Composition of Matter Substance type of matter with

Composition of Matter

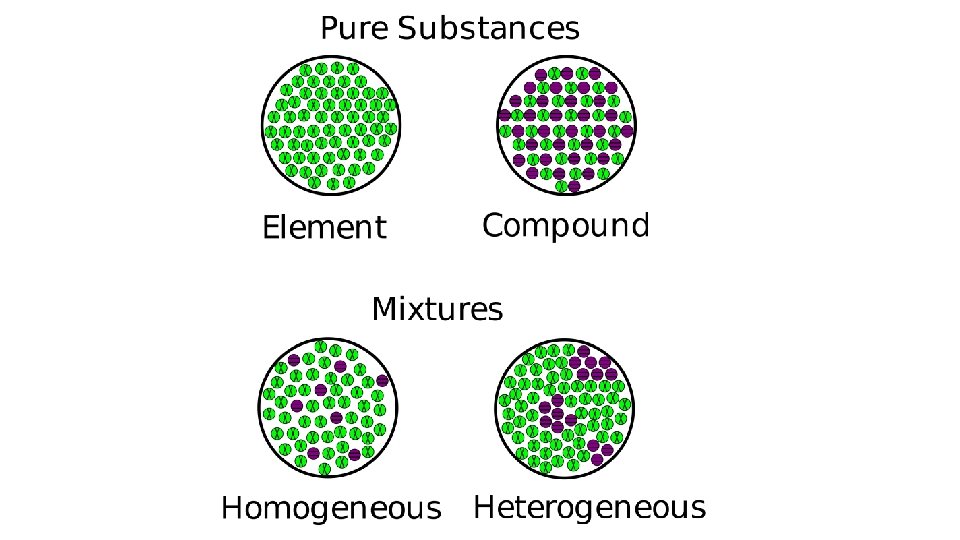
Substance – type of matter with a fixed composition that cannot be separated by physical means Element – substance made up of atoms with the same identity Ex: Gold Helium Aluminum Carbon

Compound – atoms of two or more elements chemically combined in a fixed portion Ex: water (H 2 O) carbon dioxide (CO 2) oxygen (O 2) salt
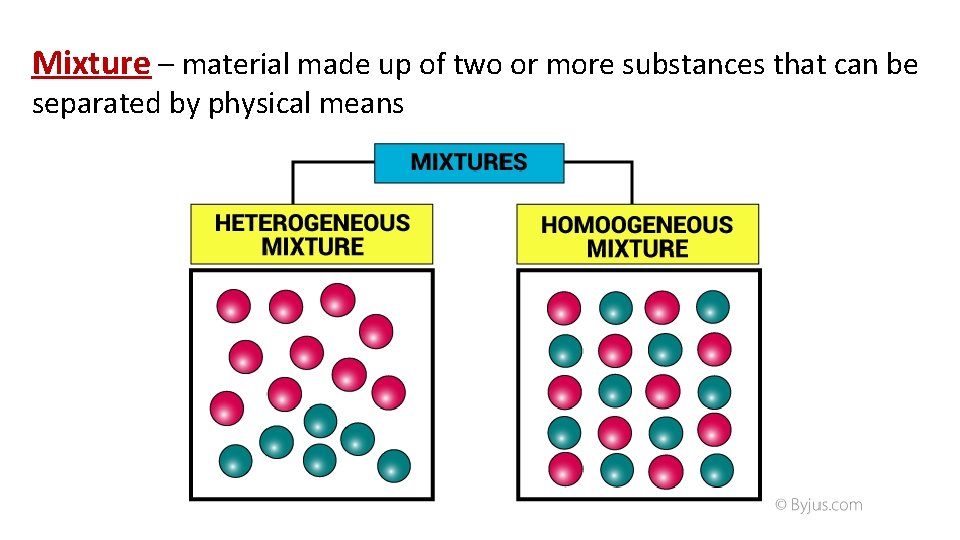
Mixture – material made up of two or more substances that can be separated by physical means
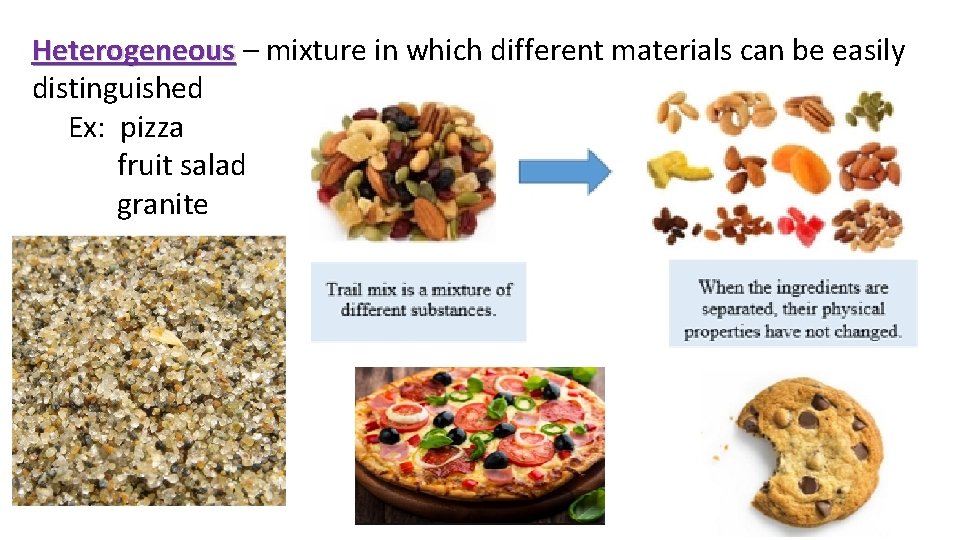
Heterogeneous – mixture in which different materials can be easily Heterogeneous distinguished Ex: pizza fruit salad granite

Homogeneous – mixture in which two or more substances are uniformly spread out Ex: vinegar, salt water
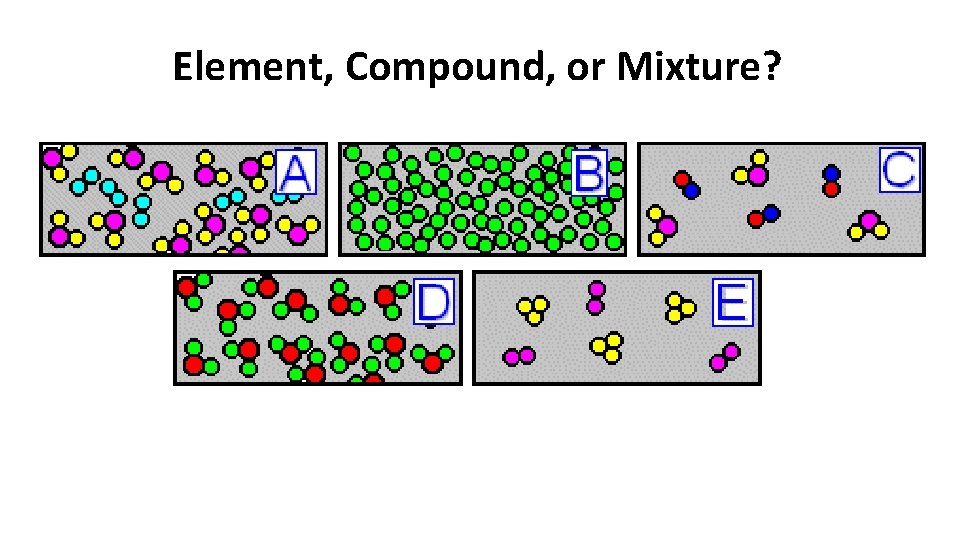
Element, Compound, or Mixture?

Types of Mixtures

Solution – homogeneous mixture of particles so small that they cannot even be seen with a microscope and will never settle to the bottom of their container Ex: vinegar soda (coke) hydrogen peroxide air

2 parts of a solution 1. Solvent – dissolving substance Ex: water 2. Solute – substance being dissolved Ex: Kool-Aid powder

Colloid – type of mixture with particles that are larger than those in solution, but still too light to settle out Ex: milk (water and fat), fog (water and air), cool whip, cytoplasm
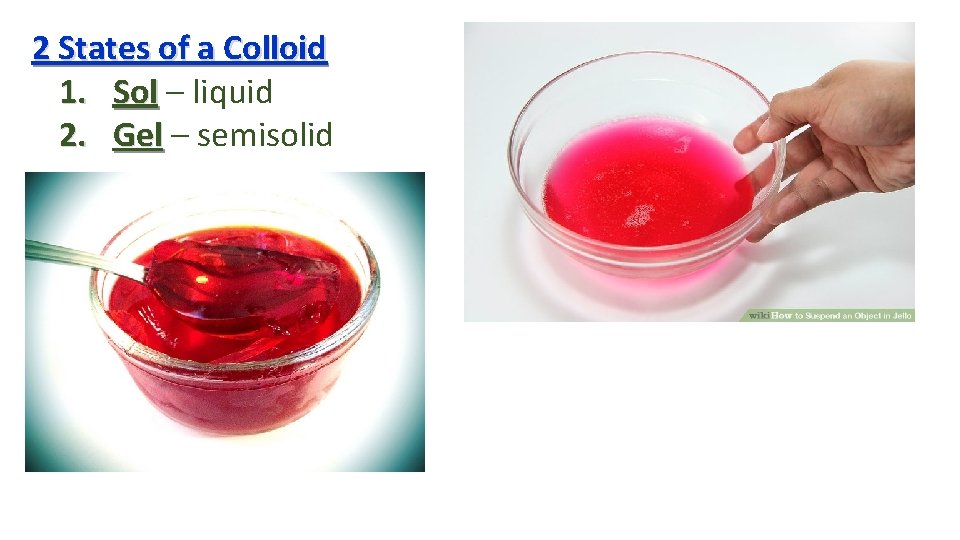
2 States of a Colloid 1. Sol – liquid Sol 2. Gel – semisolid Gel

Determining colloids is sometimes difficult so shining a bean of light at colloid will make the light scatter – this scattering of light by a colloid is called the Tyndall effect

Suspension – heterogenous mixture containing a liquid in which visible particles settle Ex: Italian dressing, muddy pond water, chocolate milk, blood *Hint – if it needs shaking to mix, then it’s a suspension

Review of Composition of Matter 1. Distinguish between a substance from a mixture. Give two examples of each. 2. Compare and Contrast How is a compound similar to a homogeneous mixture? How is it different? 3. Identify three elements and three compounds. How are they similar? How are they different? 4. Make a table that compares the properties of suspensions, colloids, and solutions. 5. Why do the words “Shake well before using” indicate that the fruit juice in a carton is a suspension? Why are these words not used on a milk container?
- Slides: 16