Competence isolation and switching in Bacillus subtilis Gavin

Competence isolation and switching in Bacillus subtilis Gavin Price Arkin Group, 8/13/06 Gavin Price Arkin Lab 8/13/06
Competence Background • Competence refers to the ability to bind, internalize, and recombine extracellular DNA • Several usage hypotheses • Food • Repair template • Gene transfer • Uptake promiscuous or homology based, depending on species • Some known competent species: • • • Streptococcus pneumoniae (pathogen) Haemophilius influenzae (pathogen) Neisseria gonorreae (pathogen) Staphyloccocus aureus (pathogen) Chlorobium limicola (photolithotroph) Thiobacillus thioparus (chemolithotroph) Psuedomonas stutzeri (heterotroph) Thermus thermophilus (heterotroph) Methylobacterium organophilum (methylotroph) Methanococcus voltae (archaebacteria) Bacillus subtilis (heterotroph) Gavin Price Arkin Lab The DNA binding and transport assembly in B. subtilis I. Chen, D. Dubnau, Nat Rev Microbiol 2, 241 (Mar, 2004). 8/13/06
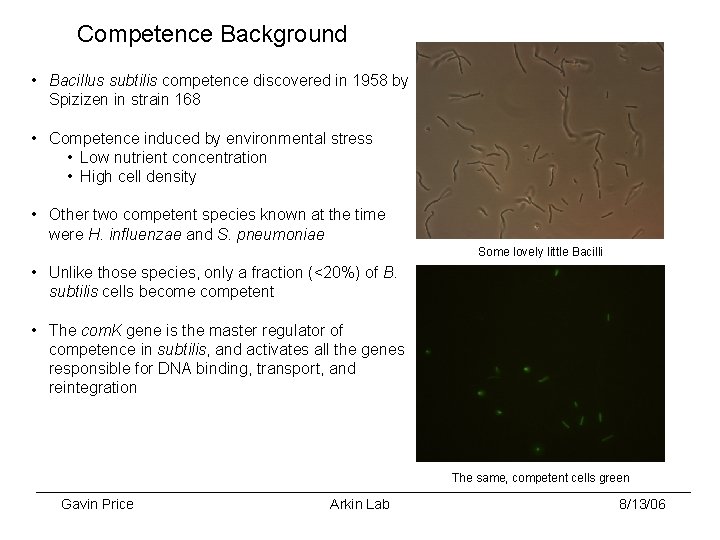
Competence Background • Bacillus subtilis competence discovered in 1958 by Spizizen in strain 168 • Competence induced by environmental stress • Low nutrient concentration • High cell density • Other two competent species known at the time were H. influenzae and S. pneumoniae Some lovely little Bacilli • Unlike those species, only a fraction (<20%) of B. subtilis cells become competent • The com. K gene is the master regulator of competence in subtilis, and activates all the genes responsible for DNA binding, transport, and reintegration The same, competent cells green Gavin Price Arkin Lab 8/13/06

Specific Aim #1 • Develop means to separate heterogeneous B. subtilis 168 populations (competent vs. non-competent and sporulating vs. non -sporulating) • Engineer cells to display streptavidin in the cell wall during entry to competence or sporulation • Immobilize competent/sporulating cells on biotin coated plates or in columns • Elute with high concentration streptavidin/biotin Gavin Price Arkin Lab 8/13/06
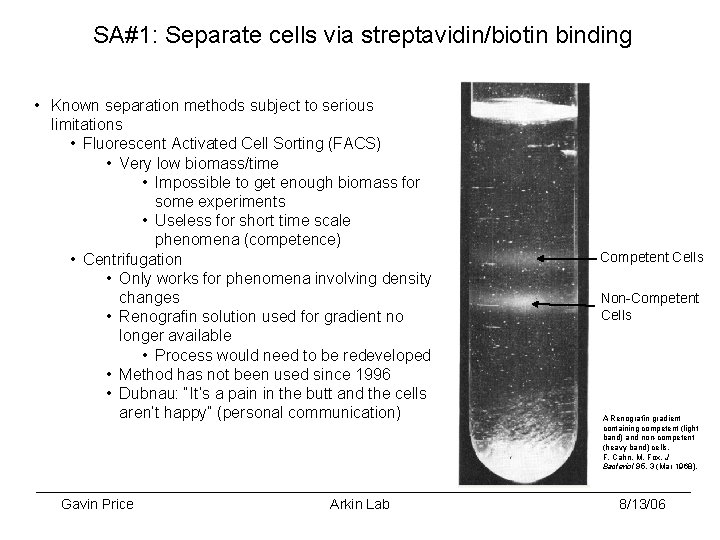
SA#1: Separate cells via streptavidin/biotin binding • Known separation methods subject to serious limitations • Fluorescent Activated Cell Sorting (FACS) • Very low biomass/time • Impossible to get enough biomass for some experiments • Useless for short time scale phenomena (competence) • Centrifugation • Only works for phenomena involving density changes • Renografin solution used for gradient no longer available • Process would need to be redeveloped • Method has not been used since 1996 • Dubnau: “It’s a pain in the butt and the cells aren’t happy” (personal communication) Gavin Price Arkin Lab Competent Cells Non-Competent Cells A Renografin gradient containing competent (light band) and non-competent (heavy band) cells. F. Cahn, M. Fox, J Bacteriol 95, 3 (Mar 1968). 8/13/06
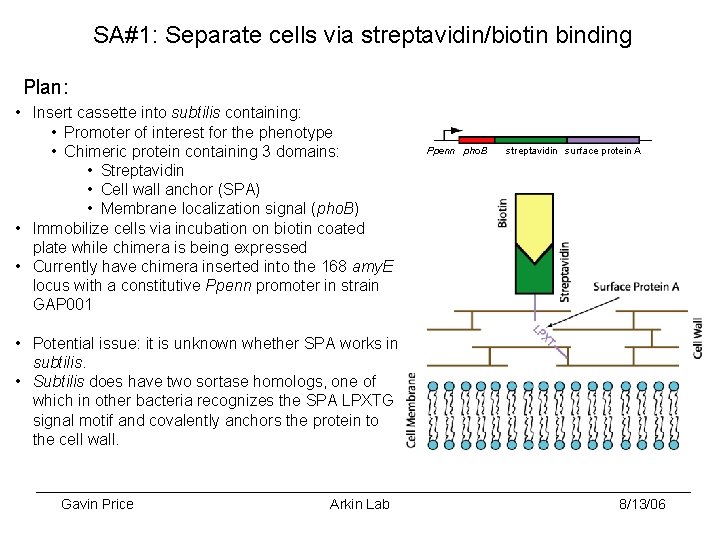
SA#1: Separate cells via streptavidin/biotin binding Plan: • Insert cassette into subtilis containing: • Promoter of interest for the phenotype • Chimeric protein containing 3 domains: • Streptavidin • Cell wall anchor (SPA) • Membrane localization signal (pho. B) • Immobilize cells via incubation on biotin coated plate while chimera is being expressed • Currently have chimera inserted into the 168 amy. E locus with a constitutive Ppenn promoter in strain GAP 001 Ppenn pho. B streptavidin surface protein A • Potential issue: it is unknown whether SPA works in subtilis. • Subtilis does have two sortase homologs, one of which in other bacteria recognizes the SPA LPXTG signal motif and covalently anchors the protein to the cell wall. Gavin Price Arkin Lab 8/13/06
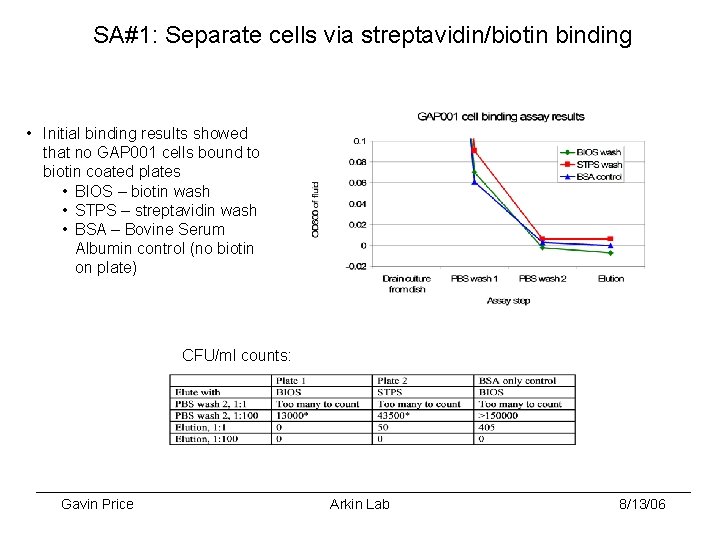
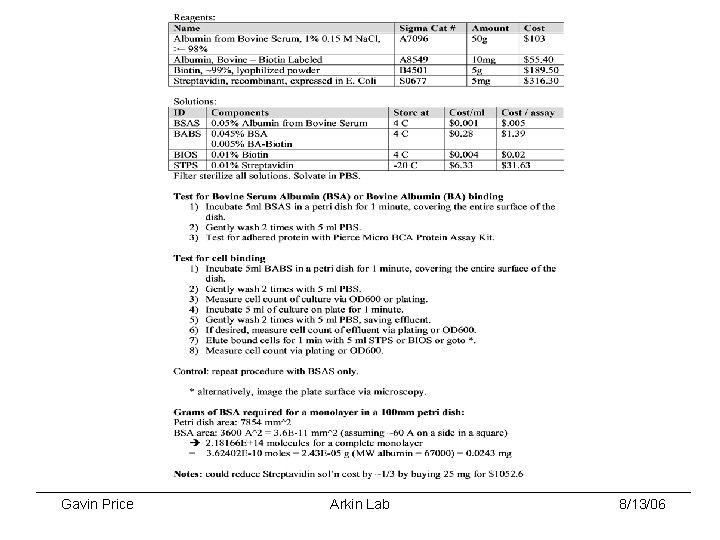
SA#1: Separate cells via streptavidin/biotin binding • Initial binding results showed that no GAP 001 cells bound to biotin coated plates • BIOS – biotin wash • STPS – streptavidin wash • BSA – Bovine Serum Albumin control (no biotin on plate) CFU/ml counts: Gavin Price Arkin Lab 8/13/06
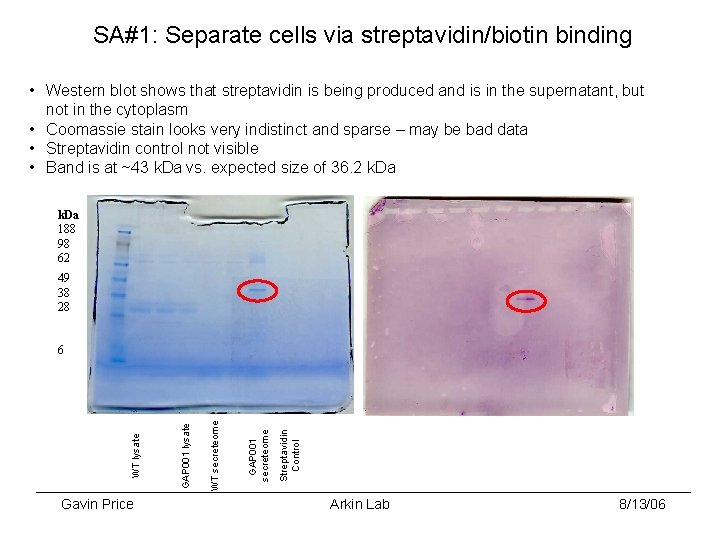
SA#1: Separate cells via streptavidin/biotin binding • Western blot shows that streptavidin is being produced and is in the supernatant, but not in the cytoplasm • Coomassie stain looks very indistinct and sparse – may be bad data • Streptavidin control not visible • Band is at ~43 k. Da vs. expected size of 36. 2 k. Da 188 98 62 49 38 28 Gavin Price Streptavidin Control GAP 001 secreteome WT secreteome GAP 001 lysate WT lysate 6 Arkin Lab 8/13/06

SA#1: Separate cells via streptavidin/biotin binding Next: • Redo Western blots with changes: • Fix gel prior to Coomassie stain • Use more concentrated streptavidin control • Use gentler lysis procedure (currently boiling for 10 min) • Resuspend pellet post lysis and run in gel • If chimera is anchored in the cell wall, likely to wind up in the pellet • If the chimera really is only in the supernatant, research alternative cell wall anchors or insert characterized sortase • Once binding is working, replace Ppenn with Pcom. K (competence) and Psp 0 IIE (sporulation) and prove separation of phenotypes via microscopy and flow cytometery Gavin Price Arkin Lab 8/13/06

Specific Aim #2 • Determine whether Rok plays a role in exit from competence • Although entry into competence is reasonably well understood, exit remains contentious • Elowitz 2006 claims that changes in Com. S concentration (expression measured by time lapse fluorescence microscopy) controls exit from competence but uses non-standard strains and media, and some of the experimental logic is questionable • A 1995 paper by Hahn and Dubnau shows that centrifuge separated cells express equal amounts of Com. S • The pathway topography suggests that Rok might play a role in competence exit, with or without Com. S Gavin Price Arkin Lab 8/13/06
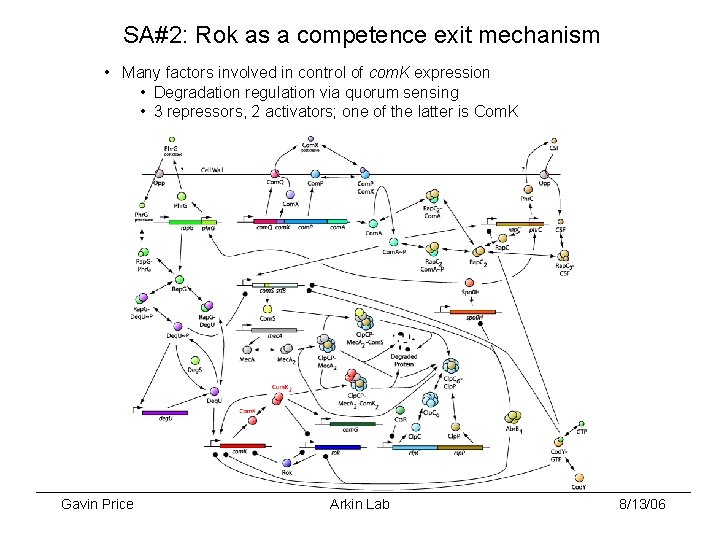
SA#2: Rok as a competence exit mechanism • Many factors involved in control of com. K expression • Degradation regulation via quorum sensing • 3 repressors, 2 activators; one of the latter is Com. K Gavin Price Arkin Lab 8/13/06
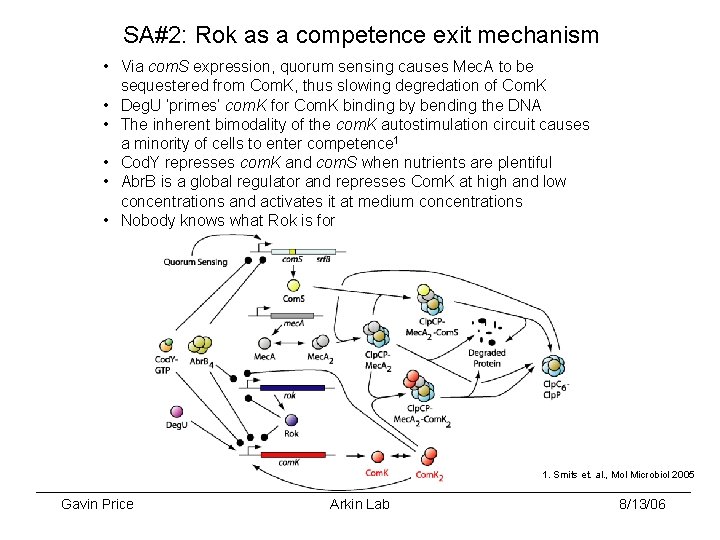
SA#2: Rok as a competence exit mechanism • Via com. S expression, quorum sensing causes Mec. A to be sequestered from Com. K, thus slowing degredation of Com. K • Deg. U ‘primes’ com. K for Com. K binding by bending the DNA • The inherent bimodality of the com. K autostimulation circuit causes a minority of cells to enter competence 1 • Cod. Y represses com. K and com. S when nutrients are plentiful • Abr. B is a global regulator and represses Com. K at high and low concentrations and activates it at medium concentrations • Nobody knows what Rok is for 1. Smits et. al. , Mol Microbiol 2005 Gavin Price Arkin Lab 8/13/06
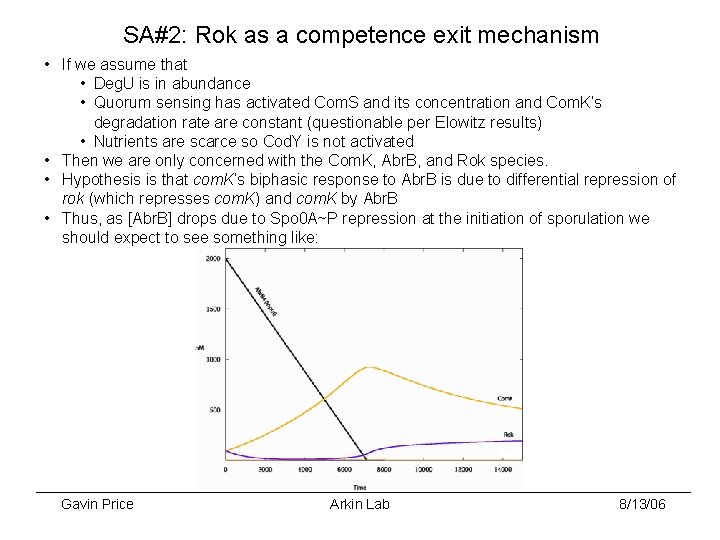
SA#2: Rok as a competence exit mechanism • If we assume that • Deg. U is in abundance • Quorum sensing has activated Com. S and its concentration and Com. K’s degradation rate are constant (questionable per Elowitz results) • Nutrients are scarce so Cod. Y is not activated • Then we are only concerned with the Com. K, Abr. B, and Rok species. • Hypothesis is that com. K’s biphasic response to Abr. B is due to differential repression of rok (which represses com. K) and com. K by Abr. B • Thus, as [Abr. B] drops due to Spo 0 A~P repression at the initiation of sporulation we should expect to see something like: Gavin Price Arkin Lab 8/13/06

SA#2: Rok as a competence exit mechanism • Hence, Rok acts as a switch to shut off competence as sporulation starts. • Possible tests for this hypothesis: • Simply measure com. K and rok expression over time with fluorescent reporters and compare against the prediction • Knockout rok. We should expect competence to start at the same time, but that some or all cells cannot exit competence. • Knockout rok and replace it with an inducible version. We should be able to regulate the fraction of cells that exit competence with varying levels of the inducer. Early induction should prevent or reduce the number of cells entering competence. • Measure reporters via microscopy (time-lapse? ) and flow cytometery. • If this explanation is correct, the fact that Com. K represses rok is strange. However, repression is only detected with overexpression of Com. K. Next: • Actually start working on the blasted thing Gavin Price Arkin Lab 8/13/06

Acknowledgements Adam Arkin Denise Wolf Funding from NIH Gavin Price Arkin Lab 8/13/06
Gavin Price Arkin Lab 8/13/06
- Slides: 16