COMPLICATIONS OF DIABETES MELLITUS CHAIR Dr B JAYAKUMAR

COMPLICATIONS OF DIABETES MELLITUS CHAIR : Dr. B. JAYAKUMAR

�ACUTE COMPLICATIONS Hypoglycemia DKA, HSS �CHRONIC COMPLICATIONS Microvascular Macrovascular

HYPOGLYCEMIA �It is a clinical syndrome in which low serum or plasma glucose levels lead to symptoms of sympathoadrenal activation (sweating, anxiety, tremor, nausea, palpitations, tachycardia) from increased secretion of counterregulatory hormones. �Neuroglycopenia occurs as the glucose level decreases further (fatigue, dizziness, headache, visual disturbances, drowsiness, difficulty speaking, inability to concentrate, abnormal behavior, confusion and loss of consciousness or seizures.

�In diabetics a blood glucose <70 mg/d. L �In diabetics - iatrogenic factors �In non diabetics – fasting _ postprandial q. RISK FACTORS – skipped or insufficient meals, unaccustomed physical exertion, misguided therapy, alcohol ingestion and drug overdose.

Source: American Diabetes Association. Standards of Medical Care in Diabetes— 2008. Diabetes Care. 2008; 31: S 12–S 54. Normal & Target Blood Glucose Ranges Normal Blood Glucose Levels in People Who Do Not Have Diabetes �Upon waking—fasting : 70 to 99 mg/d. L �After meals : 70 to 140 mg/d. L Target Blood Glucose Levels in People Who Have Diabetes �Before meals : 70 to 130 mg/d. L � 1 to 2 hours after the start of a meal : <180 mg/d. L

�Hypoglycemia can also happen during sleep. Some signs of hypoglycemia during sleep include : Ø crying out or having nightmares Ø finding pajamas or sheets damp from perspiration Ø feeling tired, irritable, or confused after waking up

_Hypoglycemia unawareness � - develops when frequent episodes of hypoglycemia lead to changes in how the body reacts to low blood glucose levels. The body stops releasing the hormone epinephrine and other stress hormones when blood glucose drops too low. The loss of the body’s ability to release stress hormones after repeated episodes of hypoglycemia is called hypoglycemia-associated autonomic failure, or HAAF.

�Other medications that can cause hypoglycemia include salicylates, including aspirin , alcohol, warfarin sulfa medications pentamidine tolbutamide, chlorpropamide, allopurinol, probenecid
TREATMENT �Oral Glucose �IV Dextrose �Glucagon 1 mg IM or SC �PREVENTION – � Patient education � Monitor blood glucose frequently

DIABETIC KETOACIDOSIS �Occurs in 5% of Pts with Type 1 DM �Precipitating factors – interruption of insulin therapy, infection, trauma, infarction and pregnancy. �Symptoms – polyuria, polydipsia, wt. loss, nausea, vomiting, and vaguely localized abdominal pain. �Signs – dehydration, tachycardia, hypotension, tachypnea, Kussmaul breathing, abdominal tenderness and fruity breath odour.

Pathophysiology �Relative or absolute insulin deficiency combined with counterregulatory hormone excess. �Decreased ratio of insulin to glucagon promotes gluconeogenesis, glycogenolysis and ketone body formation. �Ketone bodies are neutralized by bicarbonates leading to acidosis. Increased lactic acid production also contributes.

Diagnosis �Hyperglycemia (pregnancy & alcohol ingestion – euglycemic DKA) �Positive urine ketone �Metabolic acidosis (increased anion gap) �Hyponatremia, hyperkalemia, azotemia �Precipitating infection �ECG

treatment �Fluid replacement �Adequate insulin administration �Potassium repletion

�Estimate fluid deficit. Average degree of dehydration is 7 -9% of body wt. 10% if in hypotension. �Restoration of circulating volume using 0. 9% saline. �Replenish total body water deficits. �Maintenance fluid replacement.

�IV bolus of regular insulin at 0. 15 unit/kg followed by infusion at 0. 1 unit/kg/hr. �A decrease in blood glucose of 50 -75 mg/d. L/hr is appropriate. �Lesser decrements – insulin resistance �Rapid correction – osmotic encephalopathy � 5% dextrose in saline should be infused once plasma glucose reduces to 250 mg% and insulin infusion reduced to 0. 05 unit/kg/hr.

�Give the first SC injection of insulin approximately 30 min before stopping the infusion. �Potassium infusion at the rate of 10 -20 m. Eq/hr except in patients with hyperkalemia, renal failure or oliguria. �Hypokalemia – 40 m. Eq/hr

�Bicarbonate therapy – in shock or coma, severe acidosis, severe depletion of plasma bicarbonate, acidosis induced cardiac or respiratory dysfunction or severe hyperkalemia. �IV Antibiotics �Monitoring – Hourly blood glucose, serum electrolytes 2 hourly and ABG as required.

Complications of DKA �Lactic acidosis �Arterial thrombosis – stroke, MI or ischemic limb �Cerebral edema �Rebound ketoacidosis

Hyperglycemic hyperosmolar state �Occurs primarily in Pts with Type 2 DM. �Less common than DKA : incidence of <1 case per 1000 person-years. �Prototypical Pt is an elderly individual with type 2 DM, with a several week history of polyuria, wt loss and decreased oral intake that culminates in mental confusion, lethargy or coma.

�Signs – profound dehydration, hypotension, tachycardia, altered mental status. �No nausea, vomiting, abdominal pain or Kussmaul’s respiration. �Precipitating factors – stress, infection, stroke, non compliance with medications, dietary indiscretion, and alcohol and cocaine abuse

�Pathophysiology – relative insulin deficiency and inadequate fluid replacement. �Insulin def increases hepatic glucose production and impairs glucose utilization. �Hyperglycemia induces osmotic diuresis that leads to intravascular vol depletion, which is exacerbated by inadequate fluid replacement.

Diagnosis �Hyperglycemia, often >600 mg/d. L �Plasma osmolality >320 m. Osm/L �Absence of ketonemia �p. H >7. 3 �Serum bicarbonate >20 m. Eq/L �DD – hypoglycemia, hyponatremia, severe dehydration, uremia, hyperammonemia, drug overdose and sepsis.

treatment �Fluid therapy – 10 -12 L positive fluid balance over 2436 hrs. �Insulin therapy – IV bolus of 5 -10 units, followed by continuous infusion of 0. 1 -0. 15 units/kg/hr. Once plasma glucose reduces to 200 -300 mg/d. L, insulin infusion can be decreased to 1 -2 units/hr. 5% dextrose should be added. �IV antibiotics

�Monitoring of therapy – blood glucose every 30 -60 min and serum electrolytes every 1 -2 hrs. �COMPLICATIONS – thromboembolic events, cerebral edema, adult respiratory distress syndrome, rhabdomyolysis.

LACTIC ACIDOSIS �When there is lack of oxygen in blood, mitochondria cannot burn all the pyruvate produced by glycolysis; pyruvate accumulates in the cell which is converted to lactate causing lactic acidosis. �Signs – deep and rapid breathing, vomiting and abdominal pain

�COHEN-WOODS CLASSIFICATION TYPE A – reduced perfusion or oxygenation TYPE B – B 1 – underlying disease - B 2 – medication or intoxication - B 3 – inborn error of metabolism

Causes �Genetic – MELAS, Glycogen storage diseases �Drugs – Phenformin, Metformin, INH toxicity, Nucleoside Reverse Transcriptase Inhibitors Potassium cyanide Others – hypoxia and hypoperfusion, hemorrhage disease Ethanol toxicity, sepsis, shock, hepatic DKA, muscular exercise, renal hypoperfusion
�S. p. H - <7. 35 �Lactate levels >5 �TREATMENT - IV line – fluid repletion Treat cause Bicarbonate therapy –starting dose one third to half of calculated HCO 3 deficit = 0. 5 x wt in kg x (desired HCO 3 –

ACUTE SEPSIS �Sepsis is a response to infections that can lead to widespread inflammation and blood clotting. �CAUSES – urinary tract infection - pelvic infections - peri odontic infections - skin and soft tissue infections

�People with diabetes and sepsis were more likely to develop acute renal failure than people without diabetes, whereas they were less likely to develop acute respiratory failure.
Chronic complications �MICROVASCULAR – Eye disease – diabetic retinopathy - macular edema Neuropathy – sensory and motor - autonomic Nephropathy

�MACROVASCULAR – Coronary artery disease Peripheral arterial disease Cerebrovascular disease v. OTHER – Gastrointestinal, genitourinary, dermatologic Infections Cataracts, glaucoma, periodontal disease

Mechanisms of complications 1) Increased intracellular glucose leads to the formation of advanced glycosylation end products (AGEs), which cross-link proteins, accelerate atherosclerosis, promote glomerular dysfunction, reduce nitric oxide synthesis, induce endothelial dysfunction, and alter extracellular matrix composition and structure.

2) Hyperglycemia induces metabolism of glucose via sorbitol pathway. Increased sorbitol conc. alters redox potential, increases cellular osmolality, generates reactive oxygen species and leads to other types of cellular dysfunction.

3) Hyperglycemia increases the formation of diacylglycerol leading to activation of protein kinase C, which alters the transcription of genes for fibronectin, type IV collagen, contractile proteins, and extracellular matrix proteins in endothelial cells and neurons.

4) Hyperglycemia increases flux through the hexosamine pathway, which may alter function by glycosylation of proteins or by changes in gene expression of TGF-B or PAI-1.

Clinical trials �The Diabetes Control and Complications Trial (DCCT) – multicentric clinical trial – 1400 individuals with type 1 DM. � Intensive and conventional diabetes management groups. �Prospective evaluation of development of retinopathy, neuropathy and nephropathy.

�Twice Intensive Conventional Multiple daily administrtn group insulin group of insulin injections. �Extensive educational, psychological and �Quarterly nutritional, medical support. educational and clinical �evaluatn. Goal – normoglycemia �Preventn Reductionofinsymptoms Hb. A 1 c – 7. 3% � 9. 1%

Results �Improvement of glycemic control reduced proliferative and non proliferative retinopathy (47%), microalbuminuria(39%), clinical nephropathy(54%), and neuropathy(60%). �If all complications of diabetes were combined, individuals in the intensive group would experience 15. 3 more years of life without significant complications.

�The United Kingdom Prospective Diabetes Study (UKPDS) – studied the course of >5000 individuals with type 2 diabetes for >10 yrs. �Newly diagnosed individuals with type 2 DM were randomized to (1) intensive management using various combinations of insulin and OHA; or (2) conventional therapy using dietary modification and pharmacotherapy. Also randomly assigned to different antihypertensive regimens.

Results �Each percentage point reduction in A 1 c was associated with 35% reduction in microvascular complications. �Strict blood pressure control significantly reduced both microvascular and macrovascular complications.

�Kumamoto study – small trial of lean Japanese individuals with type 2 DM randomized to either intensive glycemic control or standard therapy with insulin. �Reduction in risk of nephropathy and retinopathy.

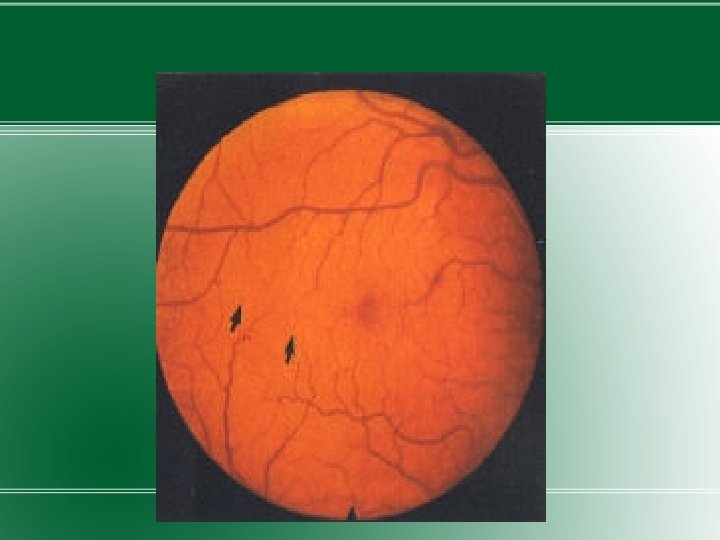
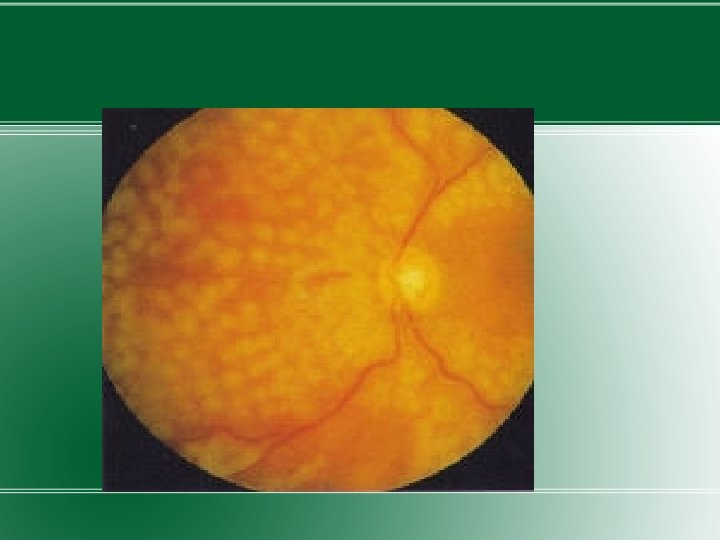
DIABETIC RETINOPATHY �DM is the leading cause of blindness in US. �Diabetic retinopathy has four stages: �Mild Nonproliferative Retinopathy. At this earliest stage, microaneurysms occur. They are small areas of balloon-like swelling in the retina's tiny blood vessels. �Moderate Nonproliferative Retinopathy. As the disease progresses, some blood vessels that nourish the retina are blocked. �Severe Nonproliferative Retinopathy. Many more blood vessels are blocked, depriving several areas of

�Proliferative Retinopathy. At this advanced stage, the signals sent by the retina for nourishment trigger the growth of new blood vessels. This condition is called proliferative retinopathy. These new blood vessels are abnormal and fragile. They grow along the retina and along the surface of the clear, vitreous gel that fills the inside of the eye. By themselves, these blood vessels do not cause symptoms or vision loss. However, they have thin, fragile walls. If they leak blood, severe vision loss and even blindness can result.


�Osmotic stress from sorbitol accumulation has been postulated as an underlying mechanism in the development of diabetic microvascular complications, including diabetic retinopathy. �Duration of DM and degree of glycemic control are the best predictors of development of retinopathy.
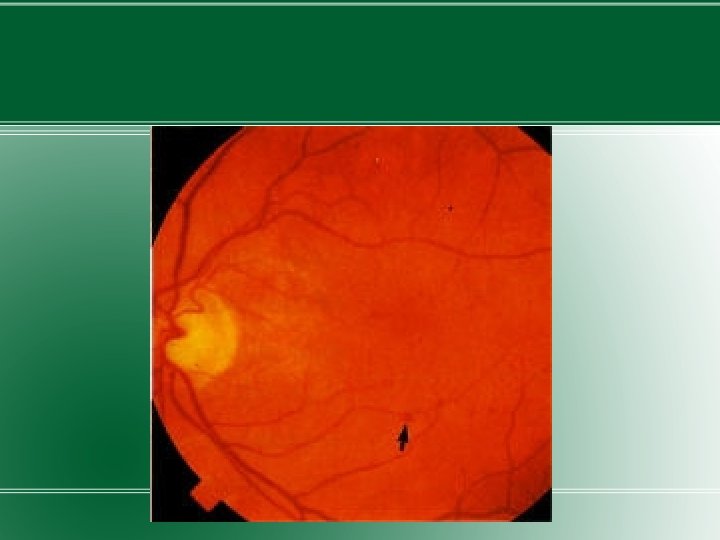

Treatment �Most effective therapy – prevention – intensive glycemic and blood pressure control. �Annual examination by ophthalmologist. �Proliferative retinopathy – panretinal laser photocoagulation

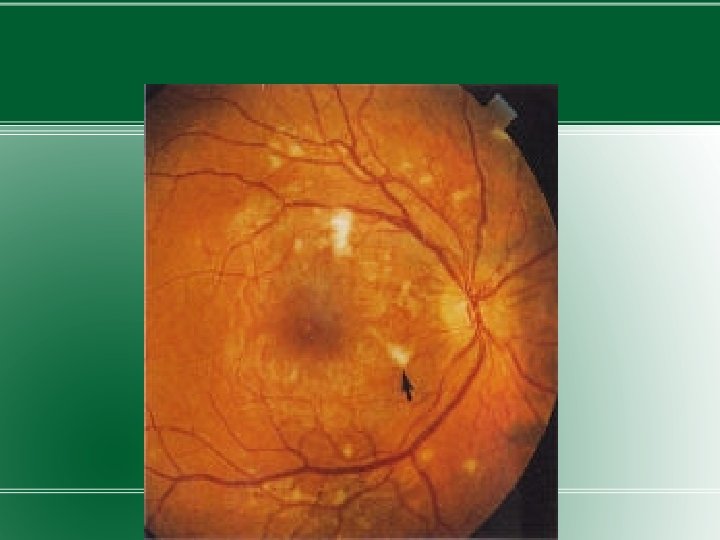
MACULAR EDEMA � - swelling of retina in diabetes due to leaking of fluid from blood vessels within the macula. �As macular edema develops, blurring develops in the middle or just to the side of the central visual field. �Lifetime risk to develop macular edema in diabetics is 10%. �Visual loss from macular edema can progress over mths. �The condition is closely related with the degree of diabetic retinopathy.

�Hypertension and fluid retention are other factors. �Fluid retention is mainly caused by loss of protein in urine. �Classified into focal and diffuse types �Focal – caused by foci of vascular abnormalities primarily microaneurysms which tend to leakage fluid. �Diffuse – caused by dilated retinal cappilaries in the retina
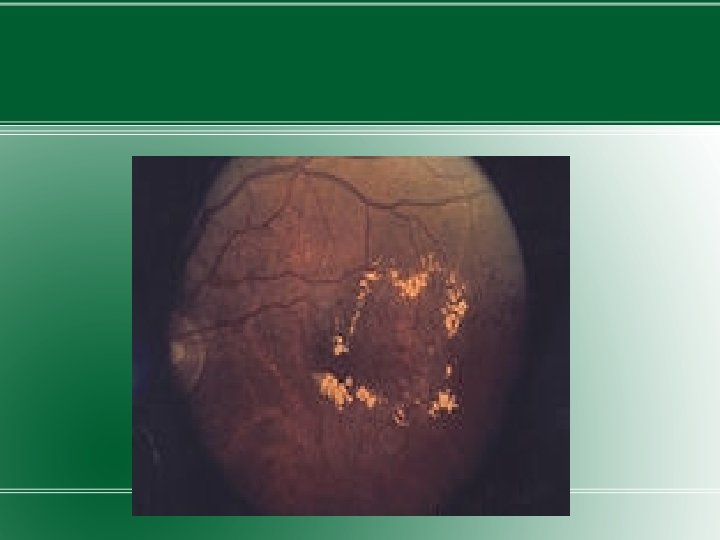

Treatment �Focal – Focal laser treatment – to close leaking microaneurysms �Diffuse – Grid laser treatment – produce retinal burn of mild to moderate intensity �Patient is rechecked several mths after treatment and if not responding, laser treatment is repeated. �Goal of treatment is to reduce chances of progressive visual loss. Visual acuity does not improve.

TRIALS q. DIABETIC RETINOPATHY STUDY (DRS) Scatter photocoagulation significantly reduces the risk of severe visual loss from proliferative diabetic retinopathy. q. EARLY TREATMENT DIABETIC RETINOPATHY STUDY (ETDRS) Focal photocoagulation reduces risk of moderate visual loss by 50% or more

�DIABETIC RETINOPATHY VITRECTOMY STUDY (DRVS) It provided guidelines for the most opportune time for vitrectomy surgery for patients with type 1 and 2 diabetes who suffered from vitreous hemorrhage or from severe PDR in eyes with useful vision.

�DIABETIC RETINOPATHY CANDESARTAN TRIALS (DIRECT) Candesartan compared to placebo reduces the progression of diabetic retinopathy in normotensive, normoalbuminuric patients with type 1 diabetes and retinopathy. An 18% reduction in incidence was observed.

DIABETIC NEUROPATHY �Diabetic neuropathy occurs in approx. 50%. �Polyneuropathy, mononeuropathy and/or autonomic neuropathy. �Correlates with duration of diabetes and glycemic control. �Risk factors – increased BMI, smoking, cardiovascular disease, elevated triglycerides, hypertension.

�Nerve damage is likely due to a combination of factors: 1) metabolic factors, such as high blood glucose, long duration of diabetes, and possibly low levels of insulin 2) neurovascular factors, leading to damage to the blood vessels that carry oxygen and nutrients to nerves 3) autoimmune factors that cause inflammation in nerves

�ADA recommends screening for distal symmetric neuropathy beginning with the initial diagnosis of diabetes and for autonomic neuropathy 5 years after diagnosis of type 1 DM and at the time of diagnosis of type 2 DM. Then screened annually for both forms of neuropathy.
Polyneuropathy �Most common form of diabetic neuropathy is distal symmetric polyneuropathy. �Symptoms – distal sensory loss, sensation of numbness, tingling, sharpness or burning sensation in the feet that spreads proximally. �O/E – sensory loss, loss of ankle reflexes and abnormal position sense.

�Sensorimotor diabetic peripheral polyneuropathy is a major risk factor foot trauma, ulceration and Charcot arthropathy; and is responsible for 50 – 75% of non- traumatic amputations. �Neuropathic pain develops in some individuals. �Pain typically involves the lower extremities, is usually present at rest and worsens at night.

�Both acute and chronic form �As diabetic neuropathy progresses, pain disappears but a sensory deficit in the lower extremities persists.

Polyradiculopathy �Diabetic polyradiculopathy is a syndrome characterised by severe disabling pain in the distribution of one or more nerve roots. �It may be accompanied by motor weakness. �Intercostal or truncal radiculopathy causes pain over thorax and abdomen.

�Involvement of lumbar plexus or femoral nerve causes severe pain in the thigh or hip and maybe associated with muscle weakness in the hip flexors or extensors ( diabetic amyotrophy ). �Usually self-limiting and resolve over 6 -12 mths.

Mononeuropathy �Dysfunction of isolated cranial or peripheral nerve. �Vascular etiology suggested, pathogenesis unknown. � 3 rd cranial nerve most common – diplopia, ptosis, ophthalmoplegia with normal pupillary constriction to light. �Sometimes IV, VI or VII are affected.

Autonomic neuropathy �Longstanding diabetics develop autonomic dysfunction involving cholinergic, noradrenergic and peptidergic systems. �Can involve cardiovascular, gastrointestinal, genitourinary, sudomotor and metabolic systems. �Resting tachycardia and orthostatic hypotension.

�Hyperhidrosis of upper extremities and anhidrosis of lower extremities result from sympathetic nervous system dysfunction. �Hypoglycemia unawareness due to reduced counterregulatory hormone release.

Treatment �Screening – sensation in the lower extremities should be documented at least annually, using either a lighttouch monofilament or a tuning fork of frequency 128 Hz. �Symptoms of diabetic neuropathy may not necessarily improve. �Risk factors for neuropathy should be treated.

�Avoidance of neurotoxins (alcohol) and smoking, supplementation with vitamins for possible deficiencies and symptomatic treatment. �Patients should check their feet daily and take precautions to prevent calluses or ulcer formation.

�Chronic painful neuropathy may respond to TCAs – Amitryptyline, nortryptyline, imipramine, desipramine SSRIs – duloxetine Anticonvulsants – gabapentin, pregabalin, carbamazepine, lamotrigine �Duloxetine and pregabalin are approved by the U. S. Food and Drug Administration specifically for treating painful diabetic peripheral neuropathy

�Since the pain may resolve over time, medications may be discontinued as progressive neuronal damage occurs �Orthostatic hypotension – non-pharmacologic measures like adequate salt intake, avoidance of dehydration and diuretics, and lower extremity support hose. Fludrocortisone (0. 1 -0. 3 mg/day), midodrine, clonidine, octreotide, yohimbine

TRIALS JOURNAL OF AMERICAN MEDICAL ASSOCIATION DEC 2 , 1998 Gabapentin for the Symptomatic Treatment of Painful Neuropathy in Patients with Type 2 DM Randomized, double blind, placebo controlled, 8 week trial between July ‘ 96 and March ‘ 97 �RESULTS – Gabapentin treated patients daily score at the study end point was significantly lower (P<. 001) compared with placebo treated patients end point score

� DIABETESCAN AUGUST 1999 �Treatment of diabetic polyneuropathy with the antioxidant alpha-lipoic acid : a 7 mth multicentric randomized trial (ALADIN III) Alpha lipoic acid in diabetic neuropathy �RESULTS – No significant differences

� NEUROLOGY MARCH 25, 2003 �Controlled release Oxycodone for pain in diabetic neuropathy �Multicentric, randomized, double blind, placebo controlled, parallel group study for 6 weeks �RESULTS – At an average dose of 37 mg/day, CR Oxycodone provided more analgesia than placebo.

� NEUROLOGY 2001 �Lamotrigine reduces painful diabetic neuropathy �Randomized, placebo controlled trial over 6 weeks �RESULTS – Global assessment of efficacy favored lamotrigine treatment over placebo

� NEUROLOGY DEC 2004 �Pregabalin relieves symptoms of Painful Diabetic Neuropathy �Multicentric, randomized, double blind, placebo controlled study �RESULTS – Patients in Pregabalin group showed improvement in end point mean pain score vs. placebo

DIABETIC NEPHROPATHY �Leading cause of DM related morbidity and mortality. � 20 – 40% of diabetics develop nephropathy �Smoking accelerates decline in renal function �Family history of diabetic nephropathy is a known risk factor.

�Pathogenesis involves the effects of soluble factors (growth factors, angiotensin II, endothelins), hemodynamic alterations in the renal microcirculation (glomerular hyperfiltration or hyperperfusion, increased glomerular capillary pressure) and structural changes in the glomerulus (increased EC matrix, basement membrane thickening, mesangial expansion, fibrosis).

�In the first years – glomerular hyperperfusion and renal hypertrophy occur with increase in GFR. �During first 5 yrs – thickening of the glomerular basement membrane, glomerular hypertrophy and mesangial volume expansion occurs. �After 5 -10 yrs – approx. 40% develop microalbuminuria

�MICROALBUMINURIA – is defined as 30 -300 mg/d in a 24 hr collection or 30 -300 mcg/mg creatinine in a spot collection (preferred method).

�Approx. 50% progress to macroalbuminuria over the next 10 yrs. �There is a steady decline in GFR and approx. 50% reach ESRD in 7 -10 yrs. �Once macroalbuminuria develops, blood pressure rises slightly and the pathologic changes are likely irreversible.

�Type 2 DM v/s type 1 DM 1) present at diagnosis 2) hypertension more common 3) microalbuminuria less predictive – maybe secondary to hypertension, CHF, prostate disease or infection

�Type IV renal tubular acidosis (hyporeninemic hypoaldosteronism) may occur in type 1 or 2 DM – propensity to develop hyperkalemia. �Predisposed to radiocontrast induced nephrotoxicity. Risk factors – preexisting nephropathy and volume depletion. Individuals with DM should be well hydrated before and after dye exposure, and the s. Cr should be monitored for 24 hrs.

�Screening for microalbuminuria should be performed in patients with type 1 DM >5 yrs, in patients with type 2 DM and during pregnancy. �Other conditions that might increase microalbuminuria are UTI, hematuria, heart failure, febrile illness, severe hyperglycemia, severe hypertension and vigorous exercise.

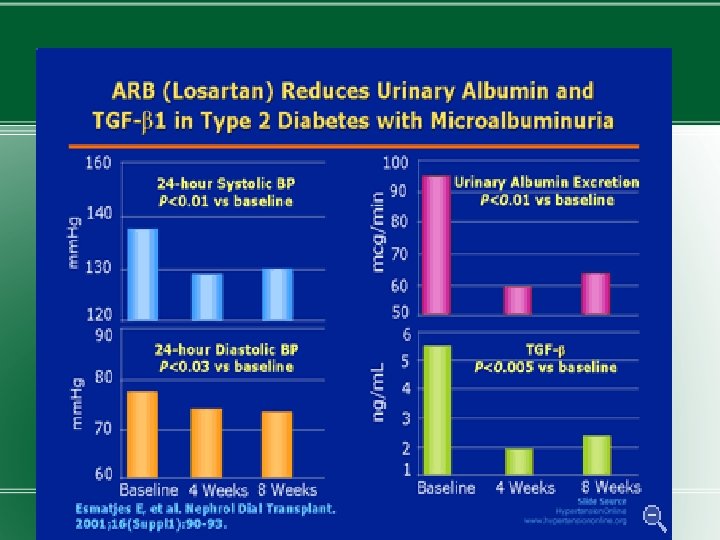
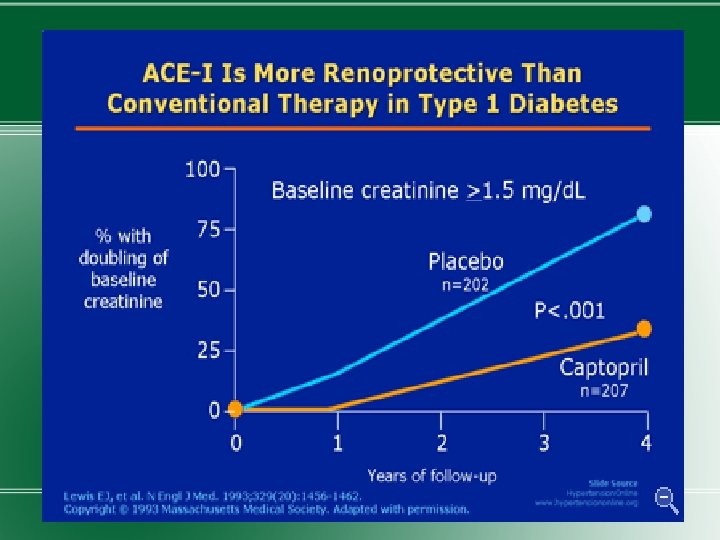
Treatment � 1) Normalization of glycemia � 2) Strict blood pressure control � 3) Administration of ACEIs or ARBs �Improved glycemic control reduces the rate at which microalbuminuria appears and progresses.

�Blood pressure should be maintained at <130/80 mm. Hg in diabetic individuals without proteinuria. A slightly lower blood pressure for those with microalbuminuria. �ACEIs or ARBs should be used to prevent progression. The drug dose is increased after 2 -3 mths of therapy till microalbuminuria disappears or max. dose reached.

�If not possible, Ca channel blockers, beta blockers or diuretics should be used. �ADA suggests modest restriction of protein intake in microalbuminuria – 0. 8 g/kg/day and <0. 8 g/kg/day. �Nephrology consultation when the estimated GFR < 60 ml/min/1. 743 m 2.


�Hemodialysis in patients with DM is associated with more frequent complications such as hypotension (due to autonomic neuropathy or loss of reflex tachycardia), more difficult vascular access and accelerated progression of retinopathy. �Atherosclerosis is the leading cause of death in patients on dialysis.

�Renal transplant from a living related donor is the preferred therapy, but requires chronic immunosuppression. �Combined pancreas-kidney transplant offers the promise of normoglycemia and freedom from dialysis.

TRIALS



THANK YOU
- Slides: 100