Composition Book Notebook setup Table of contents Page

Composition Book Notebook set-up

Table of contents Page 1 Stoichiometry

1

Stoichiometry. Standard 3 Ms. Siddall
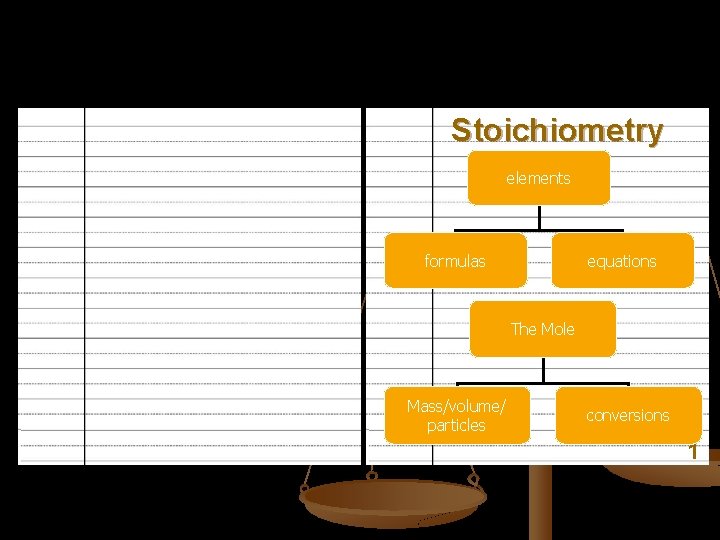
Stoichiometry elements formulas equations The Mole Mass/volume/ particles conversions 1

The periodic table n n n The periodic table displays all elements Symbols on the left represent metals Symbols on the right represent non-metals Non-metals Separation line

The periodic Table n n n 2 The periodic table displays all elements Symbols on the left represent metals Symbols on the right represent non-metals 3

Summary 1 n Identify the following elements as metals or non-metals: 1. 2. 3. 4. 5. 6. Copper Chlorine Carbon Sodium Calcium Hydrogen

The periodic Table Summary 1: Identify the following elements as metals or non-metals n n n 2 The periodic table displays all elements Symbols on the left represent metals Symbols on the right represent non-metals 3

Symbols n Each chemical symbol on the periodic table represents an element chlorine = Cl n sodium = Na n n Symbols written together represent a compound n Na. Cl = a sodium atom combined with a chlorine atom

The periodic Table Summary 1: Identify the following elements as metals or non-metals Summary 2: n n n The periodic table displays all elements Symbols on the left represent metals Symbols on the right represent nonmetals symbols Each chemical symbol on the periodic table represents an element chlorine = Cl 2 3

Notes… Summaries… 4 5

Summary 2 n Define the following words 1. 2. 3. 4. n Element: Atom: Compound: Chemical formula: Describe the similarities and differences between an atom and an element

Compounds n Covalent compound: non-metal atoms combine to form molecules n n Ionic compound: metal and non-metal atoms combine to formula units n n e. x. H 2 O and CO 2 e. x. Na. Cl, Cu. SO 4 chemical formula: a combination of symbols and numbers that describe the amount and type and of atoms that form a compound.

Summary 3 n Identify the following compounds as ionic or covalent 1. 2. 3. 4. Cu. SO 4 Na. Cl CO 2 H 2 O

Anatomy of a chemical formula Cu. SO 4(aq) Atomic symbols describe the type of atoms in the compound (copper, sulfur, oxygen) subscript numbers describe the number of atoms in the compound (1 copper, 1 sulfur, 4 oxygen) subscript letters describe the physical state of the compound (aqueous)

Summary 4 Na 2 O(s) In the above formula: 1. How many sodium atoms? 2. How many oxygen atoms? 3. What is the physical state? 4. Is the compound ionic or covalent?
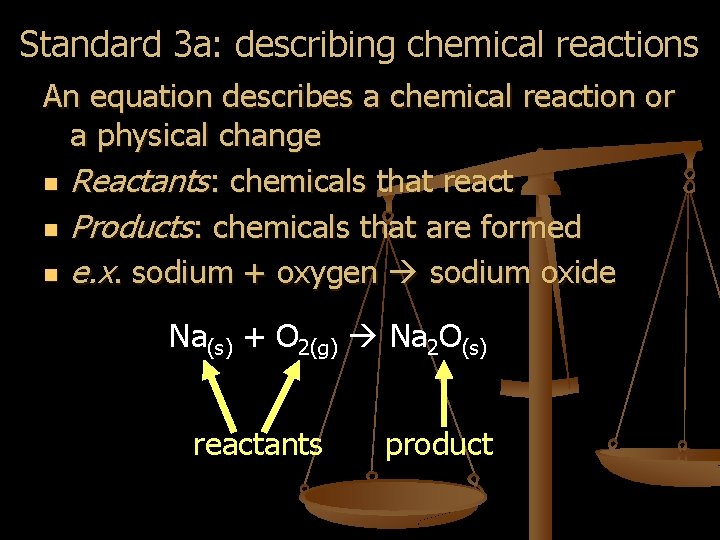
Standard 3 a: describing chemical reactions An equation describes a chemical reaction or a physical change n Reactants: chemicals that react n Products: chemicals that are formed n e. x. sodium + oxygen sodium oxide Na(s) + O 2(g) Na 2 O(s) reactants product

Summary 5 Pb(NO 3)3(aq) + KI(aq) KNO 3(aq) + Pb. I 3(s) n n n Label the reactants and the products in the reaction above Are the reactants and products covalent or ionic? How many oxygen atoms are in the compound Pb(NO 3)3?

Symbols describing chemical reactions Copy table 11. 1 (page 323)

Summary 6 1. What do the following symbols mean? n n n 2. (l) (aq) Pb(NO 3)3(aq) + KI(aq) KNO 3(aq) + Pb. I 3(s) n Which compound is solid?

Balancing chemical equations The Law of Conservation of Matter: Matter cannot be created or destroyed. n For chemical equations: The total number of each type of atom must be the same before and after the reaction Thou shall not create or destroy matter

example n Sodium reacts with oxygen to produce sodium oxide Na(s) + O 2(g) Na 2 O(s) n n n How many sodium atoms react? 1 How many sodium atoms are in the product? 2 This violates the law of conservation of matter!

Summary 7 Na. I(s) + Cl 2(g) Na. Cl(s) + I 2(s) Count the number of atoms on the reactants side Sodium ____ Iodine ____ Chlorine ____ Count the number of atoms on the products side Sodium ____ Iodine ____ Chlorine ____ Does this equation obey the law of conservation of matter?

Example: 4 Na(s) + O 2(g) 2 Na 2 O(s) SUBSCRIPTS CAN NOT CHANGE! n Coefficients are used to balance the equation n number of atoms or formulas needed in the reaction. These apply to the entire formula (all the atoms) 4 Na = 4 sodium atoms n 2 Na 2 O = 4 sodium atoms and 2 oxygen atoms n

Summary 8 2 Cu(s) + O 2(g) 2 Cu. O(s) Count the number of atoms on the reactants side copper ____ Oxygen ____ Count the number of atoms on the products side copper ____ Oxygen ____ Does this equation obey the law of conservation of matter?

Rules of Balancing Equations 1. 2. Write the equation using correct formulas. You may NOT change the formula in any way. Balance the equation using coefficients
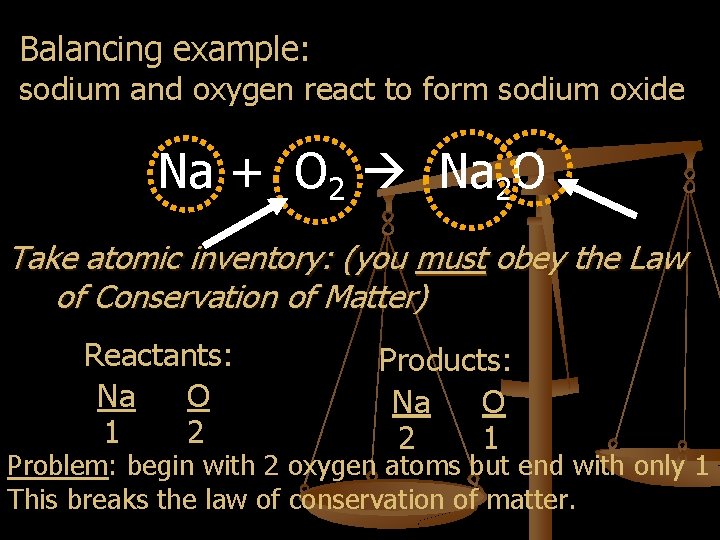
Balancing example: sodium and oxygen react to form sodium oxide Na + O 2 Na 2 O Take atomic inventory: (you must obey the Law of Conservation of Matter) Reactants: Na O 1 2 Products: Na O 2 1 Problem: begin with 2 oxygen atoms but end with only 1 This breaks the law of conservation of matter.

Na + O 2 2 Na 2 O (= Na 2 O + Na 2 O) solution: Add the coefficient ‘ 2’ in front of Na 2 O Take atomic inventory again: Reactants: Na O 1 2 Products: Na O 4 2 Problem: begin with 1 sodium atom but end with 4. This breaks the law of conservation of matter.

4 Na + O 2 2 Na 2 O solution: Add the coefficient ‘ 4’ in front of Na Take atomic inventory again: Reactants: Na O 4 2 Products: Na O 4 2 4 sodium atoms combine with 1 oxygen molecule to form 2 formula units of sodium oxide. This equation obeys the Law of Conservation of Matter.

Summary 9 n BALANCE THE FOLLOWING REACTION: H 2(g) + O 2(g) H 2 O(l)

Weird things: polyatomic ions n Ionic compounds can be formed with polyatomic ions (made from many atoms) n CO 32= one carbon and three oxygen atoms n SO 42 n OHn NH 4+ n Sometimes you can treat a polyatomic ion like one atom sometimes you must balance individual atoms

Summary 10 n Identify the polyatomic ions in the following compounds: 1. 2. 3. 4. 5. 6. CO 2 KNO 3 Na. OH Cu. Cl 2 Li 3 PO 4 NH 4 OH

Balancing with polyatomic ions n Sometimes polyatomic ions break apart in a chemical reaction and sometimes they do not n e. x. sulfate appears on both sides of the reaction so SO 4 can be treated like one atom: Mg(s) + Cu. SO 4(aq) Mg. SO 4(aq) + Cu(s) n e. x. carbonate breaks apart so atoms must be balanced individually: Ca. CO 3(aq) + HCl(aq) Ca. Cl 2(aq) + H 2 O(l) + CO 2(g)

Summary 11 n Balance the following equations: 1. Na 2 CO 3(s) + HCl(aq) Na. Cl(aq) + CO 2(g) + H 2 O(l) 2. K 2 SO 4(aq) + Ca. Cl 2(aq) Ca. SO 4(s) + KCl(l)

Types of Chemical Reactions. 1. Combination. n n Also called synthesis Two or more reactants combine to form one product n e. x. 2 Na(s) + Cl 2(g) 2 Na. Cl(s) n A + B AB http: //chemed. chem. purdue. edu/demos/main_pages/7. 1. html

Summary 12 Which equation represents a synthesis reaction? n 2 Ca(s) + O 2(g) 2 Ca. O(s) n 2 KCl. O 3(s) 2 KCl(s) + O 2(g)

2. Decomposition. n One reactant decomposes to form two or more products. n 2 H 2 O(l) 2 H 2(g) + O 2(g) n AB A + B

Summary 13 Which equation represents a decomposition reaction? n Ca(s) + O 2(g) Ca. O(s) n 2 KCl. O 3(s) 2 KCl(s) + O 2(g)
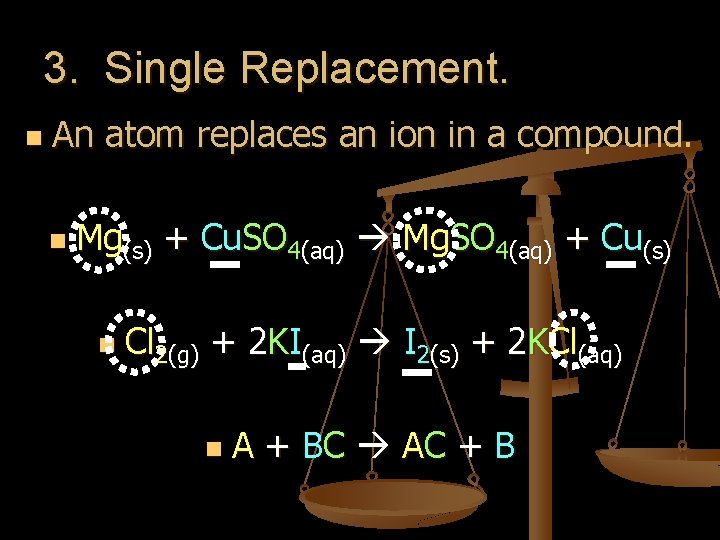
3. Single Replacement. n An atom replaces an ion in a compound. n Mg(s) + Cu. SO 4(aq) Mg. SO 4(aq) + Cu(s) n Cl 2(g) + 2 KI(aq) I 2(s) + 2 KCl(aq) n A + BC A C + B

Summary 14 Which equation represents a single replacement reaction? • 2 Na. I(s) + Cl 2(g) 2 Na. Cl(s) + I 2(s) • 2 Na. I(aq) + Pb(NO 3)2(aq) 2 Na. NO 3(aq) + Pb. I 2(s)

4. Double Replacement. n n Ions from different compounds switch places. Ca. CO 3(s) + 2 HCl(aq) Ca. Cl 2(s) + H 2 CO 3(aq) n AB + CD AD + CB

Summary 15 Which equation represents a double replacement reaction? • 2 Na. I(s) + Cl 2(g) 2 Na. Cl(s) + I 2(s) • 2 Na. I(aq) + Pb(NO 3)2(aq) 2 Na. NO 3(aq) + Pb. I 2(s)

5. Combustion reactions. n n n A compound reacts with oxygen often produces CO 2 & H 2 O e. x. C 3 H 8(g) + 5 O 2(g) 3 CO 2(g) + 4 H 2 O(g)

Summary 16 n Write the balanced equation for the reaction of CO with O 2 to form CO 2 and identify the type of reaction.

Standard 3 e: The Arithmetic of Equations. n A balanced equation shows the amount of each reactant and product needed or produced in any reaction.

Example. n n Mg(s) + 2 HCl(aq) Mg. Cl 2(aq) + H 2(g) One atom of magnesium combines with 2 formula units of HCl to form one formula unit of magnesium chloride and one molecule of hydrogen gas. n Use equation coefficients to solve quantitative problems.

Summary 17 4 Na(s) + O 2(g) 2 Na 2 O(s) 1. 2. 3. How many molecules of oxygen are needed to react with 4 atoms of sodium? How many oxygen atoms is that? How many formula units of sodium oxide are produced when 4 atoms of sodium are used?
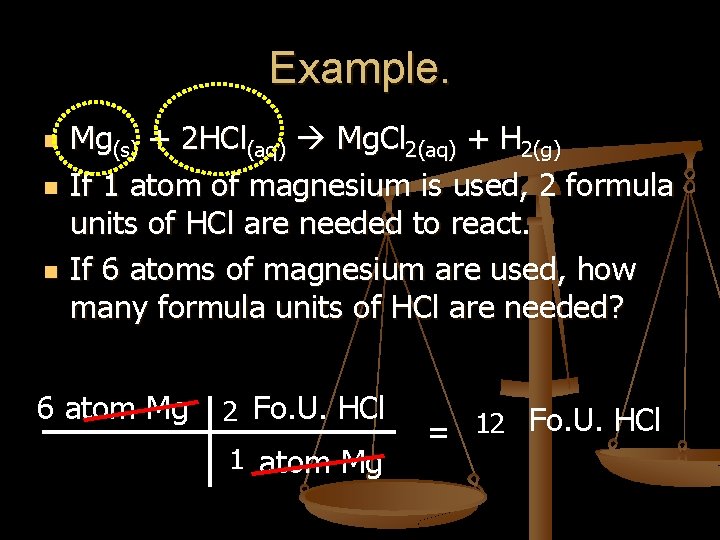
Example. n n n Mg(s) + 2 HCl(aq) Mg. Cl 2(aq) + H 2(g) If 1 atom of magnesium is used, 2 formula units of HCl are needed to react. If 6 atoms of magnesium are used, how many formula units of HCl are needed? 6 atom Mg 2 Fo. U. HCl 1 atom Mg = 12 Fo. U. HCl
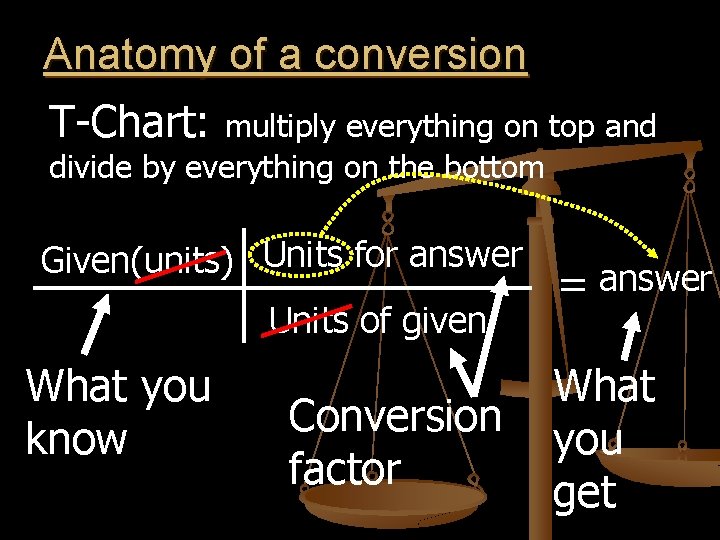
Anatomy of a conversion T-Chart: multiply everything on top and divide by everything on the bottom Given(units) Units for answer Units of given What you know Conversion factor = answer What you get

Summary 18 Mg(s) + 2 HCl(aq) Mg. Cl 2(aq) + H 2(g) n if 4 molecules of H 2 are created 1. 2. how many Fo. U. of HCl are needed? How many atoms of Mg are used?

Standard 3 b: The Mole Atoms and molecules are so small scientists must use a large number of atoms, molecules or formula units in order to observe chemical reactions. This large number is called: A Mole

Standard 3 c: Avogadro’s Number The number of particles in one mole = Avogadro’s number = 6. 02 x 1023 = 602, 000, 000, 000 Particles = atoms, molecules, formula units, donuts…

Summary 19 n How many dollars would you have if you had Avogadro’s number of dollars?

How big is a mole? If every person on earth (6 billion people) started counting 1 mole of objects (6. 02 x 1023 objects) And everyone could count one object per second it would take… Over 3 million years to count to Avogadro’s number!

n One mole of dollars would allow you to give away 1 million dollars to every person on earth every day for nearly 300, 000 years.

n One mole of paper would reach outside our solar system.

n n n If you added 1 mole of ‘special’ water molecules to the world water supply… And waited until it was mixed in… every cup of water on earth would contain over 100 molecules of ‘special’ water

n n n One mole of atoms fits in the palm of my hand. Avogadro’s number is convenient because a mole of any chemical is easily measured in the laboratory. Instead of using 6. 02 x 1023 atoms we use 1 mole of atoms.

Summary 20 4 Na(s) + O 2(g) 2 Na 2 O(s) 1. 2. 3. How many moles of O 2 are needed to react with 4 moles of sodium? How many moles of O 2 are needed to react with 2 moles of sodium? How many moles of Na 2 O are produced when 2 moles of sodium are used?
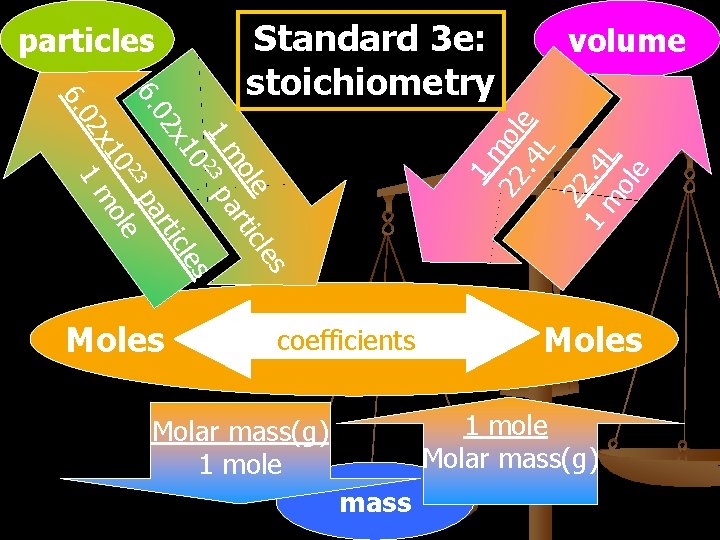
particles Moles volume 1 m 22 ole. 4 L 22 1. 4 m L ole 6. 0 le s ole tic m ar 1 23 p 0 les x 1 tic 02 ar 6. 3 p 2 e 10 ol 2 x 1 m Standard 3 e: stoichiometry coefficients Moles 1 mole Molar mass(g) 1 mole mass
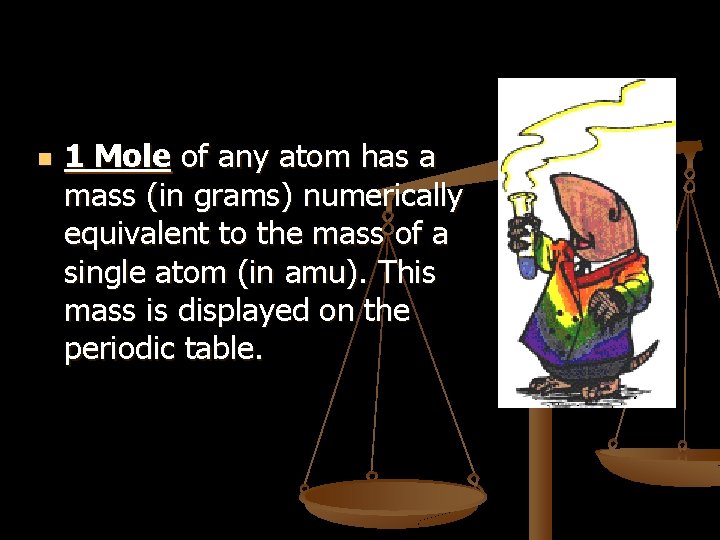
n 1 Mole of any atom has a mass (in grams) numerically equivalent to the mass of a single atom (in amu). This mass is displayed on the periodic table.

Summary 21 n What is the mass of one mole of: Aluminum? n Carbon? n

Standard 3 d: Molar Mass = the mass of one mole of any substance. n Example: Na 2 O Sodium = 23 g/mole Oxygen = 16 g/mole 1 mole Na 2 O: (2 x 23 g/mole) + 16 g/mole = The molar mass for Na 2 O = 62 g/mole

Summary 22 n calculate the molar mass of Al. Cl 3 (don’t forget units!)

examples n What is the mass of 3 moles of sodium hydroxide? Molar mass! 3 moles Na. OH 40 g Na. OH 1 mole Na. OH n = 120 g Na. OH How many moles of carbon dioxide are in a sample weighing 88 g? 88 g CO 2 1 mole CO 2 44 g CO 2 = 2 moles CO 2

Summary 23 A person produces just less than 0. 5 moles CH 4 per day. How many grams is that?

The volume of a Mole 1 mole of any GAS has a volume of 22. 4 L at standard temperature and pressure n Standard temperature = 0°C n Standard pressure = 1 atm

Summary 24 1. What is the volume of 2 moles of gas? 2. What is the volume of 0. 5 moles of gas? (At standard temperature and pressure)

examples n What is the volume of 3 moles of helium? 3 moles He 22. 4 L He 1 mole He n = 67. 2 L He How many moles of nitrogen are inside a 224 L container? 224 L 1 mole N 2 22. 4 L = 10 mole N 2

Summary 25 A person produces just less than 11. 2 L CH 4 per day. How many moles is that (assume STP conditions)?
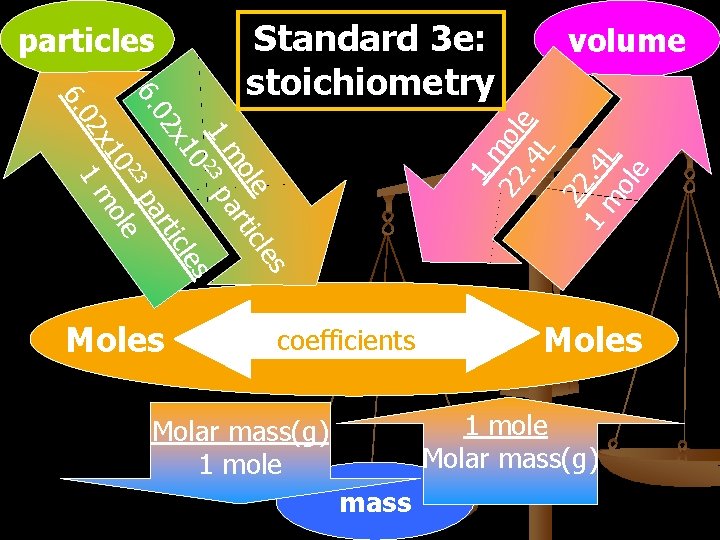
particles Moles volume 1 m 22 ole. 4 L 22 1. 4 m L ole 6. 0 le s ole tic m ar 1 23 p 0 les x 1 tic 02 ar 6. 3 p 2 e 10 ol 2 x 1 m Standard 3 e: stoichiometry coefficients Moles 1 mole Molar mass(g) 1 mole mass

Example. n n Mg(s) + 2 HCl(aq) Mg. Cl 2(aq) + H 2(g) If 72 g hydrochloric acid are used, how many liters of hydrogen gas are produced? 73 g HCl 1 mole HCl 36. 5 g HCl 1 mole H 2 22. 4 L H 2 2 moles HCl 1 mole H 2 = 22. 4 L H 2

Summary 26 1. 2. Write the balanced equation for the reaction of hydrogen (H 2(g)) with nitrogen (N 2(g)) to form ammonia (NH 3(g)). If one mole of nitrogen is used: a. b. c. d. How many moles of hydrogen are needed? How many liters (of hydrogen) is that? How many moles of NH 3(g) are produced? How many grams of NH 3(g) are produced?
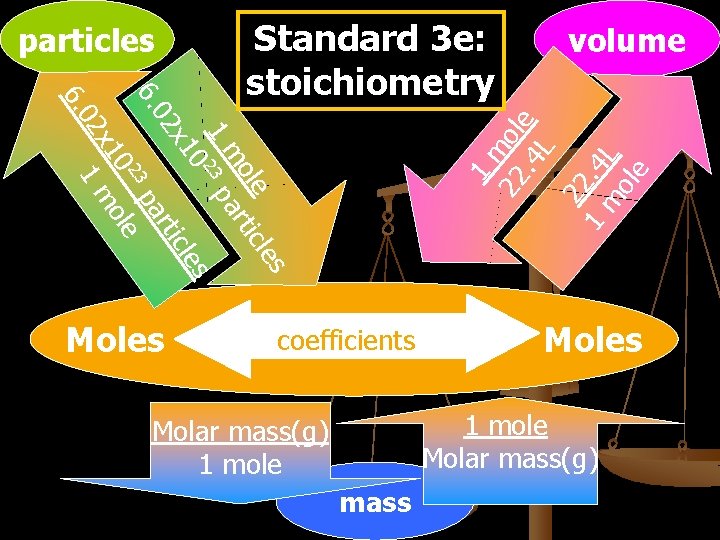
particles Moles volume 1 m 22 ole. 4 L 22 1. 4 m L ole 6. 0 le s ole tic m ar 1 23 p 0 les x 1 tic 02 ar 6. 3 p 2 e 10 ol 2 x 1 m Standard 3 e: stoichiometry coefficients Moles 1 mole Molar mass(g) 1 mole mass

examples n How many molecules are contained in 2 moles of CO 2? 2 moles CO 2 6 x 1023 molecules = 12 x 1023 molecules CO 2 1 mole CO 2 24 = 1. 2 x 10 n How many atoms are contained in 2 moles of CO 2? 2 mol CO 2 6 x 1023 molcs 3 atoms 1 mol CO 2 1 molc CO 2 = 36 x 1023 atoms = 3. 6 x 1024

Summary 27 A person produces just less than 0. 5 moles CH 4 per day. How many molecules is that?
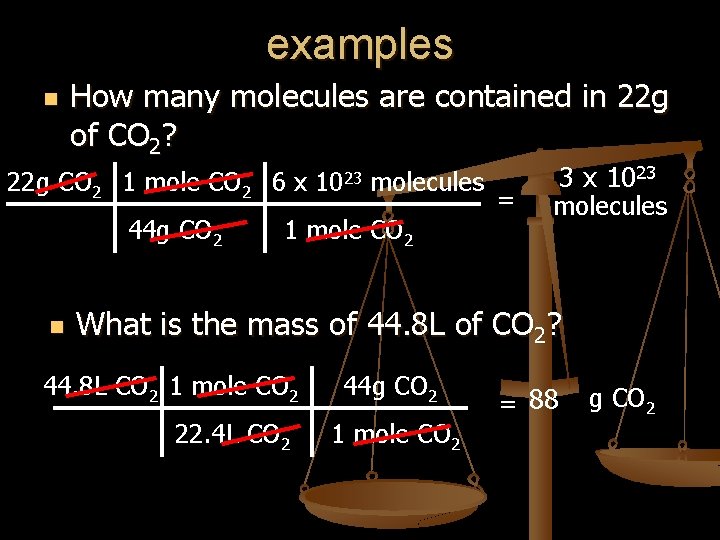
examples n How many molecules are contained in 22 g of CO 2? 22 g CO 2 1 mole CO 2 6 x 1023 molecules 44 g CO 2 n 1 mole CO 2 = 3 x 1023 molecules What is the mass of 44. 8 L of CO 2? 44. 8 L CO 2 1 mole CO 2 22. 4 L CO 2 44 g CO 2 1 mole CO 2 = 88 g CO 2

Summary 28 A cow produces about 1500 L CH 4 per day. How many grams is that?


Note: Scientific notation n n One digit before the decimal ONLY Number of digits indicates accuracy Power of 10 indicates magnitude e. x. 1. 0 x 103 Accurate to 2 digits n Number is equivalent to approximately 1, 000 n n e. x 1. 0 x 10 -3 n Number equivalent to 0. 0010

Summary 17 Write the following numbers using scientific notation: 1. 67, 000 2. 67, 141 3. 0. 01 4. 0. 0100

Example. n n Mg(s) + 2 HCl(aq) Mg. Cl 2(aq) + H 2(g) If 2 moles of magnesium reacts, how many molecules of hydrogen are produced? 2 mole Mg 1 mole H 2 1 mole Mg 6. 0 x 1023 molecules H 2 1 mol H 2 12 x 1023 = molecules H 2 1. 2 x 1024 molecules H 2

Predicting reactions (Honors only). Reactions are driven by: n The formation of a solid e. x. Pb(NO 3)2(aq) + 2 KI(aq) Pb. I 2(s) + 2 KNO 3(aq) n The formation of a gas e. x. Ca. CO 3(s) + 2 HCl(aq) Ca. Cl 2(aq) + H 2 O(l) + CO 2(g)

Solubility Rules (honors only). All compounds that contain the following ions are soluble: group 1 n NH 4+ n NO 3 n HCO 3 n C 2 H 3 O 2 n Cl. O 3 n Cl. O 4 n

Solubility Rules (honors only). n All compounds that contain Cl-, Br-, Iare soluble n n All compounds that contain SO 42 - are soluble n n Except: Ag+, Hg+, Pb 2+ Except: Ag+, Hg 2+, Pb 2+ , Ca 2+, Sr 2+, Ba 2+ All compounds that contain S 2 - are insoluble n Except: group 1, group 2, NH 4+

Examples (honors only). ALL OTHER COMPOUNDS ARE SOLIDS e. x. Pb(NO 3)2(aq) + KI(aq) KNO 3 + Pb. I 2 n n Formation of a solid or gas? PI 2 is solid therefore reaction proceeds e. x. Na. Cl(aq) + KNO 3(aq) Na. NO 3 + KCl n Formation of a solid or gas? n No! no reaction
- Slides: 87