Complement and Haemolytic Uraemic Syndrome ESPN 2008 Dr















- Slides: 20

Complement and Haemolytic Uraemic Syndrome – ESPN 2008 Dr Heather Maxwell Renal Unit RHSC, Glasgow

Haemolytic Uraemic Syndrome • Most common cause of acute renal failure in children in UK • High incidence in Scotland • Mostly D+HUS, usually E. Coli O 157 • Very occasional patients with D-HUS or atypical HUS • More known about atypical HUS • Implications for treatment

Atypical HUS • Infections other than diarrhoeal illnesses – Pneumococcal HUS – HIV, Influenza A • • • Drugs Pregnancy Familial Complement Abnormalities ADAMTS 13 Deficiency Disordered Cobalamin Metabolism

Atypical HUS • Uncommon • Up to 50% or more may have mutations in genes of complement components and regulators • Poor prognosis • Treatment options depending on the mutation • Early intervention crucial • Implications for recurrence post transplantation • Up to 40% may present with diarrhoea

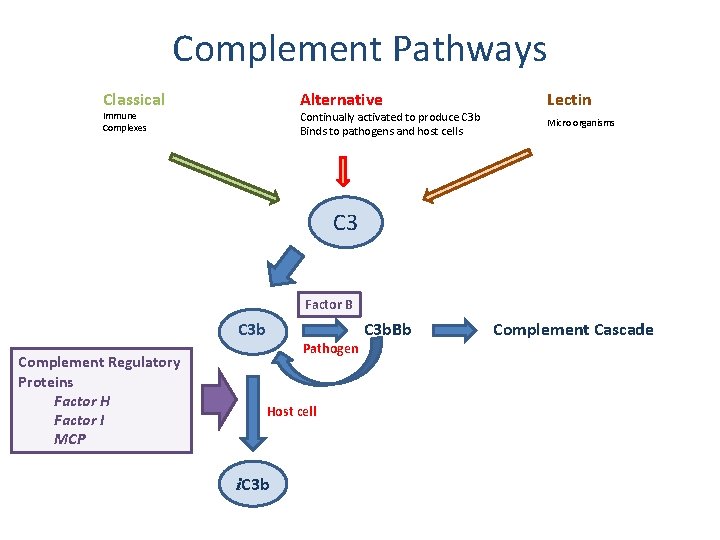
Complement Pathways Classical Alternative Continually activated to produce C 3 b Binds to pathogens and host cells Immune Complexes Lectin Micro organisms C 3 Factor B C 3 b Complement Regulatory Proteins Factor H Factor I MCP Pathogen Host cell i. C 3 b. Bb Complement Cascade

Complement Abnormalities in HUS • • • Factor H MCP Factor I C 3 Factor B – Endothelial cell damage – Platelet aggregation – Thrombus formation

French a. HUS Registry Data (n=90) Complement Abnormality Frequency Factor H 19% Most severe 60% ESRF or death within 1 year MCP 14% Often have a relapsing course but with time 30% go into ESRF Factor I 9% 50 -70% ESRF at 5 years C 3 7% Factor B 1% Factor H Antibodies 11% Combined Mutations 8% None 39% Rare 30% ESRF by 5 years

Factor H • • Most important regulator of the alternative pathway Synthesized by the liver Circulating glycoprotein 150 k. Da Binds to cell surface Prevents C 3 convertase formation and accelerates C 3 b decay Co-factor for Factor I Gene on chromosome 1 Gene - multiple short consensus repeats (SCR)

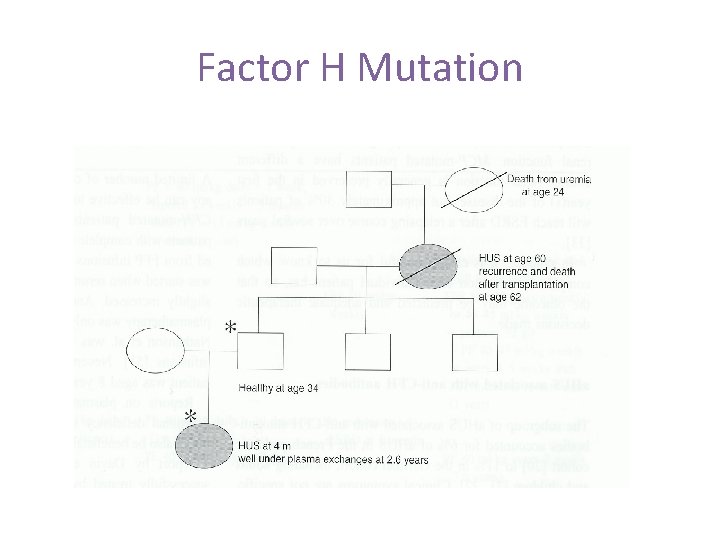
Factor H Mutations First reported 1981 Abnormal Factor H Deficiency of Factor H Majority heterozygous C 3 either normal or low >100 mutations reported Mutations vary between countries • 50% familial penetrance • Factor H autoantibodies • • Clinical Course • Reported in 19 -24% a. HUS • Early onset 3/12 to 1 year • 60% died or had ESRF within 1 year • +/- family history Treatment • Homozygous – plasma infusions • Heterozygous – Plasma Ex + FFP • Recombinant Factor H • Kidney +/- liver transplant • Complement Inhibitors

Factor H Mutation

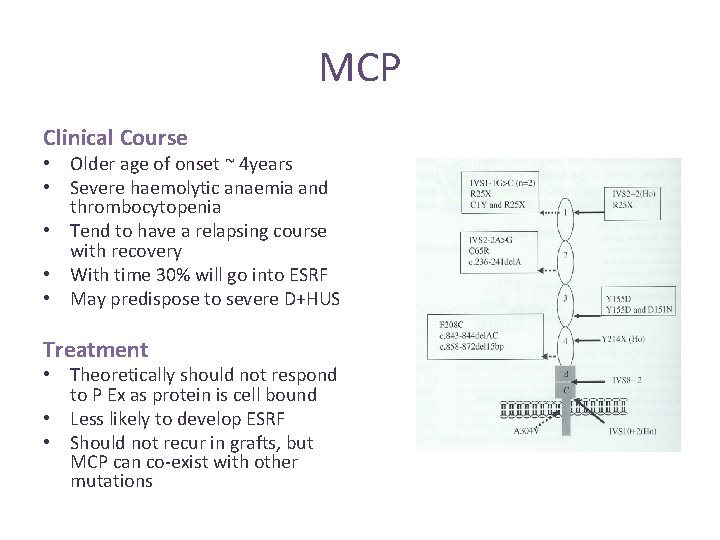
Membrane Co-Factor Protein (CD 46) Widely expressed transmembrane glycoprotein Cofactor for the cleavage of C 3 b by Factor I >20 mutations have been described 80% result in a reduction in MCP expression Results in inadequate control of complement activation on endothelial cells • MCP mutations can co-exist with other complement mutations • Less likely to develop ESRF than FH mutations • • •

MCP Clinical Course • Older age of onset ~ 4 years • Severe haemolytic anaemia and thrombocytopenia • Tend to have a relapsing course with recovery • With time 30% will go into ESRF • May predispose to severe D+HUS Treatment • Theoretically should not respond to P Ex as protein is cell bound • Less likely to develop ESRF • Should not recur in grafts, but MCP can co-exist with other mutations

Factor I • Is an enzyme – 2 chain serine protease • Gene on chromosome 4 • Soluble regulator of C 3 b requiring the co-factors FH and MCP • < 20 mutations described • Usually quantitative deficiency of Factor I Clinical Course • Young age of onset (median 2 months) • Few reports of details of treatment • 88% recurrence risk post transplantation

Complement Pathways Classical Alternative Continually activated to produce C 3 b Binds to pathogens and host cells Immune Complexes Lectin Micro organisms C 3 Factor B C 3 b Complement Regulatory Proteins Factor H Factor I MCP Pathogen Host cell i. C 3 b. Bb Complement Cascade

Atypical HUS Factor B Mutations C 3 Mutations • Rare (0 -3%) • Low C 3 levels • Gain of function • Few reports regarding mutation described clinical course, treatment or prognosis • Increases the stability of C 3 b. Bb convertase • Low C 3 • Children are immunodeficient • Factor B levels normal

Atypical HUS Anti Factor H Antibodies • 6 -11% in registry data • Presents between 3 and 14 years of age • C 3 slightly low • Factor H levels normal • Homozygous gene deletions Treatment • Plasma exchange • Immunosuppression (steroids and rituximab have been tried) No Complement Mutations Detected • Age range 25 days to 15 years • 32% in ESRF at 1 and 5 years • Some have a relapsing course

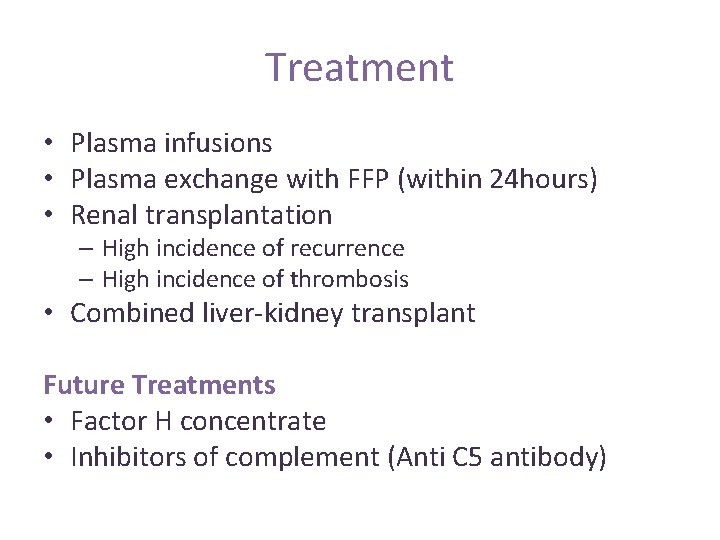
Treatment • Plasma infusions • Plasma exchange with FFP (within 24 hours) • Renal transplantation – High incidence of recurrence – High incidence of thrombosis • Combined liver-kidney transplant Future Treatments • Factor H concentrate • Inhibitors of complement (Anti C 5 antibody)

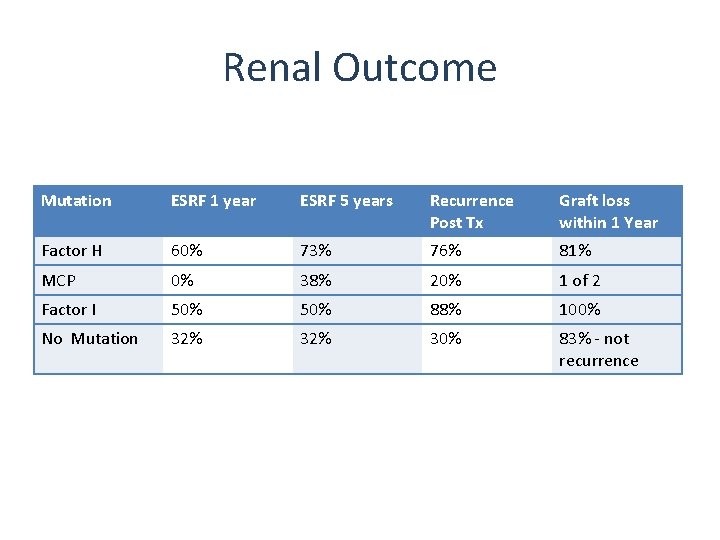
Renal Outcome Mutation ESRF 1 year ESRF 5 years Recurrence Post Tx Graft loss within 1 Year Factor H 60% 73% 76% 81% MCP 0% 38% 20% 1 of 2 Factor I 50% 88% 100% No Mutation 32% 30% 83% - not recurrence

Atypical HUS Who is atypical? • No proceeding diarrhoea • Non-bloody diarrhoea • Known family history of HUS • Children under 3 months old • Relapsing course • High index of suspicion / absence of typical features • Evidence of VTEC • Refer early

Investigation Atypical HUS Quantitative +/- Qualitative Activity • C 3 • C 4 • Factor H • Factor I • Factor B • Membrane expression of MCP • Factor H Antibodies Genetic testing • Factor H • Factor I • MCP Non-Complement Testing • ADAMTS 13 • Cobalamine metabolism – Homocysteine – Methylmalonic acid • Streptococcal infection