Complexes and Coloured Ions Do Now How do


















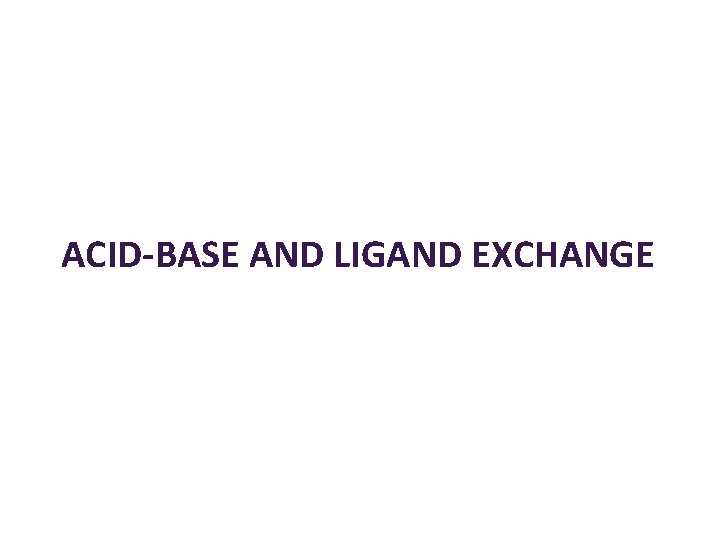
![Acid-Base Reaction [Cu(H 2 O)6]2+ + 2 OH- ? [Cu(H 2 O)6]2+ + 2 Acid-Base Reaction [Cu(H 2 O)6]2+ + 2 OH- ? [Cu(H 2 O)6]2+ + 2](https://slidetodoc.com/presentation_image_h2/73747ffb0ccd0137fdcd63c00f9646ac/image-24.jpg)


- Slides: 29

Complexes and Coloured Ions Do Now: How do we see colour? What do we mean by complimentary colour?

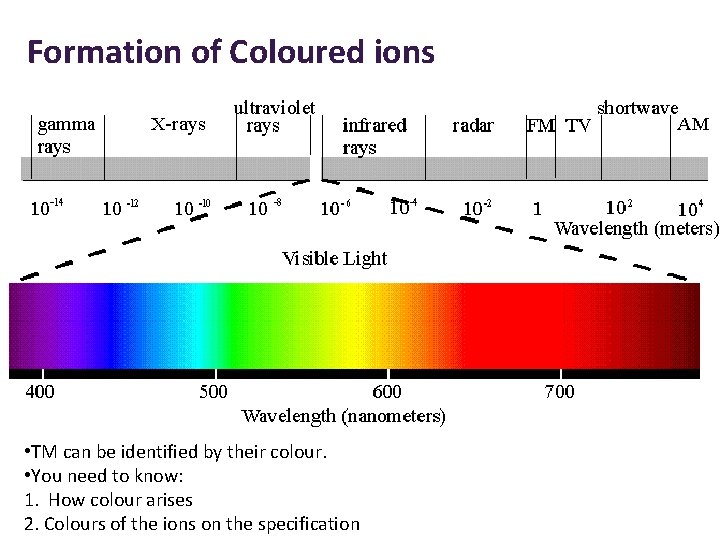
Formation of Coloured ions • TM can be identified by their colour. • You need to know: 1. How colour arises 2. Colours of the ions on the specification

What is white light? • Traditional answer – made up of the 7 colours of the rainbow • However… • Seven is just an arbitrary number • Made up of infinite number of colours!

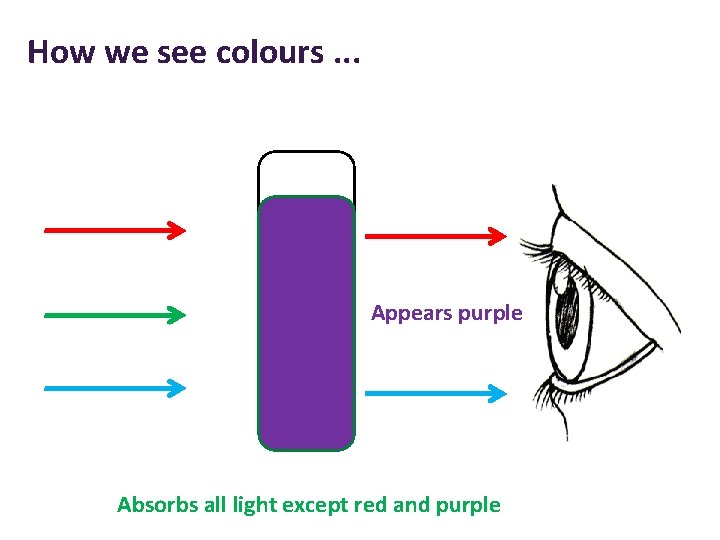
How we see colours. . . Appears purple Absorbs all light except red and purple


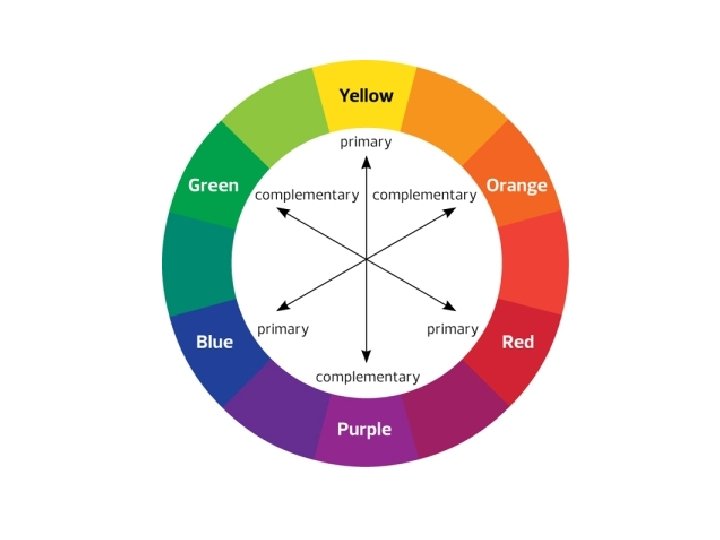
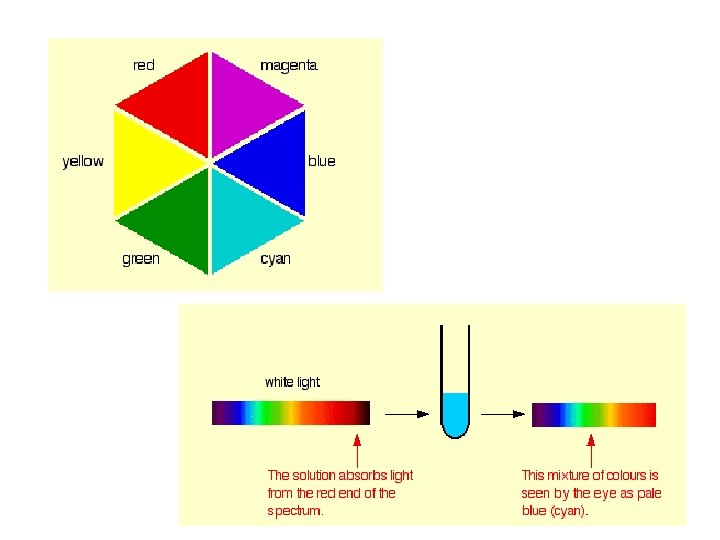
Complimentary Colours • Shown opposite each other • When white light passes through a solution containing a transition metal complex, some of the wavelengths of light are absorbed by the complex. • The light emerging will therefore contain proportionally more of the complimentary colour. • E. g. if a complex absorbs red light, the light emerging will look blue or green.


Zinc Sulfate (aq) vs. Copper (II) Sulfate (aq)

3 d energy levels

Why are Transition Metal Complexes Coloured? • TM have part filled d-orbitals. • So the e- can move from one d-orbital to another. • In a TM ATOM on its own (isolated), all of the d-orbitals are of EXACTLY the same energy • BUT in a COMPOUND, the presence of ligands makes the d-orbitals have slightly different energies. • When an e- moves from one d-orbital to another of a higher energy level, they often ABSORB energy. • This is known as ‘promotion’ or ‘excitation’ • The amount of energy absorbed depends on the difference in the energy levels • The bigger the energy, difference, the more energy the electron absorbs • The amount of energy absorbed is directly proportional to the frequency of the absorbed light and inversely proportional to the wavelength of light • For Copper 2+, the small difference means a low frequency and a high wavelength • This corresponds to red on the visible light spectrum • So blue light is transmitted

Electronic Transitions • e- occupy particular energy levels in atoms • When an atom ABSORBS energy, an e- can be promoted from its normal ground state to a higher energy level • The e- is then in an excited state • Such changes from one energy level to another = electronic transitions

Questions • Look at the colour wheel to help answer the following questions: 1) Fe 3+ (aq) ions look yellow. a) What is the complimentary colour? How would you describe the difference in energy levels? Describe the frequency and wavelength of the radiation absorbed b) c)







DIFFERENT TYPES OF REACTIONS

What do we mean by the following? • Redox reaction • Acid-Base • Ligand reaction exchange • Coordination number change

CHANGE IN OXIDATION NUMBER

Formation of Coloured ions The size of the energy gap between the d-orbitals, and so the colour is affected by changes in: 1) the oxidation state [Fe(H 2 O)6]3+ [Fe(H 2 O)6]2+ Yellow/Brown Pale Green
ACID-BASE AND LIGAND EXCHANGE
![AcidBase Reaction CuH 2 O62 2 OH CuH 2 O62 2 Acid-Base Reaction [Cu(H 2 O)6]2+ + 2 OH- ? [Cu(H 2 O)6]2+ + 2](https://slidetodoc.com/presentation_image_h2/73747ffb0ccd0137fdcd63c00f9646ac/image-24.jpg)
Acid-Base Reaction [Cu(H 2 O)6]2+ + 2 OH- ? [Cu(H 2 O)6]2+ + 2 NH 3 ? [Cu(H 2 O)4(OH)2] + 4 NH 3


CHANGE IN COORDINATION NUMBER

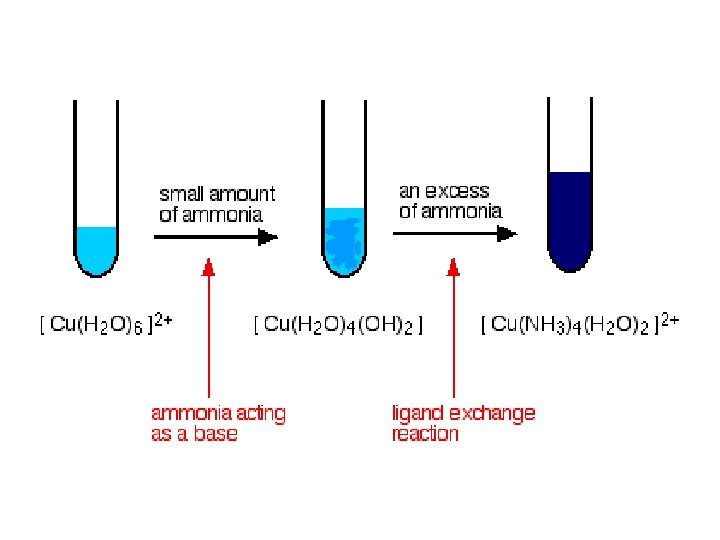
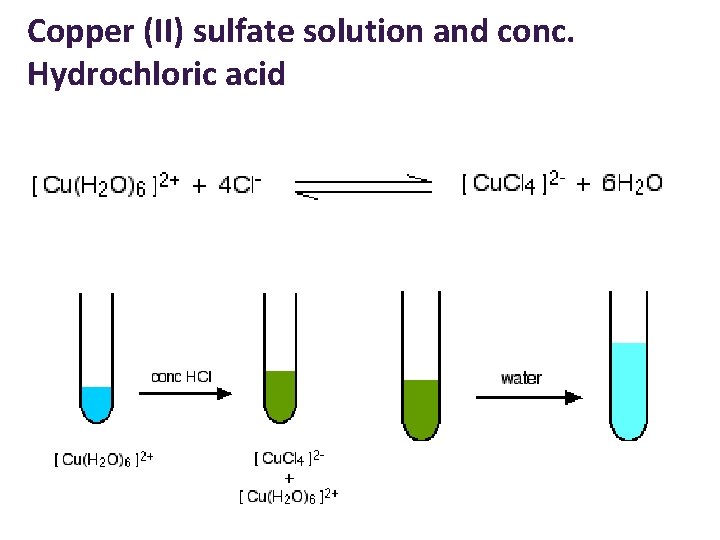
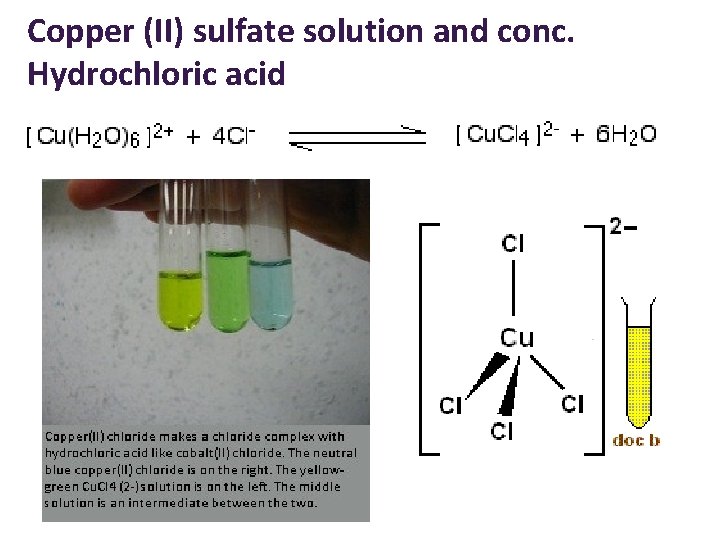
Copper (II) sulfate solution and conc. Hydrochloric acid

Copper (II) sulfate solution and conc. Hydrochloric acid

EXAM QUESTIONS