Competition in Alkali Metal Ion Phenol Clusters Disrupting



- Slides: 15

Competition in Alkali Metal Ion – Phenol Clusters: Disrupting the Intermolecular Hydrogen Bonds with Cation-p Interactions Timothy D. Vaden and James M. Lisy University of Illinois at Urbana-Champaign

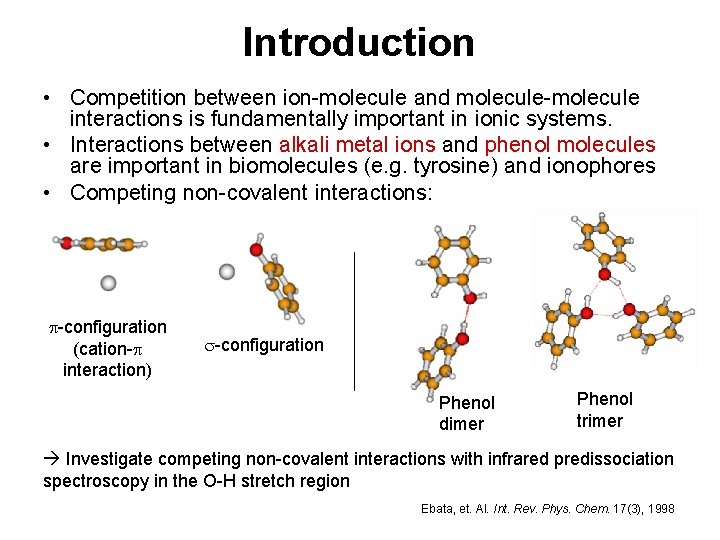
Introduction • Competition between ion-molecule and molecule-molecule interactions is fundamentally important in ionic systems. • Interactions between alkali metal ions and phenol molecules are important in biomolecules (e. g. tyrosine) and ionophores • Competing non-covalent interactions: p-configuration (cation-p interaction) s-configuration Phenol dimer Phenol trimer Investigate competing non-covalent interactions with infrared predissociation spectroscopy in the O-H stretch region Ebata, et. Al. Int. Rev. Phys. Chem. 17(3), 1998

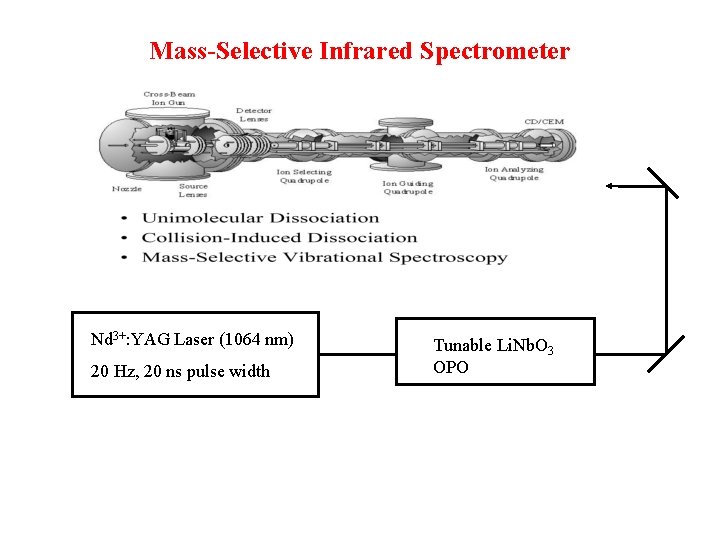
Mass-Selective Infrared Spectrometer Nd 3+: YAG Laser (1064 nm) 20 Hz, 20 ns pulse width Tunable Li. Nb. O 3 OPO

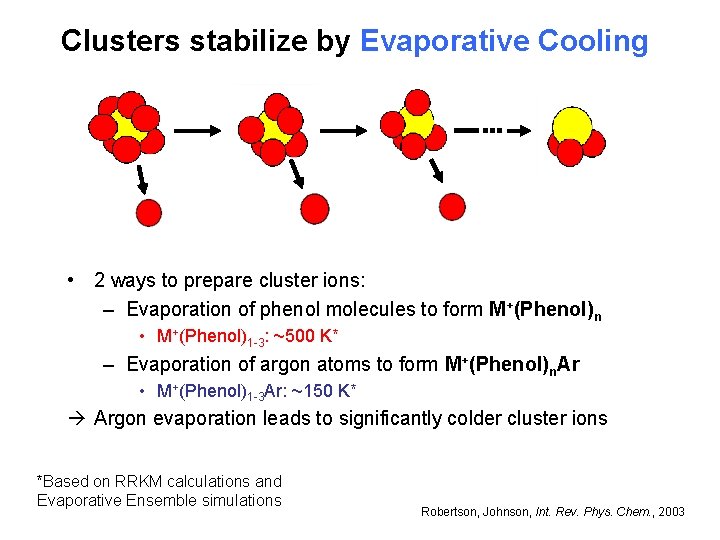
Clusters stabilize by Evaporative Cooling • 2 ways to prepare cluster ions: – Evaporation of phenol molecules to form M+(Phenol)n • M+(Phenol)1 -3: ~500 K* – Evaporation of argon atoms to form M+(Phenol)n. Ar • M+(Phenol)1 -3 Ar: ~150 K* Argon evaporation leads to significantly colder cluster ions *Based on RRKM calculations and Evaporative Ensemble simulations Robertson, Johnson, Int. Rev. Phys. Chem. , 2003

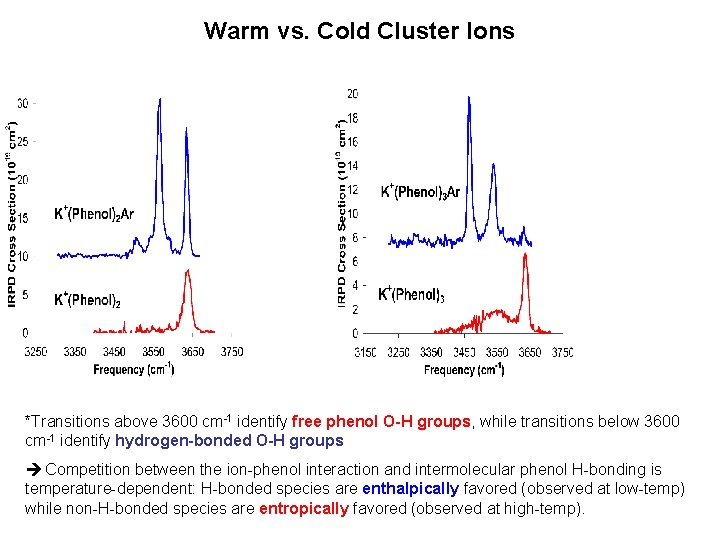
Warm vs. Cold Cluster Ions *Transitions above 3600 cm-1 identify free phenol O-H groups, while transitions below 3600 cm-1 identify hydrogen-bonded O-H groups Competition between the ion-phenol interaction and intermolecular phenol H-bonding is temperature-dependent: H-bonded species are enthalpically favored (observed at low-temp) while non-H-bonded species are entropically favored (observed at high-temp).

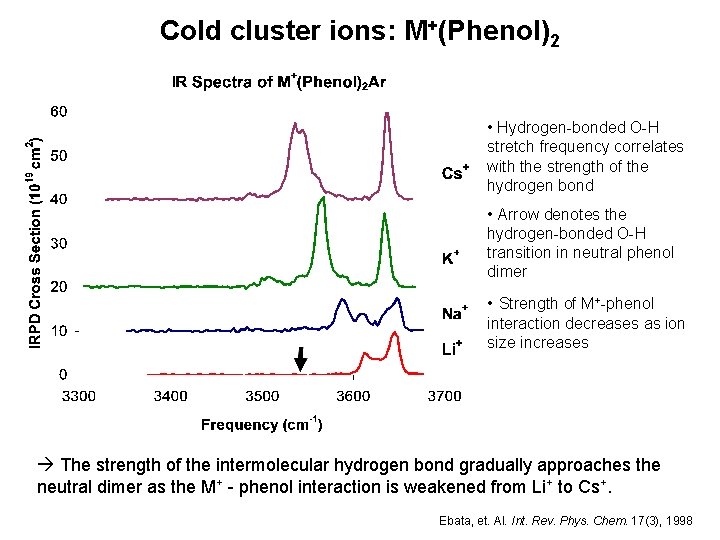
Cold cluster ions: M+(Phenol)2 • Hydrogen-bonded O-H stretch frequency correlates with the strength of the hydrogen bond • Arrow denotes the hydrogen-bonded O-H transition in neutral phenol dimer • Strength of M+-phenol interaction decreases as ion size increases The strength of the intermolecular hydrogen bond gradually approaches the neutral dimer as the M+ - phenol interaction is weakened from Li+ to Cs+. Ebata, et. Al. Int. Rev. Phys. Chem. 17(3), 1998

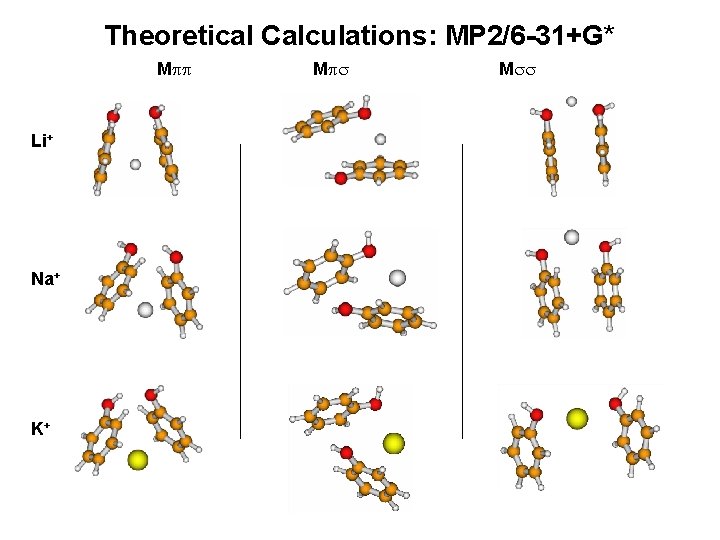
Theoretical Calculations: MP 2/6 -31+G* Mpp Li+ Na+ K+ Mps Mss

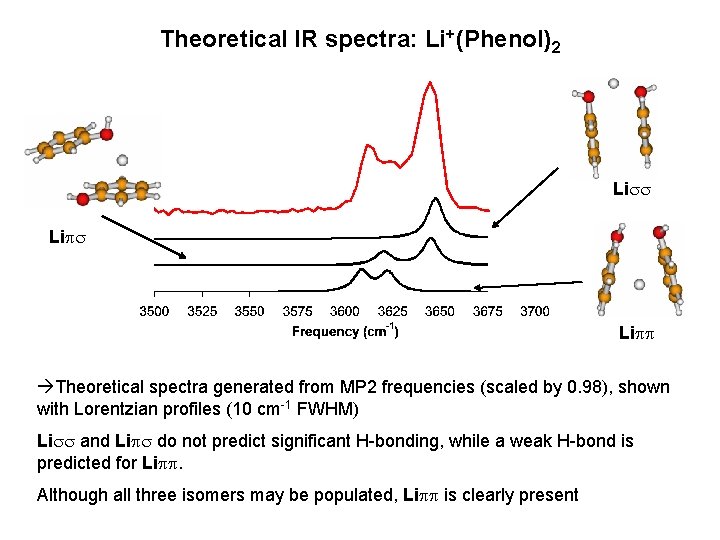
Theoretical IR spectra: Li+(Phenol)2 Liss Lipp Theoretical spectra generated from MP 2 frequencies (scaled by 0. 98), shown with Lorentzian profiles (10 cm-1 FWHM) Liss and Lips do not predict significant H-bonding, while a weak H-bond is predicted for Lipp. Although all three isomers may be populated, Lipp is clearly present

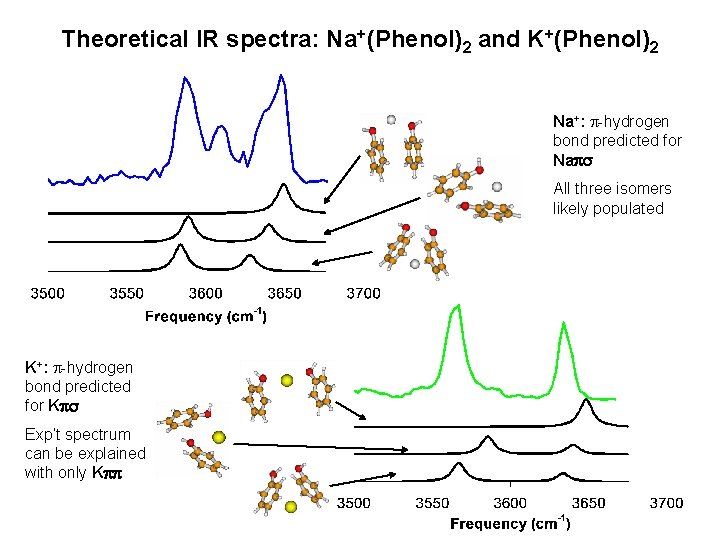
Theoretical IR spectra: Na+(Phenol)2 and K+(Phenol)2 Na+: p-hydrogen bond predicted for Naps All three isomers likely populated K+: p-hydrogen bond predicted for Kps Exp’t spectrum can be explained with only Kpp

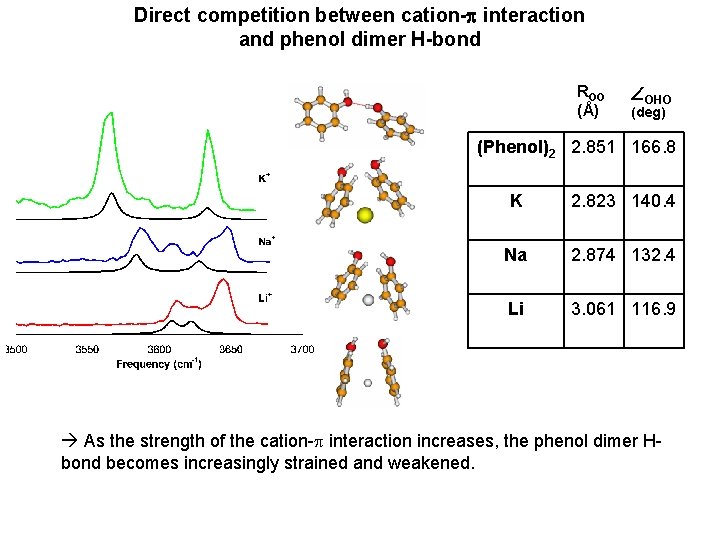
Direct competition between cation-p interaction and phenol dimer H-bond ROO (Å) OHO (deg) (Phenol)2 2. 851 166. 8 K 2. 823 140. 4 Na 2. 874 132. 4 Li 3. 061 116. 9 As the strength of the cation-p interaction increases, the phenol dimer Hbond becomes increasingly strained and weakened.

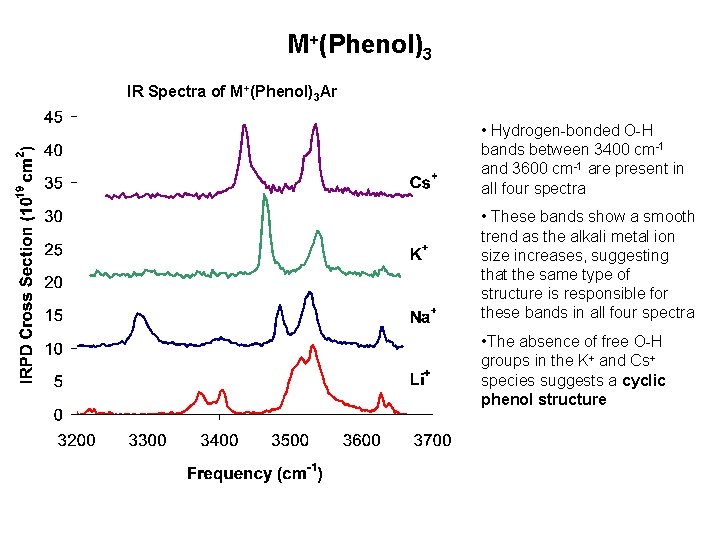
M+(Phenol)3 IR Spectra of M+(Phenol)3 Ar • Hydrogen-bonded O-H bands between 3400 cm-1 and 3600 cm-1 are present in all four spectra • These bands show a smooth trend as the alkali metal ion size increases, suggesting that the same type of structure is responsible for these bands in all four spectra • The absence of free O-H groups in the K+ and Cs+ species suggests a cyclic phenol structure

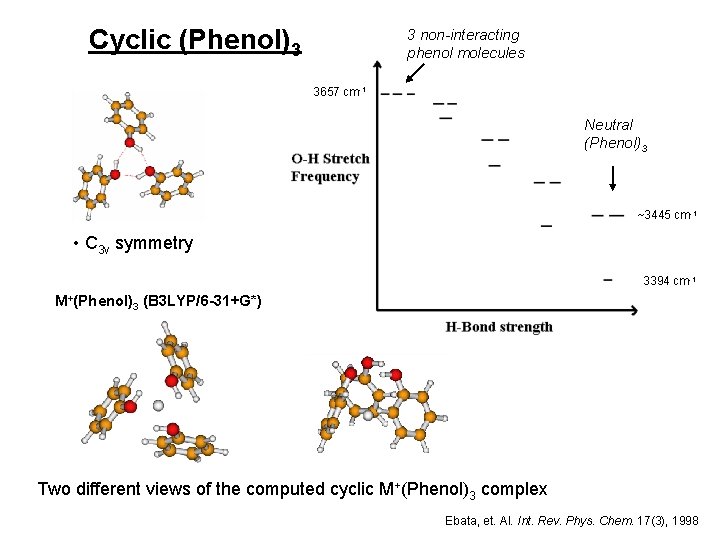
Cyclic (Phenol)3 3 non-interacting phenol molecules 3657 cm-1 Neutral (Phenol)3 ~3445 cm-1 • C 3 v symmetry 3394 cm-1 M+(Phenol)3 (B 3 LYP/6 -31+G*) Two different views of the computed cyclic M+(Phenol)3 complex Ebata, et. Al. Int. Rev. Phys. Chem. 17(3), 1998

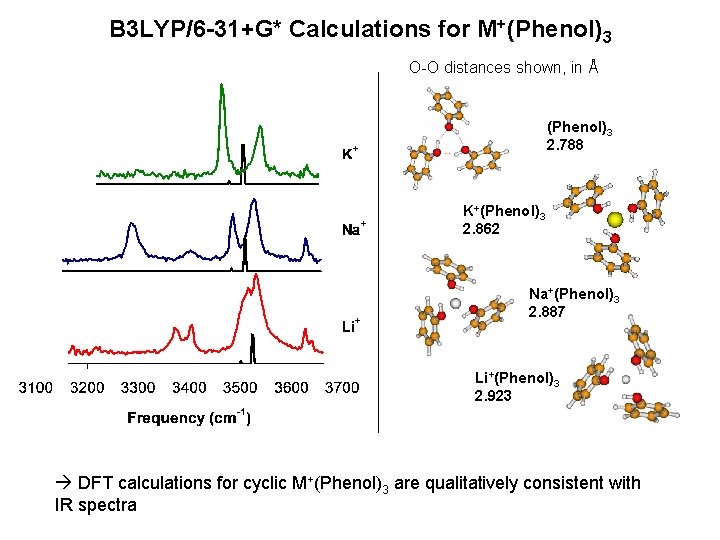
B 3 LYP/6 -31+G* Calculations for M+(Phenol)3 O-O distances shown, in Å (Phenol)3 2. 788 K+(Phenol)3 2. 862 Na+(Phenol)3 2. 887 Li+(Phenol)3 2. 923 DFT calculations for cyclic M+(Phenol)3 are qualitatively consistent with IR spectra

Conclusions • Cation-p interaction competes against the intermolecular phenol H-bonding – M+(Phenol)2 : linear phenol dimer – M+(Phenol)3 : cyclic phenol trimer • Competition between ion-phenol interactions and intermolecular phenol H-bonding is temperaturedependent – Phenol H-bonds remain intact at low temperatures but are completely ruptured at higher temperatures

Acknowledgements • • NSF CHE-0072178 and CHE-0415859 Petroleum Research Fund Merck Research Laboratories Fellowship Lisy Group Members – James Lisy – Dorothy Miller – Jason Rodriguez – Matthew Ackerman – Natalie Rebacz