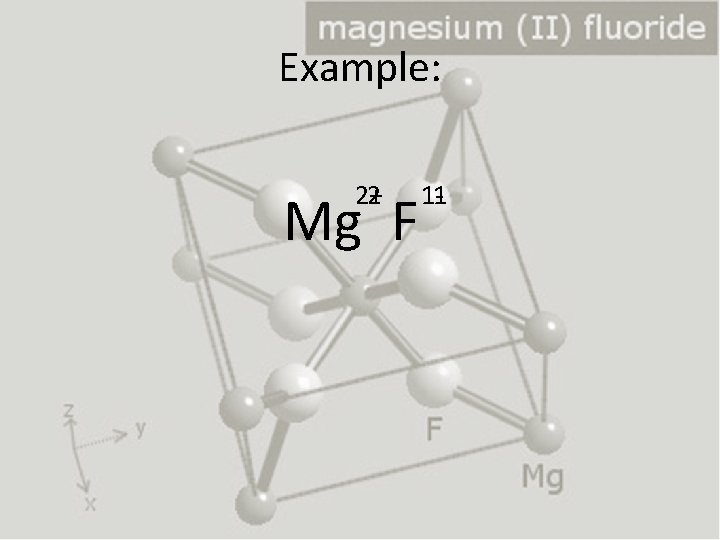
Predicting the number of cations & anions in an ionic compound • Example: Magnesium Fluoride – Magnesium cation has a +2 charge – Fluoride has a -1 charge • Mg 2+F-1 • Rule: Erase charges, replace superscripts with subscripts and switch the numbers between the ions

Example: 2+ 2 Mg F 1 -1

Why? • Magnesium cation: +2 charge • Fluoride anion: -1 charge • It takes TWO fluoride ions to cancel out the charge of the magnesium cation. -1 -1 2+

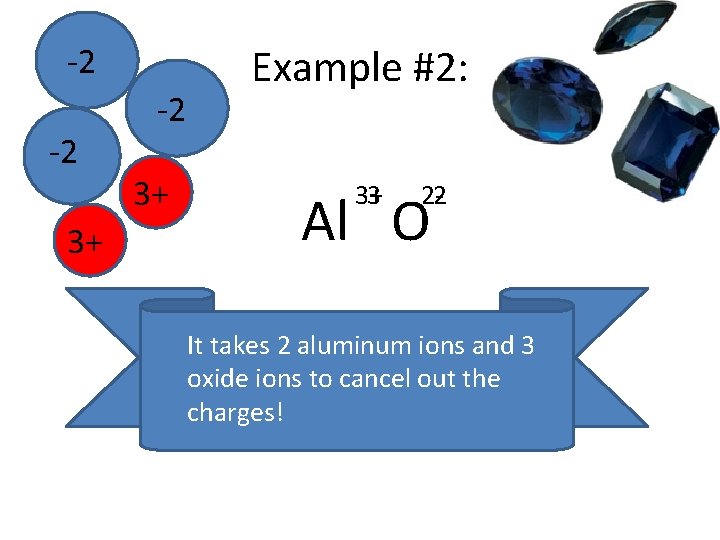
-2 -2 3+ Example #2: 3+ 3 2 -2 Al O It takes 2 aluminum ions and 3 oxide ions to cancel out the charges!

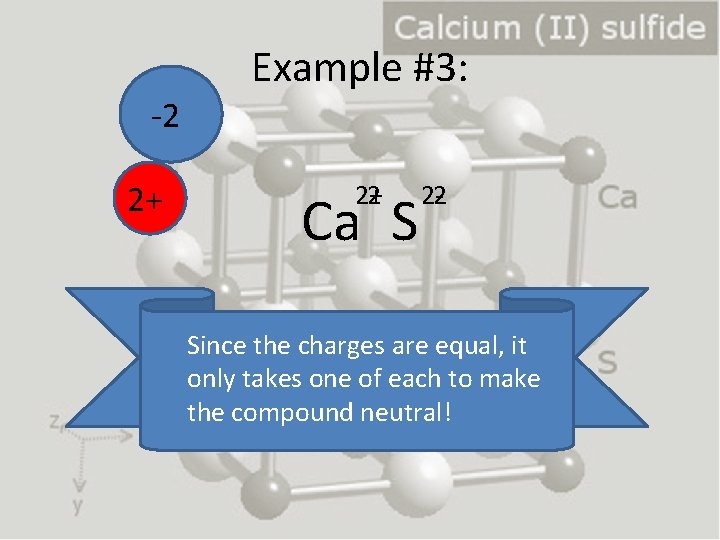
-2 2+ Example #3: 2+ 2 Ca S 2 -2 Since the charges are equal, it only takes one of each to make the compound neutral!

Ionic bonds with transition metals • Many transition metals can take on more than one charge as an ion: – Cu+, Cu 2+ – Fe 2+, Fe 3+ • • If Cu+ bonds to F-, you get Cu. F If Cu 2+ bonds to F-, you get Cu. F 2 Both cannot be called “Copper Fluoride” What can we do?

The answer! • Designate the charge of the cation in the name by using Roman numerals! • Cu. F contains a copper cation of charge +1 – We call this Copper (I) Fluoride • Cu. F 2 contains a copper cation of charge +2 – We call this Copper (II) Fluoride • Rule: When working with transition metals, put the charge of the cation in parentheses using Roman numerals!


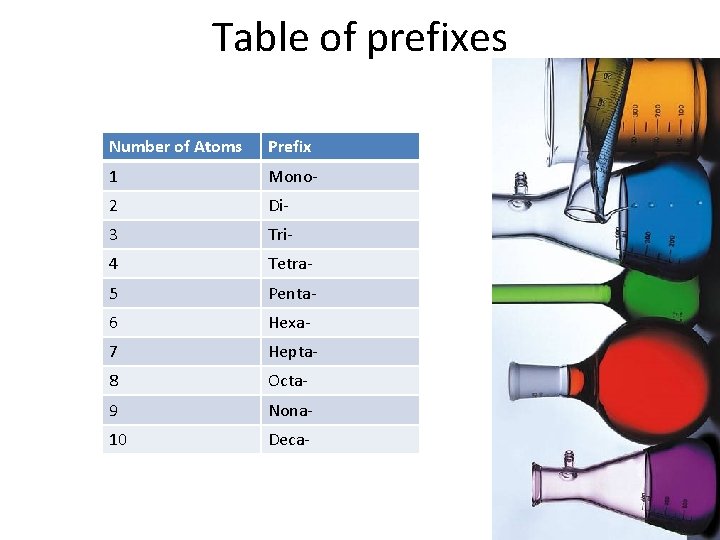
Naming Covalent Compounds • Let’s look at one we already know: – Carbon Dioxide (CO 2) • The first element is named regularly (carbon) • The second element gets an “-ide” • The number of each element is represented by a prefix. • Rule: If there is only one atom of the first element, it does not get a prefix. • Otherwise, the first and second elements get prefixes according to the following table:

Table of prefixes Number of Atoms Prefix 1 Mono- 2 Di- 3 Tri- 4 Tetra- 5 Penta- 6 Hexa- 7 Hepta- 8 Octa- 9 Nona- 10 Deca-

Examples Number of Atoms Prefix 1 Mono- 2 Di- 3 Tri- 4 Tetra- 5 Penta- 6 Hexa- 7 Hepta- 8 Octa- 9 Nona- 10 Deca- • H 2 O • N 2 O 4 • P 4 S 3 • Si. I 4 • PCl 3 • P 4 O 10

Remember the Name • Chemistry Remix • Follow along with the lyrics on the back of your HRL