Complications of 90 Y Transarterial Radioembolization of Hepatic

Complications of 90 Y Transarterial Radioembolization of Hepatic Tumors: A Primer for Interventional Radiology Residents and Fellows Nanda Venkatanarasimha, MBBS, MRCP (UK), FRCR (UK), EBIR, FRANZCR (Aus); Apoorva Gogna, MBBS, FRCR (UK), FAMS; Ravi Muli Jogi, MBBS, FRANZCR (Aus); Karthikeyan Damodharan, MBBS, MRCP (UK), FRCR (UK); Shaun Xavier Chan, MBBS, FRCR (UK); David Chee Eng Ng, MBBS, MRCP (UK), FAMS; Kelvin Siu Hoong Loke, MBBS, MRCP (UK), FAMS; Farah G. Irani, MBBS, MD, FRCR (UK), FAMS; Richard Haou Gong Lo, MBBS, MD, FRCR (UK), FAMS; Sivanathan Chandramohan, MBBS, MRCS (UK), FRCR (UK), EBIR

From the Department of Vascular and Interventional Radiology (N. V. , A. G. , R. M. J. , K. D. , S. X. C. , F. G. I. , R. H. G. L. , S. C. ), and Department of Nuclear Medicine and Molecular Imaging (D. C. E. N. , K. S. H. L. ), Singapore General Hospital, Outram Road, Singapore 169608. Address correspondence to N. V. (e-mail: drnanda 29@gmail. com). Presented as an education exhibit at the 2017 RSNA Annual Meeting. The authors D. C. E. N. , K. S. H. L. , R. H. G. L. , and F. G. I. have provided disclosures; all other authors have disclosed no relevant relationships. Disclosures of Conflicts of Interest. —D. C. E. N. Activities related to the present article: research grant, honorarium, and hotel accommodation and airfare for attendance at relevant conferences from Sirtex. Activities not related to the present article: disclosed no relevant relationships. Other activities: disclosed no relevant relationships. K. S. H. L. Activities related to the present article: disclosed no relevant relationships. Activities not related to the present article: payment for lectures from Sirtex masterclass 2017. Other activities: disclosed no relevant relationships. R. H. G. L. Activities related to the present article: disclosed no relevant relationships. Activities not related to the present article: disclosed no relevant relationships. Other activities: proctor for Sirtex Medical Singapore. F. G. I. Activities related to the present article: disclosed no relevant relationships. Activities not related to the present article: payment for lectures at regional conferences and related travel expenses from BARD, Boston Scientific, and Sirtex. Other activities: disclosed no relevant relationships.

Content Organization § Introduction § Learning objectives § Patient selection criteria and dosimetry § Classification § Case examples with management § Lessons learned from quality assurance rounds § Conclusion Radioembolization of all cases presented in this exhibit was performed with resin-based yttrium 90 ( 90 Y)–labeled microspheres (SIR-Spheres; Sirtex Medical, North Sydney, Australia).

Introduction § 90 Y radioembolization treatment is generally safe, and serious complications are rare. § Complications may occur from nontarget deposition of 90 Y microspheres, radiationinduced parenchymal injury to liver, or technical reasons related to angiography and embolization. § Most complications related to 90 Y radioembolization can be avoided with meticulous attention to patient selection, techniques, and dose optimization.

Learning Objectives § Everyone involved in treating patients with radioembolization should be familiar with the wide spectrum of complications and strategies to prevent and manage complications when they occur. § After viewing this presentation, the reader should be able to: Ø Describe and illustrate complications related to transarterial radioembolization of liver tumors Ø Describe valuable tips to prevent and manage complications that are based on lessons learned from quality assurance exercises Ø Explain the combination of interventional radiology techniques that can help prevent complications

Patient Selection Criteria Careful scrutiny of every case at a multidisciplinary meeting equals successful radioembolization procedure and in-turn reduction in complication rates! Inclusion criteria Exclusion criteria § Unresectable primary liver cancer and/or liver-dominant metastases § Age older than 18 years § Eastern Cooperative Oncology Group grade less than or equal to 2 § Serum bilirubin level less than 2 mg/d. L § Serum creatinine level less than 2 mg/d. L § Platelet count more than 60 x 109/L § Can undergo angiography and selective visceral vascular catheterization § Child-Pugh score greater than B 7 § Lung shunt fraction (LSF) greater than 20% for resin or estimated radiation dose to lungs more than 30 Gy from a single session or cumulative dose of 50 Gy from repeated treatment § Uncorrectable hepatoenteric shunts § Uncorrectable arterioportal macro shunts § Significant extrahepatic disease § Receiving bevacizumab or sorafenib★ ★Relative contraindication, should be stopped prior to procedure Venkatanarasimha et al. Clin Radiol 2017; 30: 752– 764

Preprocedural Planning § Ensure appropriate patient selection; review inclusion and exclusion criteria carefully § Obtain relevant up-to-date laboratory test results and correct abnormal results where appropriate: full blood count, coagulation, and renal panel § Liver panel assessment. Child-Pugh score is most commonly used. § Preprocedural multiphasic computed tomographic (CT) or magnetic resonance (MR) imaging provides excellent information on tumor vascularity; enhancement characteristic of the tumor; hepatic arterial, portal, and venous anatomy; and potential nontarget arteries to avoid. Ideally done within 6 weeks of workup. § Tumor feeding vessels can be identified and depicted with three-dimensional (3 D) volume-rendering techniques, which can shorten interventional radiology procedural time and thus radiation. Continues on next slide Paprottka et al. Cardiovasc Intervent Radiol 2012; 35: 454 -462

Preprocedural Planning § CT or MR imaging can also help assess the biliary tree for risks of bacterial contamination, such as previous hepatoenteric anastomosis or sphincterotomy. § CT allows estimation of future remnant liver volume and tumor volume. This can be done either manually or with automated segmentation techniques. § Volume assessment is valuable when planning radiation segmentectomy or when considering tumor downstaging with a view for future resection. § Order appropriate preprocedural medications: Ø Antibiotics for patients at risk for biliary contamination Ø Steroids and antihistamines may be needed in patients with contrast material allergy.

Technique § Transarterial radioembolization requires an initial planning hepatic angiography as a separate procedure, typically performed 1– 2 weeks prior to 90 Y implantation. § Planning angiography allows tumor mapping; select appropriate injection point(s) and take steps to prevent (eg, embolization) ectopic delivery of 90 Y. § Planning angiography is combined with 20– 100 -µm-sized particles of technetium 99 m (99 m. Tc)– labeled macroaggregated albumin (MAA) scintigraphy. Anticipated distribution and activity of 90 Y microspheres is simulated by the injected 99 m. Tc MAA particles that match the size of 90 Y microspheres. § Nuclear medicine physician can use the injected activity to calculate the amount of liver-to–lung shunt ratio and tumor-to–normal liver ratio to enable dose prescription. Continues on next slide Salem et al. Tech Vasc Interv Radiol 2007; 10: 12– 29

Technique § Standard transfemoral or transradial arterial access § 4 - 0 r 5 -F catheters are used to perform coeliac axis and superior mesenteric angiography. § We always use microcatheters and microwire, which are advanced coaxially via the parent catheter for superselective hepatic angiography. Ø Microcatheters facilitate cannulation of small and tortuous vessels and minimize vessel injury. § Intra-arterial CT (with hybrid angiography-CT machine) performed at planning angiography Ø Allows accurate volumetric assessment of tumor coverage by the vessel being evaluated Ø Improves operator confidence to detect nonhepatic or replaced vessels that may be difficult to detect at digital subtraction angiography (DSA) § During 90 Y procedure, cannulate same branches where 99 m. Tc MAA was injected, verify the position with angiographic results, and then deliver 90 Y microspheres. Burgmans et al. Eur J Radiol 2012; 81: 3979– 3984
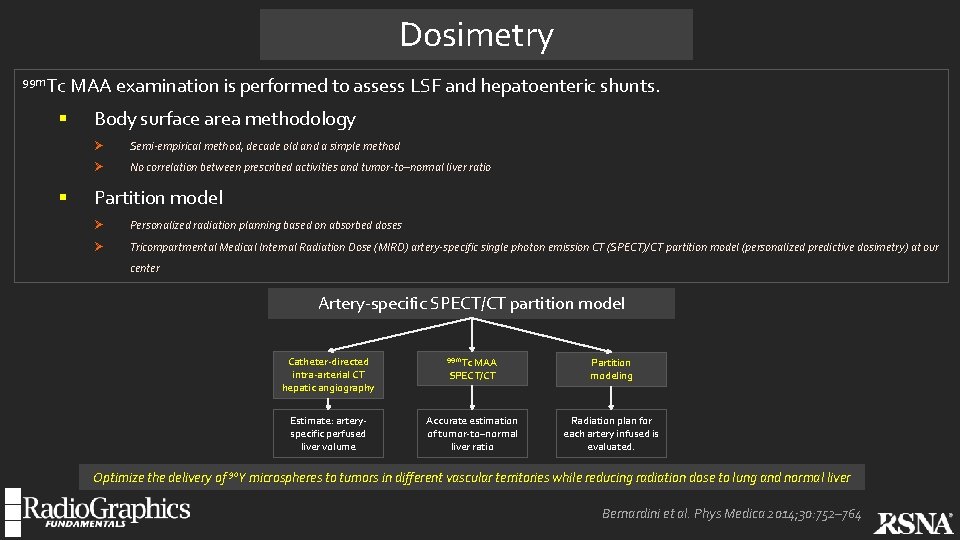
Dosimetry 99 m. Tc § § MAA examination is performed to assess LSF and hepatoenteric shunts. Body surface area methodology Ø Semi-empirical method, decade old and a simple method Ø No correlation between prescribed activities and tumor-to–normal liver ratio Partition model Ø Personalized radiation planning based on absorbed doses Ø Tricompartmental Medical Internal Radiation Dose (MIRD) artery-specific single photon emission CT (SPECT)/CT partition model (personalized predictive dosimetry) at our center Artery-specific SPECT/CT partition model Catheter-directed intra-arterial CT hepatic angiography Estimate: arteryspecific perfused liver volume 99 m. Tc MAA SPECT/CT Partition modeling Accurate estimation of tumor-to–normal liver ratio Radiation plan for each artery infused is evaluated. Optimize the delivery of 90 Y microspheres to tumors in different vascular territories while reducing radiation dose to lung and normal liver Bernardini et al. Phys Medica 2014; 30: 752– 764
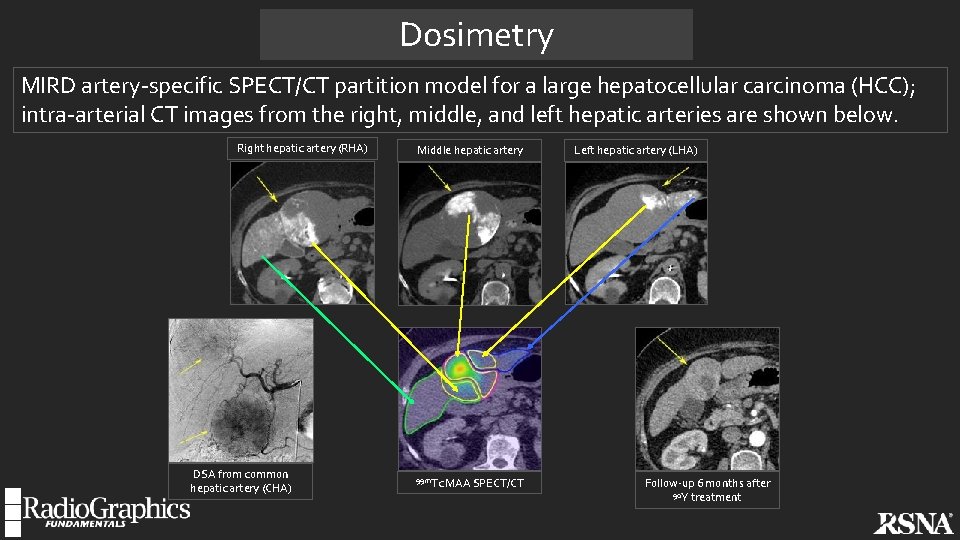
Dosimetry MIRD artery-specific SPECT/CT partition model for a large hepatocellular carcinoma (HCC); intra-arterial CT images from the right, middle, and left hepatic arteries are shown below. Right hepatic artery (RHA) DSA from common hepatic artery (CHA) Middle hepatic artery 99 m. Tc MAA SPECT/CT Left hepatic artery (LHA) Follow-up 6 months after 90 Y treatment

Dosimetry Advantages of compartment modeling and artery-specific partition model § Personalized treatment dose § Determination of tumor-to–normal liver ratio § More accurate calculation of organ-specific radiation absorbed dose § Allows optimal treatment dose calculation within known safe radiation limits for each partition § Scientifically superior to empirical body surface area methodology, particularly in tumors with multiple feeding arteries or tumors with hypovascular components § Greater physician confidence Kao et al. J Nucl Med 2012; 53: 1– 10

Classification Pulmonary § § § Radiation pneumonitis Atelectasis Pleural effusion Gastrointestinal § § Ulceration Pancreatitis Hepatobiliary § § Radiation-induced liver disease (REILD) Radiation cholecystitis Fibrosis Portal hypertension Dermatologic § Radiation dermatitis Hematologic § § Thrombocytopenia Lymphopenia Miscellaneous § § Splenic radiation Diaphragmatic perforation Vascular § § Prior transarterial chemoembolization (TACE) and vessel dissection Antineoangiogenic treatment Although glass- and resin-based 90 Y microspheres differ in their physical properties, their clinical outcomes are similar!

Pulmonary § Incidence of radiation pneumonitis less than 1% if standard dosimetry models are used § Usually develops 4– 6 weeks after 90 Y radioembolization § Restrictive respiratory dysfunction is seen. § Chest radiograph or CT image shows typical bat wing appearance with relative sparing of perihilar region. § Initial ground-glass changes may resolve or may progress to fibrosis and respiratory failure. § Presence of microspheres on transbronchial biopsy specimens is confirmatory. It is a diagnosis of exclusion in the absence of pathologic confirmation. § Bevacizumab and sorafenib are radiosensitizers, and use should be stopped for 2– 4 weeks before the procedure. § Corticosteroids are the treatment of choice. Pentoxifylline can help. § Other pleuropulmonary complications of transarterial radioembolization include atelectasis and pleural effusions. Leung et al. Int J Radiat Oncol Biol Phys 1995; 33: 919– 924
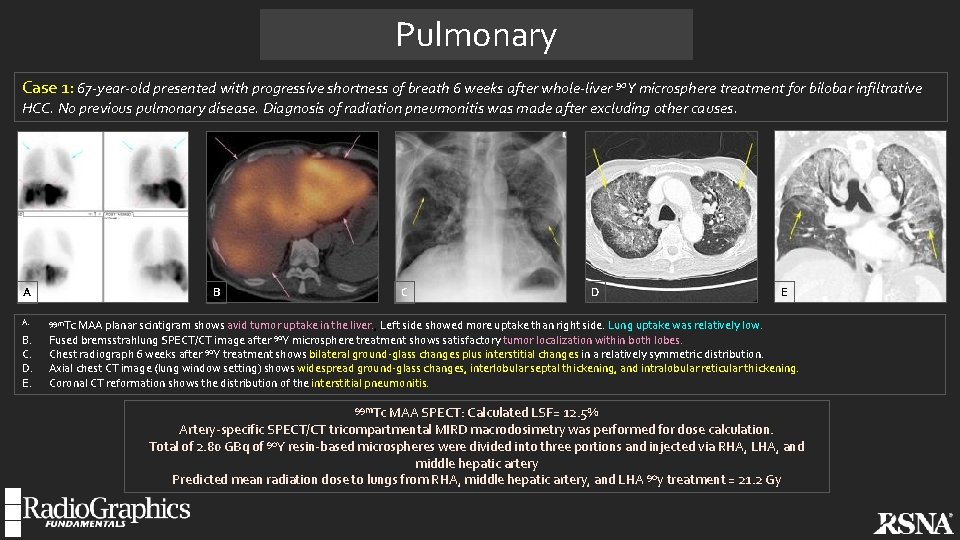
Pulmonary Case 1: 67 -year-old presented with progressive shortness of breath 6 weeks after whole-liver 90 Y microsphere treatment for bilobar infiltrative HCC. No previous pulmonary disease. Diagnosis of radiation pneumonitis was made after excluding other causes. B A A. B. C. D. E. C D E 99 m. Tc MAA planar scintigram shows avid tumor uptake in the liver. , Left side showed more uptake than right side. Lung uptake was relatively low. Fused bremsstrahlung SPECT/CT image after 90 Y microsphere treatment shows satisfactory tumor localization within both lobes. Chest radiograph 6 weeks after 90 Y treatment shows bilateral ground-glass changes plus interstitial changes in a relatively symmetric distribution. Axial chest CT image (lung window setting) shows widespread ground-glass changes, interlobular septal thickening, and intralobular reticular thickening. Coronal CT reformation shows the distribution of the interstitial pneumonitis. 99 m. Tc MAA SPECT: Calculated LSF= 12. 5% Artery-specific SPECT/CT tricompartmental MIRD macrodosimetry was performed for dose calculation. Total of 2. 80 GBq of 90 Y resin-based microspheres were divided into three portions and injected via RHA, LHA, and middle hepatic artery Predicted mean radiation dose to lungs from RHA, middle hepatic artery, and LHA 90 y treatment = 21. 2 Gy
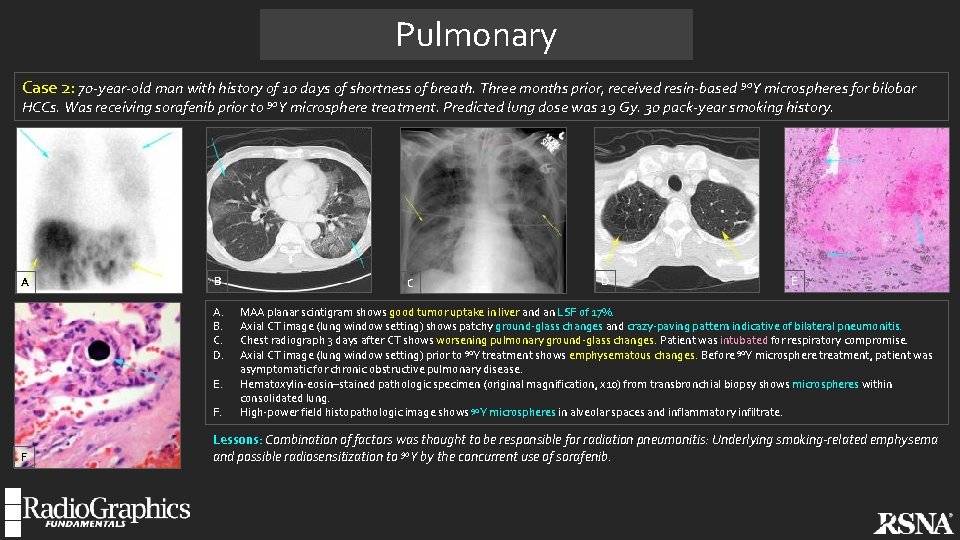
Pulmonary Case 2: 70 -year-old man with history of 10 days of shortness of breath. Three months prior, received resin-based 90 Y microspheres for bilobar HCCs. Was receiving sorafenib prior to 90 Y microsphere treatment. Predicted lung dose was 19 Gy. 30 pack-year smoking history. A B A. B. C. D. E. F. F C D E MAA planar scintigram shows good tumor uptake in liver and an LSF of 17%. Axial CT image (lung window setting) shows patchy ground-glass changes and crazy-paving pattern indicative of bilateral pneumonitis. Chest radiograph 3 days after CT shows worsening pulmonary ground-glass changes. Patient was intubated for respiratory compromise. Axial CT image (lung window setting) prior to 90 Y treatment shows emphysematous changes. . Before 90 Y microsphere treatment, patient was asymptomatic for chronic obstructive pulmonary disease. Hematoxylin-eosin–stained pathologic specimen (original magnification, x 10) from transbronchial biopsy shows microspheres within consolidated lung. High-power field histopathologic image shows 90 Y microspheres in alveolar spaces and inflammatory infiltrate. Lessons: Combination of factors was thought to be responsible for radiation pneumonitis: Underlying smoking-related emphysema and possible radiosensitization to 90 Y by the concurrent use of sorafenib.
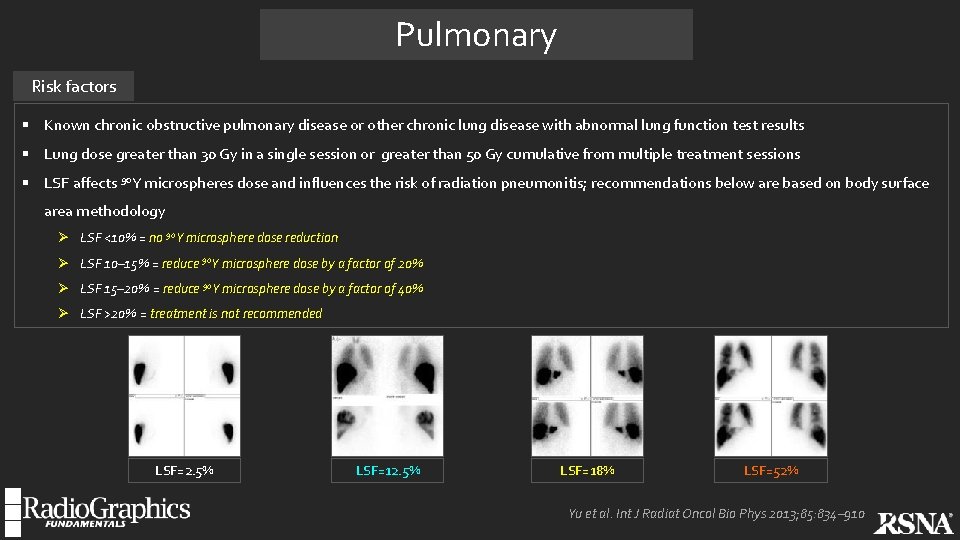
Pulmonary Risk factors § Known chronic obstructive pulmonary disease or other chronic lung disease with abnormal lung function test results § Lung dose greater than 30 Gy in a single session or greater than 50 Gy cumulative from multiple treatment sessions § LSF affects 90 Y microspheres dose and influences the risk of radiation pneumonitis; recommendations below are based on body surface area methodology Ø LSF <10% = no 90 Y microsphere dose reduction Ø LSF 10– 15% = reduce 90 Y microsphere dose by a factor of 20% Ø LSF 15– 20% = reduce 90 Y microsphere dose by a factor of 40% Ø LSF >20% = treatment is not recommended LSF=2. 5% LSF=18% LSF=52% Yu et al. Int J Radiat Oncol Bio Phys 2013; 85: 834– 910
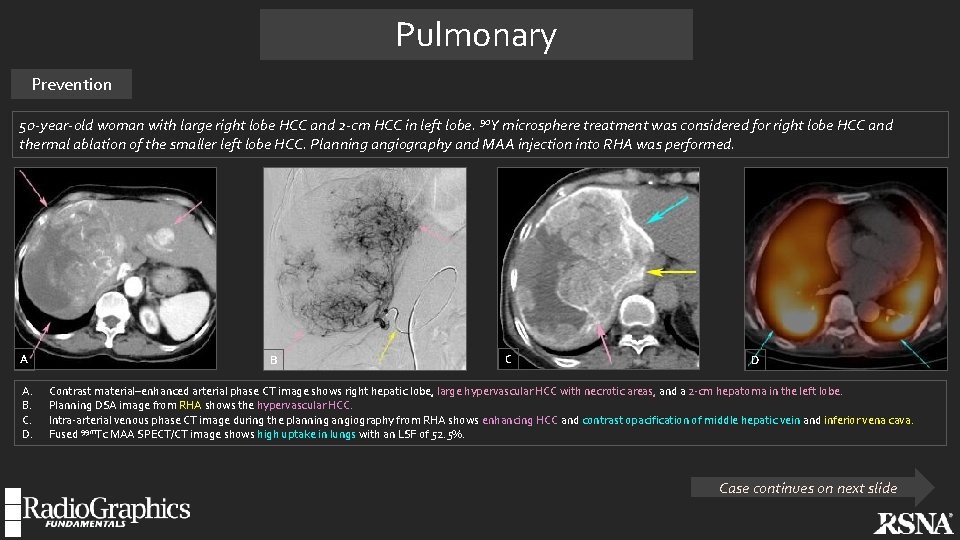
Pulmonary Prevention 50 -year-old woman with large right lobe HCC and 2 -cm HCC in left lobe. 90 Y microsphere treatment was considered for right lobe HCC and thermal ablation of the smaller left lobe HCC. Planning angiography and MAA injection into RHA was performed. A A. B. C. D. B C D Contrast material–enhanced arterial phase CT image shows right hepatic lobe, large hypervascular HCC with necrotic areas, and a 2 -cm hepatoma in the left lobe. Planning DSA image from RHA shows the hypervascular HCC. Intra-arterial venous phase CT image during the planning angiography from RHA shows enhancing HCC and contrast opacification of middle hepatic vein and inferior vena cava. Fused 99 m. Tc MAA SPECT/CT image shows high uptake in lungs with an LSF of 52. 5%. Case continues on next slide
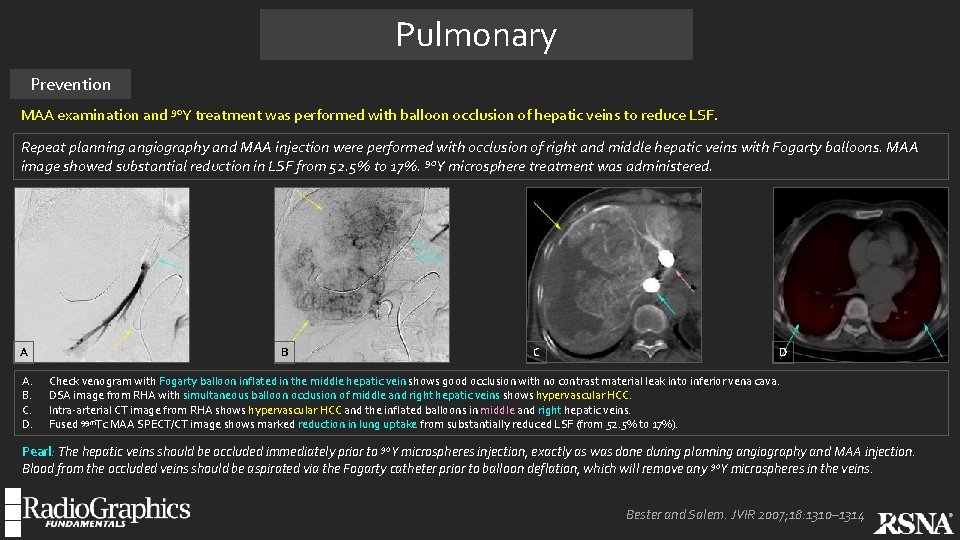
Pulmonary Prevention MAA examination and 90 Y treatment was performed with balloon occlusion of hepatic veins to reduce LSF. Repeat planning angiography and MAA injection were performed with occlusion of right and middle hepatic veins with Fogarty balloons. MAA image showed substantial reduction in LSF from 52. 5% to 17%. 90 Y microsphere treatment was administered. A A. B. C. D. B C D Check venogram with Fogarty balloon inflated in the middle hepatic vein shows good occlusion with no contrast material leak into inferior vena cava. DSA image from RHA with simultaneous balloon occlusion of middle and right hepatic veins shows hypervascular HCC. Intra-arterial CT image from RHA shows hypervascular HCC and the inflated balloons in middle and right hepatic veins. Fused 99 m. Tc MAA SPECT/CT image shows marked reduction in lung uptake from substantially reduced LSF (from 52. 5% to 17%). Pearl: The hepatic veins should be occluded immediately prior to 90 Y microspheres injection, exactly as was done during planning angiography and MAA injection. Blood from the occluded veins should be aspirated via the Fogarty catheter prior to balloon deflation, which will remove any 90 Y microspheres in the veins. Bester and Salem. JVIR 2007; 18: 1310– 1314

Gastrointestinal § Incidence of 2. 9%– 4. 8% § Ulcer starts on serosal site. § Resistant to proton pump inhibitors and more than 50% require surgical interventions § Cone-beam CT or intra-arterial CT review is highly valuable in identifying unexpected anatomic variants. § MAA SPECT/CT is more sensitive than planar imaging at depicting gastric uptake. § Occasionally, SPECT/CT images may show unexpected gastric uptake not seen at planning angiography or cone-beam CT or intra-arterial CT; these cases may warrant repeat angiography or preclude 90 Y treatment. § Diagnostic pitfalls: § Free pertechnetate at SPECT/CT § Misregistration at SPECT/CT Murthy et al. Radio. Graphics 2005; 25: S 41–S 55
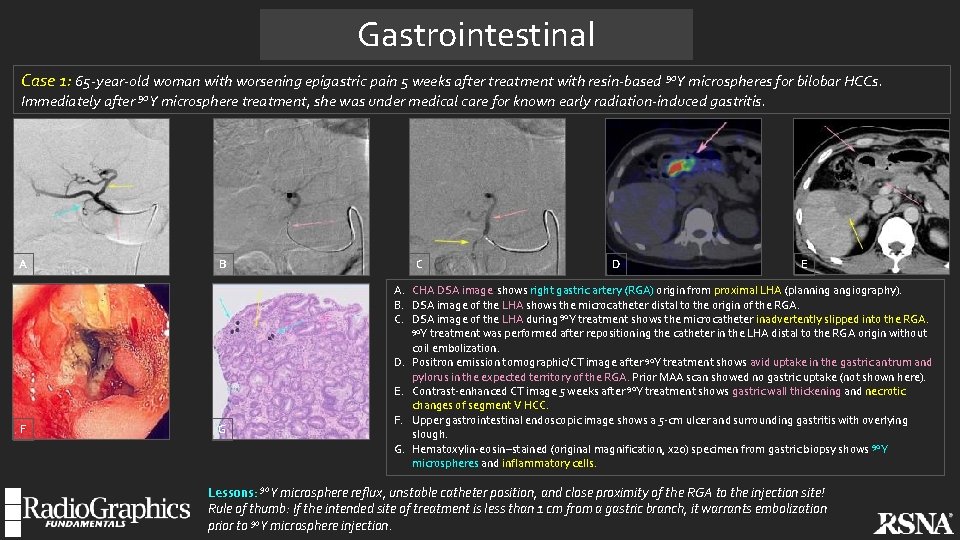
Gastrointestinal Case 1: 65 -year-old woman with worsening epigastric pain 5 weeks after treatment with resin-based 90 Y microspheres for bilobar HCCs. Immediately after 90 Y microsphere treatment, she was under medical care for known early radiation-induced gastritis. A F B G C D E A. CHA DSA image-shows right gastric artery (RGA) origin from proximal LHA (planning angiography). B. DSA image of the LHA shows the microcatheter distal to the origin of the RGA. C. DSA image of the LHA during 90 Y treatment shows the microcatheter inadvertently slipped into the RGA. 90 Y treatment was performed after repositioning the catheter in the LHA distal to the RGA origin without coil embolization. D. Positron emission tomographic/CT image after 90 Y treatment shows avid uptake in the gastric antrum and pylorus in the expected territory of the RGA. Prior MAA scan showed no gastric uptake (not shown here). E. Contrast-enhanced CT image 5 weeks after 90 Y treatment shows gastric wall thickening and necrotic changes of segment V HCC. F. Upper gastrointestinal endoscopic image shows a 5 -cm ulcer and surrounding gastritis with overlying slough. G. Hematoxylin-eosin–stained (original magnification, x 20) specimen from gastric biopsy shows 90 Y microspheres and inflammatory cells. Lessons: 90 Y microsphere reflux, unstable catheter position, and close proximity of the RGA to the injection site! Rule of thumb: If the intended site of treatment is less than 1 cm from a gastric branch, it warrants embolization prior to 90 Y microsphere injection.
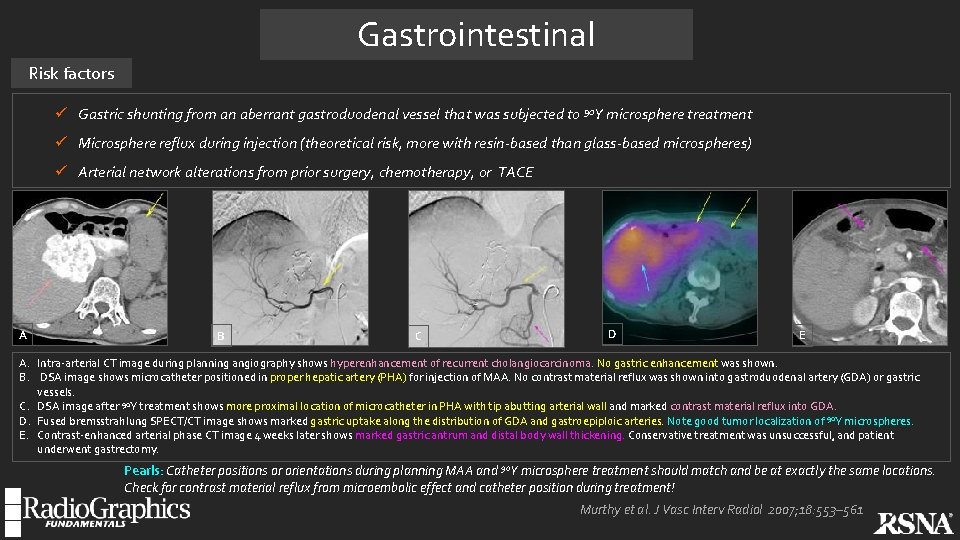
Gastrointestinal Risk factors ü Gastric shunting from an aberrant gastroduodenal vessel that was subjected to 90 Y microsphere treatment ü Microsphere reflux during injection (theoretical risk, more with resin-based than glass-based microspheres) ü Arterial network alterations from prior surgery, chemotherapy, or TACE A B C D E A. Intra-arterial CT image during planning angiography shows hyperenhancement of recurrent cholangiocarcinoma. No gastric enhancement was shown. B. DSA image shows microcatheter positioned in proper hepatic artery (PHA) for injection of MAA. No contrast material reflux was shown into gastroduodenal artery (GDA) or gastric vessels. C. DSA image after 90 Y treatment shows more proximal location of microcatheter in PHA with tip abutting arterial wall and marked contrast material reflux into GDA. D. Fused bremsstrahlung SPECT/CT image shows marked gastric uptake along the distribution of GDA and gastroepiploic arteries. Note good tumor localization of 90 Y microspheres. E. Contrast-enhanced arterial phase CT image 4 weeks later shows marked gastric antrum and distal body wall thickening. Conservative treatment was unsuccessful, and patient underwent gastrectomy. Pearls: Catheter positions or orientations during planning MAA and 90 Y microsphere treatment should match and be at exactly the same locations. Check for contrast material reflux from microembolic effect and catheter position during treatment! Murthy et al. J Vasc Interv Radiol 2007; 18: 553– 561
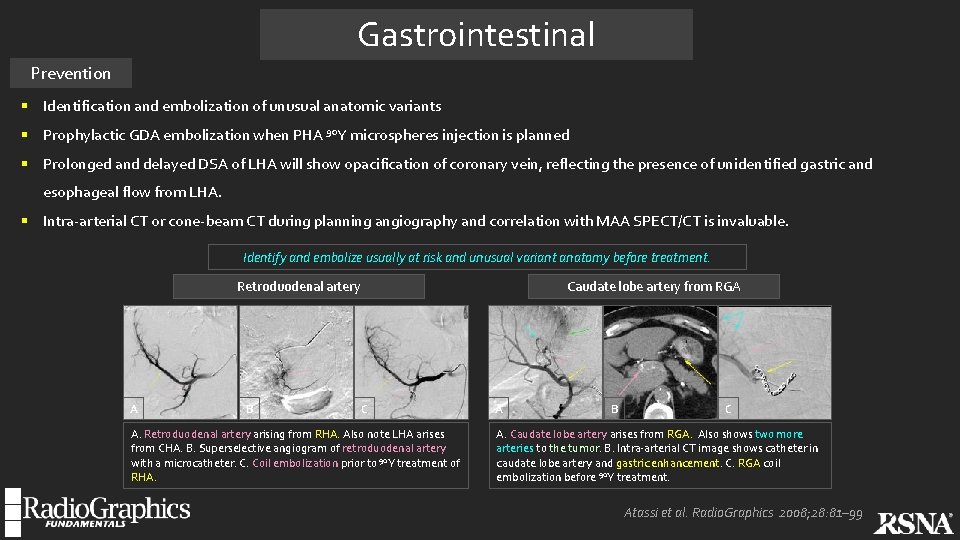
Gastrointestinal Prevention § Identification and embolization of unusual anatomic variants § Prophylactic GDA embolization when PHA 90 Y microspheres injection is planned § Prolonged and delayed DSA of LHA will show opacification of coronary vein, reflecting the presence of unidentified gastric and esophageal flow from LHA. § Intra-arterial CT or cone-beam CT during planning angiography and correlation with MAA SPECT/CT is invaluable. Identify and embolize usually at risk and unusual variant anatomy before treatment. Retroduodenal artery A B Caudate lobe artery from RGA C A. Retroduodenal artery arising from RHA. Also note LHA arises from CHA. B. Superselective angiogram of retroduodenal artery with a microcatheter. C. Coil embolization prior to 90 Y treatment of RHA. A B C A. Caudate lobe artery arises from RGA. Also shows two more arteries to the tumor. B. Intra-arterial CT image shows catheter in caudate lobe artery and gastric enhancement. C. RGA coil embolization before 90 Y treatment. Atassi et al. Radio. Graphics 2008; 28: 81– 99

What and When to Embolize? § Embolization of at-risk hepatoenteric communicating vessels should be performed. § Coil embolization is a safe and effective method in preventing nontarget 90 Y microspheres deposition. § Vessel location for treatment and vessel size further dictate the need for embolization. § Vessels that may need prophylactic embolization include Ø GDA, RGA, and inferior esophageal artery Ø Falciform artery and cystic artery Ø Supraduodenal and retroduodenal arteries § Routine use of intra-arterial CT at our institution for planning angiography has significantly reduced coil embolization rates because of greater operator confidence. § Shorter time interval between prophylactic embolization and 90 Y microsphere treatment may be preferred to avoid the risk of developing new hepatoenteric collateral flow. Salem et al. Tech Vasc Interv Radiol 2007; 10: 12– 29
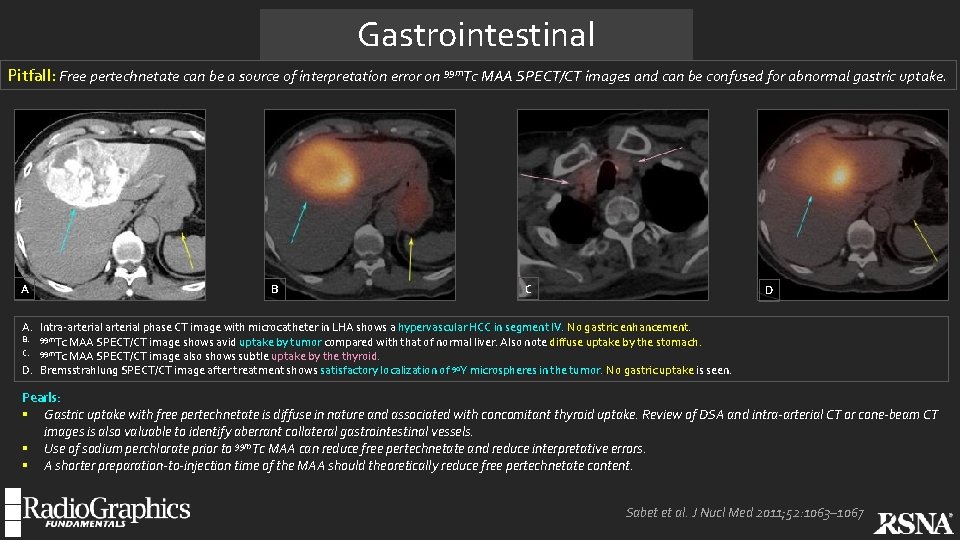
Gastrointestinal Pitfall: Free pertechnetate can be a source of interpretation error on 99 m. Tc MAA SPECT/CT images and can be confused for abnormal gastric uptake. A B C D A. Intra-arterial phase CT image with microcatheter in LHA shows a hypervascular HCC in segment IV. No gastric enhancement. B. 99 m. Tc MAA SPECT/CT image shows avid uptake by tumor compared with that of normal liver. Also note diffuse uptake by the stomach. C. 99 m. Tc MAA SPECT/CT image also shows subtle uptake by the thyroid. D. Bremsstrahlung SPECT/CT image after treatment shows satisfactory localization of 90 Y microspheres in the tumor. . No gastric uptake is seen. Pearls: § Gastric uptake with free pertechnetate is diffuse in nature and associated with concomitant thyroid uptake. Review of DSA and intra-arterial CT or cone-beam CT images is also valuable to identify aberrant collateral gastrointestinal vessels. § Use of sodium perchlorate prior to 99 m. Tc MAA can reduce free pertechnetate and reduce interpretative errors. § A shorter preparation-to-injection time of the MAA should theoretically reduce free pertechnetate content. Sabet et al. J Nucl Med 2011; 52: 1063– 1067

Hepatobiliary § Incidence of REILD is 0%– 5% in large studies. § Usually starts 4– 8 weeks after radioembolization § Jaundice, mild ascites, elevated bilirubin level, and alkaline phosphatase level § On pathologic findings, sinusoidal obstruction or hepatic veno-occlusive disease is seen. § Risk factors: ü Pre-existing liver dysfunction or cirrhosis ü Previous radioembolization or external radiotherapy ü Single-session whole-lobe treatment, then sequential lobar treatment at 6 -week intervals § Other hepatobiliary complications (eg, clinically significant hepatic fibrosis, portal hypertension, hepatic abscess, symptomatic cholecystitis, and biliary stricture) are rare. Braat et al. Eur J Gastroenterol Hepatol 2017; 29: 144– 152
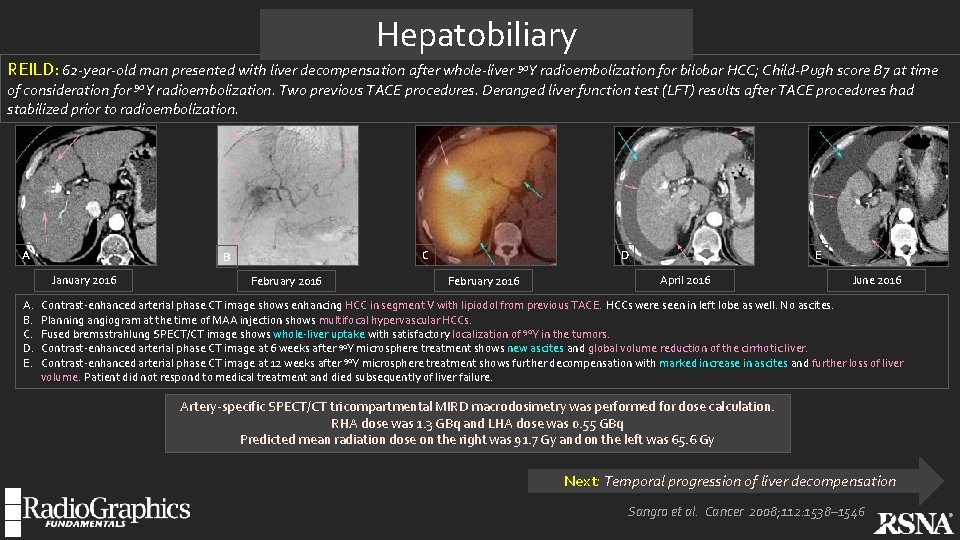
Hepatobiliary REILD: 62 -year-old man presented with liver decompensation after whole-liver 90 Y radioembolization for bilobar HCC; Child-Pugh score B 7 at time of consideration for 90 Y radioembolization. Two previous TACE procedures. Deranged liver function test (LFT) results after TACE procedures had stabilized prior to radioembolization. A January 2016 A. B. C. D. E. C B February 2016 D February 2016 E April 2016 June 2016 Contrast-enhanced arterial phase CT image shows enhancing HCC in segment V with lipiodol from previous TACE. . HCCs were seen in left lobe as well. No ascites. Planning angiogram at the time of MAA injection shows multifocal hypervascular HCCs. Fused bremsstrahlung SPECT/CT image shows whole-liver uptake with satisfactory localization of 90 Y in the tumors. Contrast-enhanced arterial phase CT image at 6 weeks after 90 Y microsphere treatment shows new ascites and global volume reduction of the cirrhotic liver. Contrast-enhanced arterial phase CT image at 12 weeks after 90 Y microsphere treatment shows further decompensation with marked increase in ascites and further loss of liver volume. . Patient did not respond to medical treatment and died subsequently of liver failure. Artery-specific SPECT/CT tricompartmental MIRD macrodosimetry was performed for dose calculation. RHA dose was 1. 3 GBq and LHA dose was 0. 55 GBq Predicted mean radiation dose on the right was 91. 7 Gy and on the left was 65. 6 Gy Next: Temporal progression of liver decompensation Sangro et al. Cancer 2008; 112: 1538– 1546
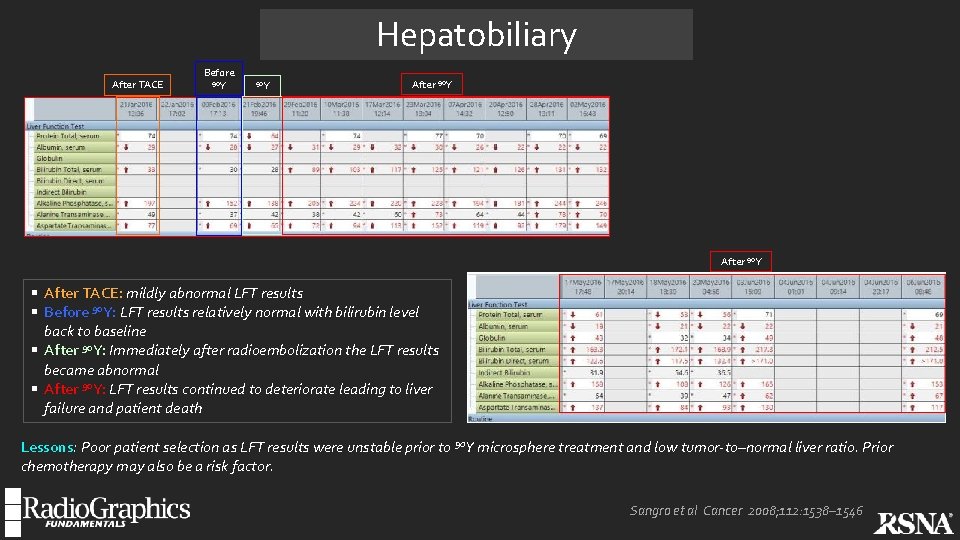
Hepatobiliary After TACE Before 90 Y After 90 Y § After TACE: mildly abnormal LFT results § Before 90 Y: LFT results relatively normal with bilirubin level back to baseline § After 90 Y: Immediately after radioembolization the LFT results became abnormal § After 90 Y: LFT results continued to deteriorate leading to liver failure and patient death Lessons: Poor patient selection as LFT results were unstable prior to 90 Y microsphere treatment and low tumor-to–normal liver ratio. Prior chemotherapy may also be a risk factor. Sangro et al Cancer 2008; 112: 1538– 1546
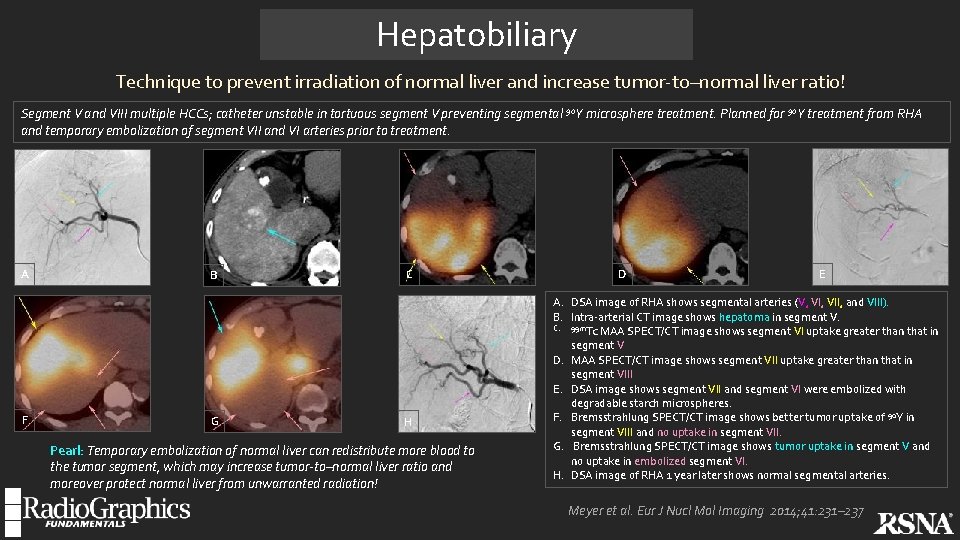
Hepatobiliary Technique to prevent irradiation of normal liver and increase tumor-to–normal liver ratio! Segment V and VIII multiple HCCs; catheter unstable in tortuous segment V preventing segmental 90 Y microsphere treatment. Planned for 90 Y treatment from RHA and temporary embolization of segment VII and VI arteries prior to treatment. A F B G C H Pearl: Temporary embolization of normal liver can redistribute more blood to the tumor segment, which may increase tumor-to–normal liver ratio and moreover protect normal liver from unwarranted radiation! D E A. DSA image of RHA shows segmental arteries (V, VII, and VIII). B. Intra-arterial CT image shows hepatoma in segment V. C. 99 m. Tc MAA SPECT/CT image shows segment VI uptake greater than that in segment V D. MAA SPECT/CT image shows segment VII uptake greater than that in segment VIII E. DSA image shows segment VII and segment VI were embolized with degradable starch microspheres. F. Bremsstrahlung SPECT/CT image shows better tumor uptake of 90 Y in segment VIII and no uptake in segment VII. G. Bremsstrahlung SPECT/CT image shows tumor uptake in segment V and no uptake in embolized segment VI. H. DSA image of RHA 1 year later shows normal segmental arteries. Meyer et al. Eur J Nucl Mol Imaging 2014; 41: 231– 237
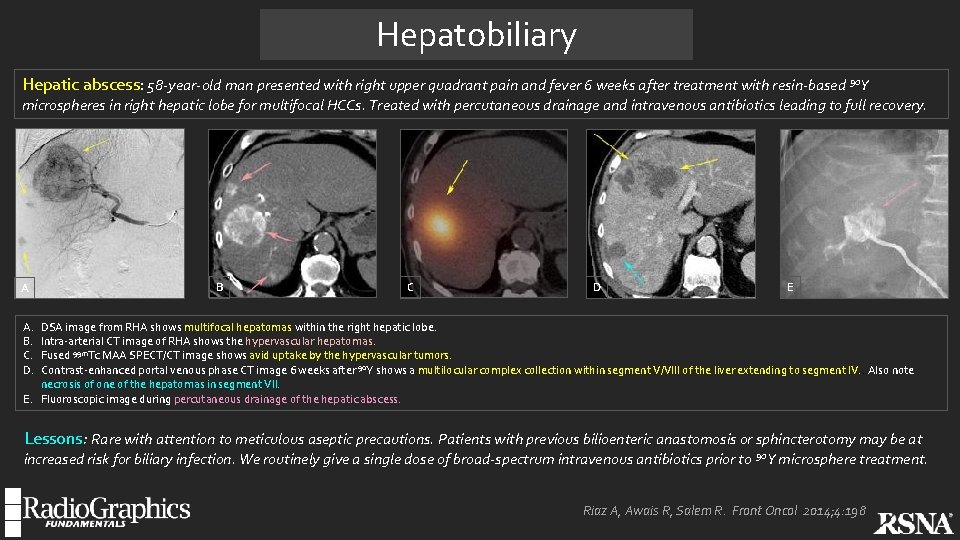
Hepatobiliary Hepatic abscess: 58 -year-old man presented with right upper quadrant pain and fever 6 weeks after treatment with resin-based 90 Y microspheres in right hepatic lobe for multifocal HCCs. Treated with percutaneous drainage and intravenous antibiotics leading to full recovery. A B C D E A. B. C. D. DSA image from RHA shows multifocal hepatomas within the right hepatic lobe. Intra-arterial CT image of RHA shows the hypervascular hepatomas. Fused 99 m. Tc MAA SPECT/CT image shows avid uptake by the hypervascular tumors. Contrast-enhanced portal venous phase CT image 6 weeks after 90 Y shows a multilocular complex collection within segment V/VIII of the liver extending to segment IV. . Also note necrosis of one of the hepatomas in segment VII. E. Fluoroscopic image during percutaneous drainage of the hepatic abscess. Lessons: Rare with attention to meticulous aseptic precautions. Patients with previous bilioenteric anastomosis or sphincterotomy may be at increased risk for biliary infection. We routinely give a single dose of broad-spectrum intravenous antibiotics prior to 90 Y microsphere treatment. Riaz A, Awais R, Salem R. Front Oncol 2014; 4: 198
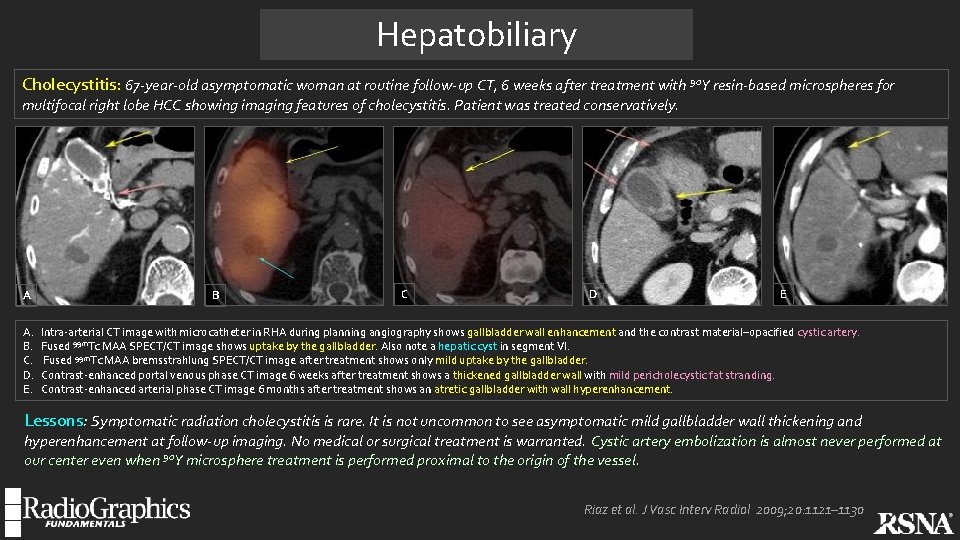
Hepatobiliary Cholecystitis: 67 -year-old asymptomatic woman at routine follow-up CT, 6 weeks after treatment with 90 Y resin-based microspheres for multifocal right lobe HCC showing imaging features of cholecystitis. Patient was treated conservatively. A A. B. C. D. E. B C D E Intra-arterial CT image with microcatheter in RHA during planning angiography shows gallbladder wall enhancement and the contrast material–opacified cystic artery. Fused 99 m. Tc MAA SPECT/CT image shows uptake by the gallbladder. Also note a hepatic cyst in segment VI. Fused 99 m. Tc MAA bremsstrahlung SPECT/CT image after treatment shows only mild uptake by the gallbladder. Contrast-enhanced portal venous phase CT image 6 weeks after treatment shows a thickened gallbladder wall with mild pericholecystic fat stranding. Contrast-enhanced arterial phase CT image 6 months after treatment shows an atretic gallbladder with wall hyperenhancement. Lessons: Symptomatic radiation cholecystitis is rare. It is not uncommon to see asymptomatic mild gallbladder wall thickening and hyperenhancement at follow-up imaging. No medical or surgical treatment is warranted. Cystic artery embolization is almost never performed at our center even when 90 Y microsphere treatment is performed proximal to the origin of the vessel. Riaz et al. J Vasc Interv Radiol 2009; 20: 1121– 1130

Dermatologic § Rare § Can present with paraumbilical pain and rash § Inadvertent entry of 90 Y microspheres into the anterior abdominal vessels § Risk factor: ü Presence of a prominent falciform artery Prevention § Identification of falciform artery, which may be seen only at delayed imaging. Note the vessel is of a small caliber and can have a varying origin. § Intra-arterial CT detection of falciform artery is better than DSA or 99 m. Tc MAA SPECT/CT detection. § Close scrutiny of the 99 m. Tc MAA planar or SPECT/CT scans can be helpful. § Prophylactic coil embolization or topical application of ice packs over the anterior abdomen immediately prior to 90 Y microsphere treatment can prevent radiation dermatitis. Leong et al. J Vasc Interv Radiol 2009; 20: 833– 836
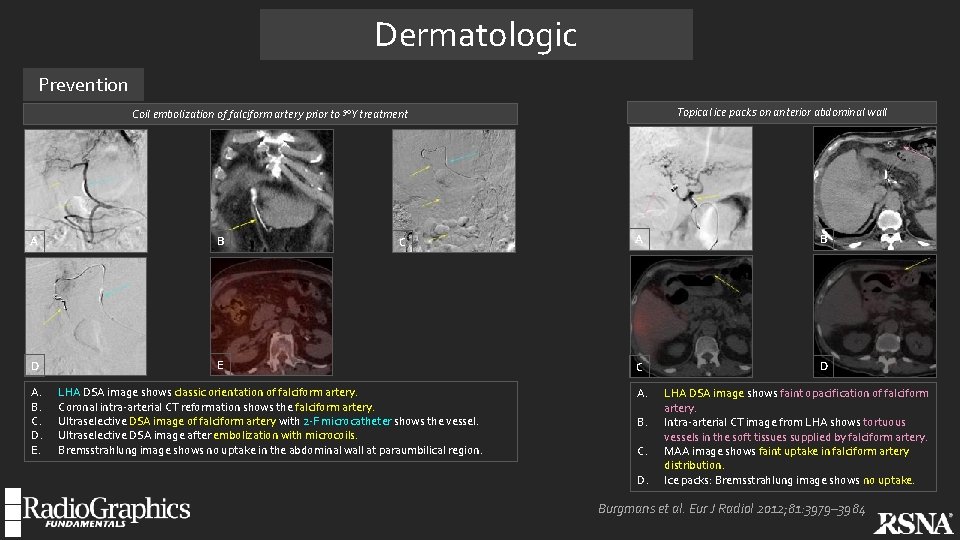
Dermatologic Prevention Topical ice packs on anterior abdominal wall Coil embolization of falciform artery prior to 90 Y treatment A B D E A. B. C. D. E. C LHA DSA image shows classic orientation of falciform artery. Coronal intra-arterial CT reformation shows the falciform artery. Ultraselective DSA image of falciform artery with 2 -F microcatheter shows the vessel. Ultraselective DSA image after embolization with microcoils. Bremsstrahlung image shows no uptake in the abdominal wall at paraumbilical region. A B C D A. B. C. D. LHA DSA image shows faint opacification of falciform artery. Intra-arterial CT image from LHA shows tortuous vessels in the soft tissues supplied by falciform artery. MAA image shows faint uptake in falciform artery distribution. Ice packs: Bremsstrahlung image shows no uptake. Burgmans et al. Eur J Radiol 2012; 81: 3979– 3984

Hematologic § Lymphopenia Ø Reported after radioembolization with glass-based microspheres Ø More than 25% decrease in counts is seen in the majority Ø Asymptomatic, no opportunistic infections reported § Thrombocytopenia Ø Splenomegaly can occur after radioembolization which in turn can lower the platelet count. Ø No bleeding episodes reported from radioembolization-induced thrombocytopenia § Neutropenia Ø Rarely significant Lam MG, Banerjee A, Louie JD, Sze DY. Cardiovasc Intervent Radiol 2014; 37: 1009– 1017
Vascular § Artery dissection Ø Higher risk in patients receiving sorafenib and vascular endothelial growth factor inhibitor (bevacizumab) Ø Previous TACE also increases the risk because of prior arterial damage. Ø These vessels are friable and more prone to perforation and pseudoaneurysm formation. § Abnormal flow Ø Peripheral hepatic arterial pruning and increased risk of vasospasm is seen in patients receiving bevacizumab. These can increase the risk of 90 Y microsphere reflux during treatment. Ø Prior TACE—multiple stenosis and occlusions can result in occult hepatoenteric collaterals and reflux. Prevention § Stop use of bevacizumab or sorafenib 2– 4 weeks prior to planning angiography and transarterial 90 Y treatment or TACE. § Review vascular anatomy including the access vessels on pretreatment diagnostic CT and MR images. § Strongly recommend to use microcatheters and microwire for the procedures Brown DB. Semin Intervent Radiol 2011; 28: 142– 146
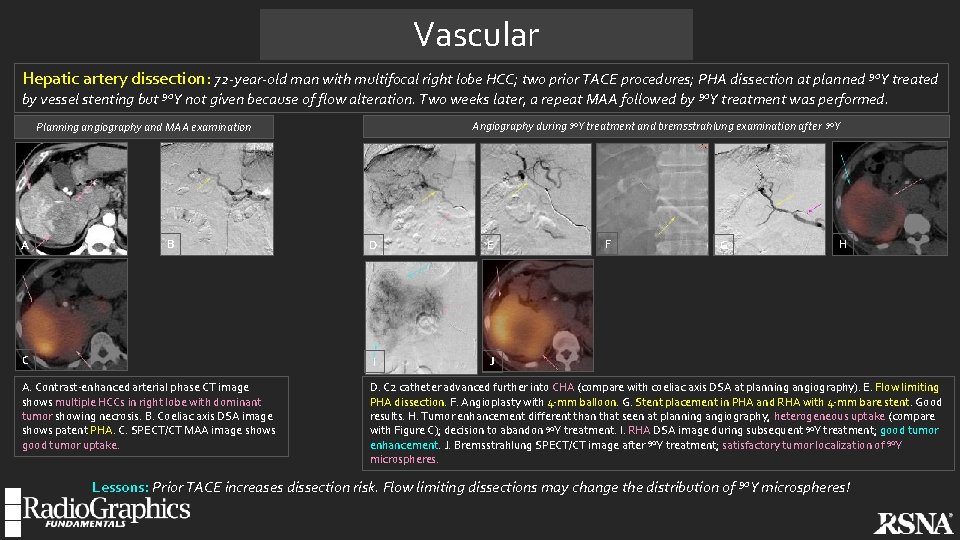
Vascular Hepatic artery dissection: 72 -year-old man with multifocal right lobe HCC; two prior TACE procedures; PHA dissection at planned 90 Y treated by vessel stenting but 90 Y not given because of flow alteration. Two weeks later, a repeat MAA followed by 90 Y treatment was performed. Angiography during 90 Y treatment and bremsstrahlung examination after Planning angiography and MAA examination B F H D E C I J A. Contrast-enhanced arterial phase CT image shows multiple HCCs in right lobe with dominant tumor showing necrosis. B. Coeliac axis DSA image shows patent PHA. C. SPECT/CT MAA image shows good tumor uptake. D. C 2 catheter advanced further into CHA (compare with coeliac axis DSA at planning angiography). E. Flow limiting PHA dissection. F. Angioplasty with 4 -mm balloon. G. Stent placement in PHA and RHA with 4 -mm bare stent. Good results. H. Tumor enhancement different than that seen at planning angiography, heterogeneous uptake (compare with Figure C); decision to abandon 90 Y treatment. I. RHA DSA image during subsequent 90 Y treatment; good tumor enhancement. J. Bremsstrahlung SPECT/CT image after 90 Y treatment; satisfactory tumor localization of 90 Y microspheres. A G 90 Y Lessons: Prior TACE increases dissection risk. Flow limiting dissections may change the distribution of 90 Y microspheres!
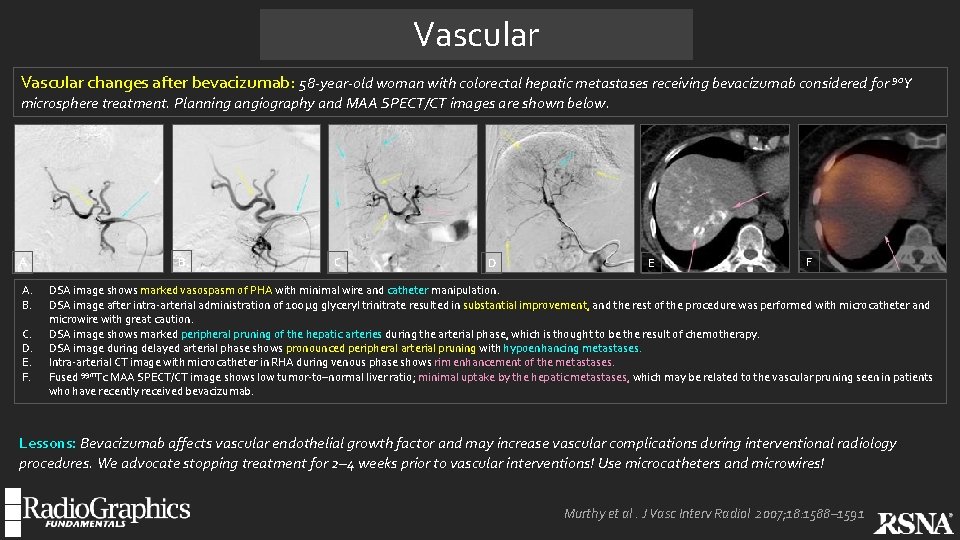
Vascular changes after bevacizumab: 58 -year-old woman with colorectal hepatic metastases receiving bevacizumab considered for 90 Y microsphere treatment. Planning angiography and MAA SPECT/CT images are shown below. A A. B. C. D. E. F. B C D E F DSA image shows marked vasospasm of PHA with minimal wire and catheter manipulation. DSA image after intra-arterial administration of 100 µg glyceryl trinitrate resulted in substantial improvement, and the rest of the procedure was performed with microcatheter and microwire with great caution. DSA image shows marked peripheral pruning of the hepatic arteries during the arterial phase, which is thought to be the result of chemotherapy. DSA image during delayed arterial phase shows pronounced peripheral arterial pruning with hypoenhancing metastases. Intra-arterial CT image with microcatheter in RHA during venous phase shows rim enhancement of the metastases. Fused 99 m. Tc MAA SPECT/CT image shows low tumor-to–normal liver ratio; minimal uptake by the hepatic metastases, which may be related to the vascular pruning seen in patients who have recently received bevacizumab. Lessons: Bevacizumab affects vascular endothelial growth factor and may increase vascular complications during interventional radiology procedures. We advocate stopping treatment for 2– 4 weeks prior to vascular interventions! Use microcatheters and microwires! Murthy et al. J Vasc Interv Radiol 2007; 18: 1588– 1591
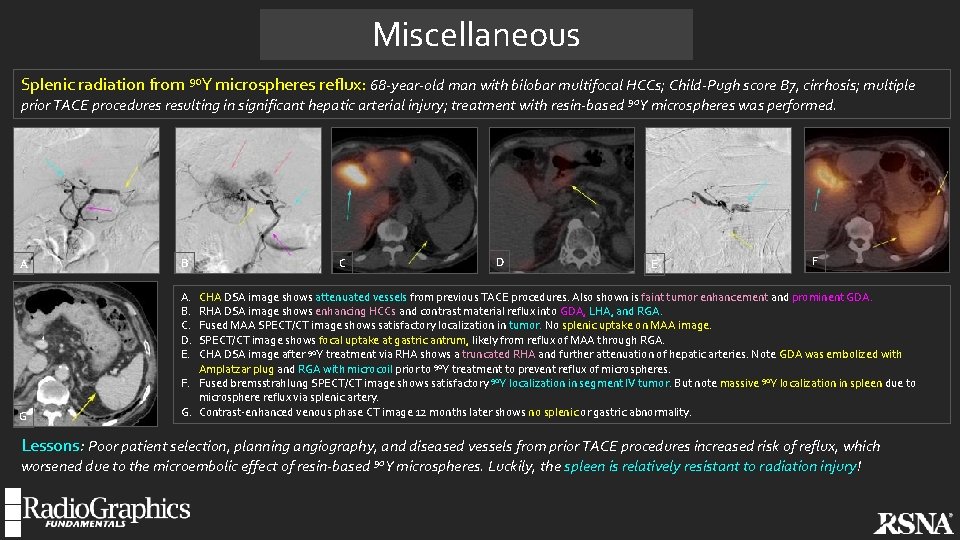
Miscellaneous Splenic radiation from 90 Y microspheres reflux: 68 -year-old man with bilobar multifocal HCCs; Child-Pugh score B 7, cirrhosis; multiple prior TACE procedures resulting in significant hepatic arterial injury; treatment with resin-based 90 Y microspheres was performed. A B C D E F A. B. C. D. E. G CHA DSA image shows attenuated vessels from previous TACE procedures. Also shown is faint tumor enhancement and prominent GDA. RHA DSA image shows enhancing HCCs and contrast material reflux into GDA, LHA, and RGA. Fused MAA SPECT/CT image shows satisfactory localization in tumor. No splenic uptake on MAA image. SPECT/CT image shows focal uptake at gastric antrum, likely from reflux of MAA through RGA. CHA DSA image after 90 Y treatment via RHA shows a truncated RHA and further attenuation of hepatic arteries. Note GDA was embolized with Amplatzar plug and RGA with microcoil prior to 90 Y treatment to prevent reflux of microspheres. F. Fused bremsstrahlung SPECT/CT image shows satisfactory 90 Y localization in segment IV tumor. But note massive 90 Y localization in spleen due to microsphere reflux via splenic artery. G. Contrast-enhanced venous phase CT image 12 months later shows no splenic or gastric abnormality. Lessons: Poor patient selection, planning angiography, and diseased vessels from prior TACE procedures increased risk of reflux, which worsened due to the microembolic effect of resin-based 90 Y microspheres. Luckily, the spleen is relatively resistant to radiation injury!
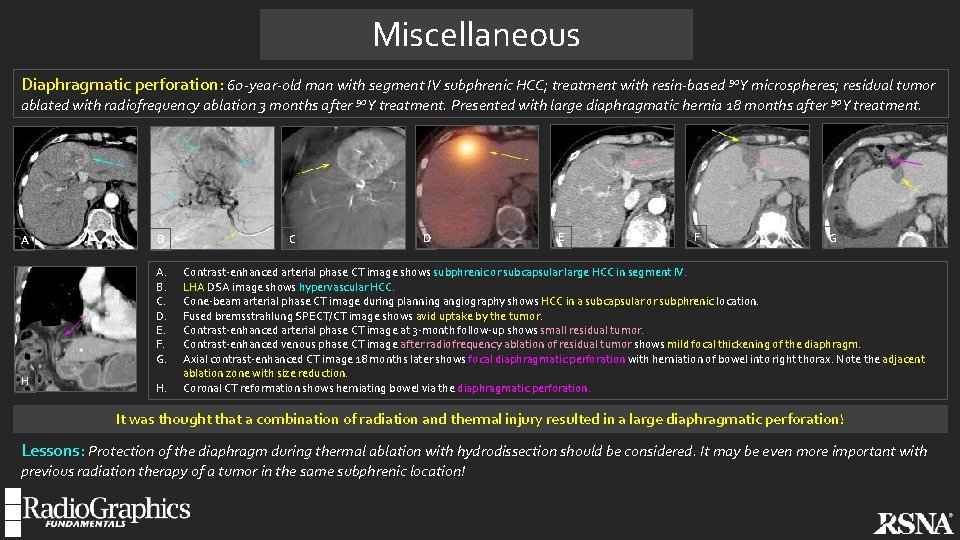
Miscellaneous Diaphragmatic perforation: 60 -year-old man with segment IV subphrenic HCC; treatment with resin-based 90 Y microspheres; residual tumor ablated with radiofrequency ablation 3 months after 90 Y treatment. Presented with large diaphragmatic hernia 18 months after 90 Y treatment. A B A. B. C. D. E. F. G. H H. C D E F G Contrast-enhanced arterial phase CT image shows subphrenic or subcapsular large HCC in segment IV. LHA DSA image shows hypervascular HCC. Cone-beam arterial phase CT image during planning angiography shows HCC in a subcapsular or subphrenic location. Fused bremsstrahlung SPECT/CT image shows avid uptake by the tumor. Contrast-enhanced arterial phase CT image at 3 -month follow-up shows small residual tumor. Contrast-enhanced venous phase CT image after radiofrequency ablation of residual tumor shows mild focal thickening of the diaphragm. Axial contrast-enhanced CT image 18 months later shows focal diaphragmatic perforation with herniation of bowel into right thorax. Note the adjacent ablation zone with size reduction. Coronal CT reformation shows herniating bowel via the diaphragmatic perforation. It was thought that a combination of radiation and thermal injury resulted in a large diaphragmatic perforation! Lessons: Protection of the diaphragm during thermal ablation with hydrodissection should be considered. It may be even more important with previous radiation therapy of a tumor in the same subphrenic location!
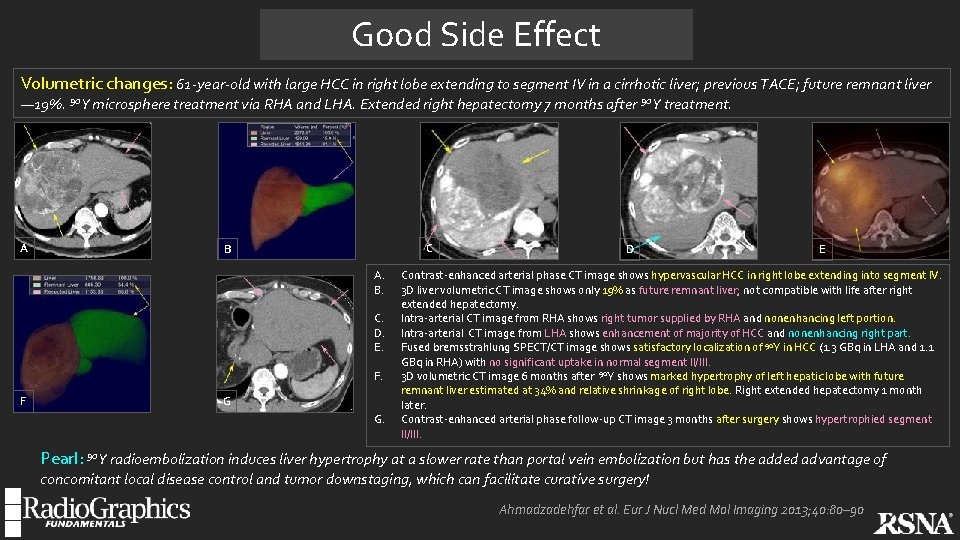
Good Side Effect Volumetric changes: 61 -year-old with large HCC in right lobe extending to segment IV in a cirrhotic liver; previous TACE; future remnant liver — 19%. 90 Y microsphere treatment via RHA and LHA. Extended right hepatectomy 7 months after 90 Y treatment. A C B A. B. C. D. E. F. F G G. D E Contrast-enhanced arterial phase CT image shows hypervascular HCC in right lobe extending into segment IV. 3 D liver volumetric CT image shows only 19% as future remnant liver; not compatible with life after right extended hepatectomy. Intra-arterial CT image from RHA shows right tumor supplied by RHA and nonenhancing left portion. Intra-arterial CT image from LHA shows enhancement of majority of HCC and nonenhancing right part. Fused bremsstrahlung SPECT/CT image shows satisfactory localization of 90 Y in HCC (1. 3 GBq in LHA and 1. 1 GBq in RHA) with no significant uptake in normal segment II/III. 3 D volumetric CT image 6 months after 90 Y shows marked hypertrophy of left hepatic lobe with future remnant liver estimated at 34% and relative shrinkage of right lobe. Right extended hepatectomy 1 month later. Contrast-enhanced arterial phase follow-up CT image 3 months after surgery shows hypertrophied segment II/III. Pearl: 90 Y radioembolization induces liver hypertrophy at a slower rate than portal vein embolization but has the added advantage of concomitant local disease control and tumor downstaging, which can facilitate curative surgery! Ahmadzadehfar et al. Eur J Nucl Med Mol Imaging 2013; 40: 80– 90

Conclusion § Serious adverse events after 90 Y radioembolization are rare. § Complications can be mitigated by appropriate patient selection, attention to proper angiographic techniques, and adoption of standardized dosimetry models. § Discussion at multidisciplinary meetings involving hepatologist, interventional radiologist, nuclear medicine physician, surgeons, and oncologists allows meticulous patient selection. § Review of preprocedural cross-sectional imaging and systematic evaluation at planning angiography is important to prevent complications. § Use of cone-beam CT or intra-arterial CT at planning angiography is paramount to identify and avoid at-risk DSA-occult hepatoenteric communications. § Finally, early identification of unexpected complications and appropriate management is essential to reduce morbidity and mortality.

References • • • Ahmadzadehfar H, Meyer C, Ezziddin S, et al. Hepatic volume changes induced by radioembolization with 90 Y resin microspheres: a single-centre study. Eur J Nucl Med Mol Imaging 2013; 40: 80– 90. Atassi B, Bangash AK, Baharani A, et al. Multimodality imaging following 90 Y radioembolization: a comprehensive review and pictorial essay Radio. Graphics 2008; 28: 81– 99. Bernardini M, Smadja C, Faraggi M, et al. Liver selective internal radiation therapy with (90)Y resin microspheres: comparison between pre-treatment activity calculation methods. Phys Med 2014; 30: 752– 764. Bester L, Salem R. Reduction of arteriohepatovenous shunting by temporary balloon occlusion in patients undergoing radioembolization. J Vasc Interv Radiol 2007; 18: 1310– 1314. Braat MN, van Erpecum KJ, Zonnenberg BA, et al. Radioembolization-induced liver disease: a systematic review. Eur J Gastroenterol Hepatol 2017; 29: 144– 152. Brown DB. Hepatic artery dissection in a patient on bevacizumab resulting in pseudoaneurysm formation. Semin Intervent Radiol 2011; 28: 142– 146. Burgmans MC, Too CW, Kao YH, et al. Computed tomography hepatic arteriography has a hepatic falciform artery detection rate that is much higher than that of digital subtraction angiography and 99 m. Tc-MAA SPECT/CT: implications for planning 90 Y radioembolization? Eur J Radiol 2012; 81: 3979– 3984. Kao YH, Tan AE, Burgmans MC, et al. Image-guided personalized predictive dosimetry by artery-specific SPECT/CT partition modeling for safe and effective 90 Y radioembolization. J Nucl Med 2012; 53: 1– 10. Lam MG, Banerjee A, Louie JD, Sze DY. Splenomegaly-associated thrombocytopenia after hepatic yttrium-90 radioembolization. Cardiovasc Intervent Radiol 2014; 37: 1009– 1017. Leong QM, Lai HK, Lo RG, et al. Radiation dermatitis following radioembolization for hepatocellular carcinoma: a case for prophylactic embolization of a patent falciform artery. J Vasc Interv Radiol 2009; 20: 833– 836. Leung TW, Lau WY, Ho SK, et al. Radiation pneumonitis after selective internal radiation treatment with intraarterial 90 yttrium-microspheres for inoperable hepatic tumors. Int J Radiat Oncol Biol Phys 1995; 33: 919– 924. Meyer C, Pieper CC, Ezziddin S, et al. Feasibility of temporary protective embolization of normal liver tissue using degradable starch microspheres during radioembolization of liver tumours. Eur J Nucl Mol Imaging 2014; 41: 231– 237.

References • • • Murthy R, Nunez R, Szklaruk J, et al. Yttrium-90 microsphere therapy for hepatic malignancy: devices, indications, technical considerations, and potential complications. Radio. Graphics 2005; 25: S 41–S 55. Murthy R, Brown DB, Salem R, et al. Gastrointestinal complications associated with hepatic arterial Yttrium-90 microsphere therapy. J Vasc Interv Radiol 2007; 18: 553– 561. Murthy R, Eng C, Krishnan S, et al. Hepatic yttrium-90 radioembolotherapy in metastatic colorectal cancer treated with cetuximab or bevacizumab. J Vasc Interv Radiol 2007; 18: 1588– 1591. Paprottka PM, Jakobs TF, Reiser MF, Hoffmann RT. Practical vascular anatomy in the preparation of radioembolization. Cardiovasc Intervent Radiol 2012; 35: 454– 462. Riaz A, Awais R, Salem R. Side effects of yttrium-90 radioembolization. Front Oncol 2014; 4: 198. Riaz A, Lewandowski RJ, Kulik LM, et al. Complications following radioembolization with yttrium-90 microspheres: a comprehensive literature review. J Vasc Interv Radiol 2009; 20: 1121– 1130. Sabet A, Ahmadzadehfar H, Muckle M, et al. Significance of oral administration of sodium perchlorate in planning liver-directed radioembolization. J Nucl Med 2011; 52: 1063– 1067. Salem R, Lewandowski RJ, Sato KT, et al. Technical aspects of radioembolization with 90 Y microspheres. Tech Vasc Interv Radiol 2007; 10: 12– 29. Sangro B, Gil-Alzugaray B, Rodriguez J, et al. Liver disease induced by radioembolization of liver tumors: description and possible risk factors. Cancer 2008; 112: 1538– 1546 Venkatanarasimha N , Gogna A, Tong KTA, et al. Radioembolisation of hepatocellular carcinoma: a primer. Clin Radiol 2017; 30: 752– 764 Yu N, Srinivas SM, Difilippo FP, et al. Lung dose calculation with SPECT/CT for ⁹⁰Yittrium radioembolization of liver cancer. Int J Radiat Oncol Bio Phys 2013; 85: 834– 910

Suggested Readings • Bilbao JL, Iezzi R, Goldberg SN, Sami A, Akhan O, Giuliante F, Pompili M, Crocetti L, Malagari K et al. The ten commandments of hepatic radioembolization: expert discussion and report from mediterranean interventional onocolgy (MIOLive) congress 2017. Eur Review for Med Pharmacol Sci 2017; 21: 4014– 4021. • Gaba RC. Planning arteriography for yttrium-90 microsphere radioembolisation. Semin Intervent Radiol 2015; 32: 428– 38. • Kao YH, Steinberg JD, Tay Y-S, Lim GKY, Yan J, Townsend DW, et al. Post-radioembolization yttrium-90 PET/CT. part 1: diagnostic reporting. EJNMMI Res 2013; 3: 56 • Kao Y-H, Steinberg JD, Tay Y-S, Lim GK, Yan J, Townsend DW, et al. Post-radioembolization yttrium-90 PET/CT. part 2: dose-response and tumor predictive dosimetry for resin microspheres. EJNMMI Res 2013; 3: 57. • Kuo JC, Tazbirkova A, Allen R, Kosmider S, Gibbs P, Yip D. Serious hepatic complications of selective internal radiaton therapy with yttrium-90 microsphere radioembolization for unresectable liver tumors. Asia Pac J Clin Oncol 2014; 10: 266– 272. • Paprottka PM, Jakobs TF, Reiser MF, Hoffmann RT. Practical vascular anatomy in the preparation of radioembolization. Cardiovasc Intervent Radiol 2012; 35: 454– 62. • Paprottka PM, Schmidt GP, Trumm CG, Hoffmann RT, Reiser MF, Jakobs TF. Changes in normal liver and spleen volume after radioembolization with (90)Y-resin microspheres in metastatic breast cancer patients: findings and clinical significance. Cardiovasc Intervent Radiol 2011; 34: 964– 972. • Peterson JL, Vallow LA, Johnson DW, Heckman MG, Diehl NN, Smith AA et al. Complications after 90 Y microsphere radioembolization for unresectable hepatic tumors: an evaluation of 112 patients. Brachytherapy 2013; 12: 573 -579. • Piana PM, Gonsalves CF, Sato T, Anne PR, Mc. Cann JW, Bar Ad V et al. Toxicities after radioembolization with yttrium-90 SIR-spheres: incidence and contributing risk factors at a single center. J Vasc Interv Radiol 2011; 22: 1373 -1379. • Riaz A, Lewandowski RJ, Kulik LM, Mulcahy MF, Sato KT, Ryu RK, Omary RA, Salem R. Complications following radioembolization with yttrium-90 microspheres: a comprehensive literature review. J Vasc Interv Radiol 2009; 20: 1121– 1130. • Sag AA, Savin MA, Lal NR, Mehta RR. Yttrium-90 radioembolization of malignant tumors of the liver: gallbladder effects. AJR Am J Roentgenol 2014; 202: 1130– 1135.
- Slides: 45