Compensation Issues in Pathology for 2017 and Beyond

Compensation Issues in Pathology for 2017 and Beyond Jonathan L. Myles, MD, FCAP Chair, CAP Economic Affairs Committee Pathology Advisor, AMA-RUC October 26, 2016
Policy Drivers of Health Care Reform Promote Primary Care Expand Access to Preventative Care Promote Use of EHRs Strengthen Care Coordination Shift Payment Incentives From Volume to Value Misvalued Fee for Service Payment Initiative © 2016 College of American Pathologists. All rights reserved. 2

CAP Now and Future Policy Agenda Protect the value of pathology services © 2016 College of American Pathologists. All rights reserved. Ensure pathologists can adapt to new payment models Sustain a favorable laboratory regulatory environment 3

CAP Now and Future Policy Agenda Protect the value of pathology services © 2016 College of American Pathologists. All rights reserved. 4

47% of pathology CPT codes have been targeted by CMS since 2006 © 2016 College of American Pathologists. All rights reserved. 5

Medicare Payments for Pathology Codes by CMS increased 51% since 2006 © 2016 College of American Pathologists. All rights reserved. 6
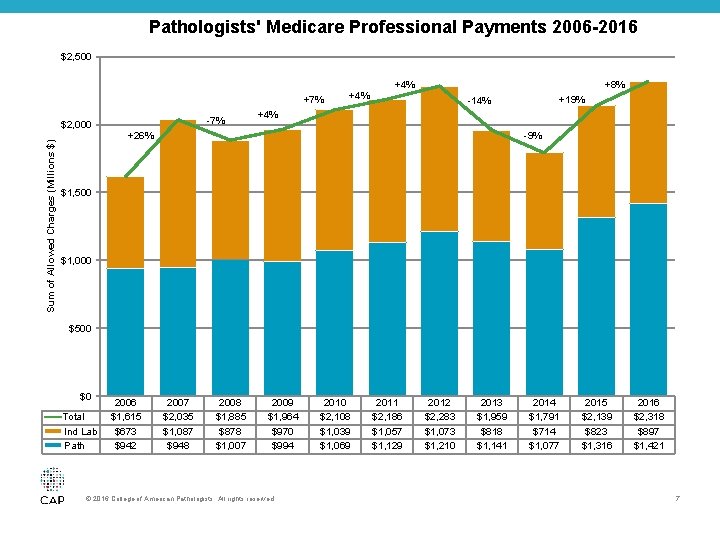
Pathologists' Medicare Professional Payments 2006 -2016 $2, 500 +7% Sum of Allowed Charges (Millions $) $2, 000 -7% +4% +8% +4% +19% -14% +26% -9% $1, 500 $1, 000 $500 $0 Total Ind Lab Path 2006 $1, 615 $673 $942 2007 $2, 035 $1, 087 $948 2008 $1, 885 $878 $1, 007 2009 $1, 964 $970 $994 © 2016 College of American Pathologists. All rights reserved. 2010 $2, 108 $1, 039 $1, 069 2011 $2, 186 $1, 057 $1, 129 2012 $2, 283 $1, 073 $1, 210 2013 $1, 959 $818 $1, 141 2014 $1, 791 $714 $1, 077 2015 $2, 139 $823 $1, 316 2016 $2, 318 $897 $1, 421 7

CAP Advocacy on Medicare Payment • CAP continues to advocate the CMS on Medicare reimbursement: – Advocating directly to the CMS throughout the year through face-to-face meetings – Via the CAP’s seat at the AMA/Specialty Society Relative Value Scale Update Committee – Submitting formal comments on fee schedules and other Medicare regulations © 2016 College of American Pathologists. All rights reserved. 8

Proposed 2017 Medicare Physician Fee Schedule • Proposed 2017 Medicare Physician Fee Schedule was released on July 7 – CAP members received a STATLINE Alert with initial analysis of this proposed ruling • CAP has engaged with the Centers for Medicare & Medicaid Services (CMS) • Final rule on or about November 1, 2016 © 2016 College of American Pathologists. All rights reserved. 9

Proposed Payment for Pathology Services CY 2017 Specialty Pathology Independent Laboratory Allowed Charges (millions) Impact of Work RVU PE RVU Changes Impact of MP RVU Changes Combined Impact $1, 127 0% -2% $701 0% -5% 0% -5% • Reflects averages by specialty (based on Medicare utilization) • For individual physicians and practices, the impact depends on mix of services and payers (Medicare and non-Medicare) • Physicians receive pay from other Medicare payment systems o For instance, independent laboratories receive 83% of their Medicare revenue from CLFS © 2016 College of American Pathologists. All rights reserved. 10

Proposed Payment for Pathology Services Due to Misvalued Code Initiatives • CMS proposed some increases sought by the CAP for pathology services, including – Pathology add-on services – Microslide consultations – Prostate G-code – Tumor immunohistochemistry • CMS proposed an update to many of the payment rates and accepted some of CAP’s and American Medical Association (AMA) Relative Value Update Committee’s (RUC) recommendations © 2016 College of American Pathologists. All rights reserved. 11

Proposed Payment for Pathology Services Due to Misvalued Code Initiatives • CMS proposed decreases in the physician work and practice expense of some services – Flow Cytometry • Reductions in Total RVUs of 20% or greater will be phased in over a two year period • CMS considers a 19% reduction as the maximum 1 year reduction for any service, and then phases in the remainder in the following year © 2016 College of American Pathologists. All rights reserved. 12

Proposed Work RVUs for Pathology Addon Services • Due to the CAP’s advocacy, the CMS again increased the value of add-on services for immunohistochemistry, immunofluorescence studies, and in situ hybridization – CMS proposes to increase the valuation from a 24% discount from the base code in 2016 to a 20% discount in 2017 – This proposed policy change represents a $2. 93 million increased payment to pathologists based on Medicare volume – $2. 75 million of this impact is attributed to CPT code 88341 – CAP continues to maintain the difference in physician work from the base code to the add-on service is diminutive © 2016 College of American Pathologists. All rights reserved. 13

Reimbursement Policy Changes • CMS proposed to impact pathologists practice expense RVUs by making wide-ranging standardizations to clinical labor tasks and time across codes – CAP advocated that pathology services are unique and should not be subject to blanket standardizing edits o Importantly, CMS did not appear to consider how differing batch and block sizes across codes would interact with the new standard times – The Agency mostly agreed with the CAP and only finalized 6 of the 17 proposed standardizations – The CAP continues to work with the agency on this and other practice expense issues © 2016 College of American Pathologists. All rights reserved. 14
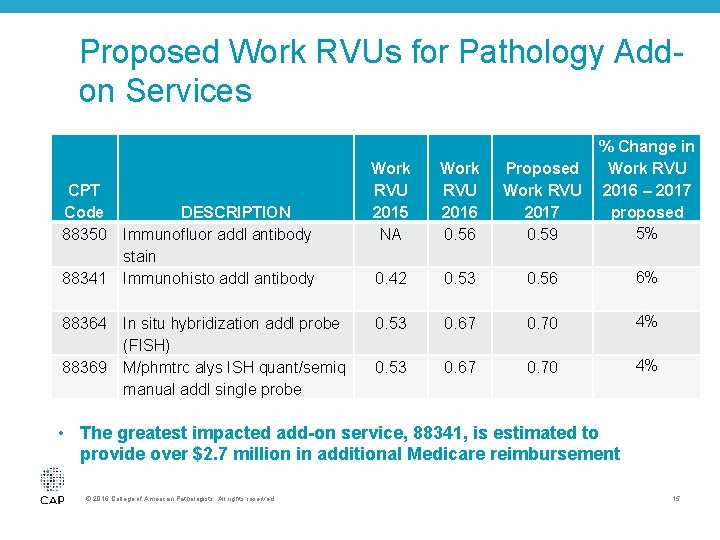
Proposed Work RVUs for Pathology Addon Services CPT Code 88350 88341 88364 88369 DESCRIPTION Immunofluor addl antibody stain Immunohisto addl antibody In situ hybridization addl probe (FISH) M/phmtrc alys ISH quant/semiq manual addl single probe % Change in Proposed Work RVU 2016 – 2017 proposed 5% 0. 59 Work RVU 2015 NA Work RVU 2016 0. 56 0. 42 0. 53 0. 56 6% 0. 53 0. 67 0. 70 4% • The greatest impacted add-on service, 88341, is estimated to provide over $2. 7 million in additional Medicare reimbursement © 2016 College of American Pathologists. All rights reserved. 15
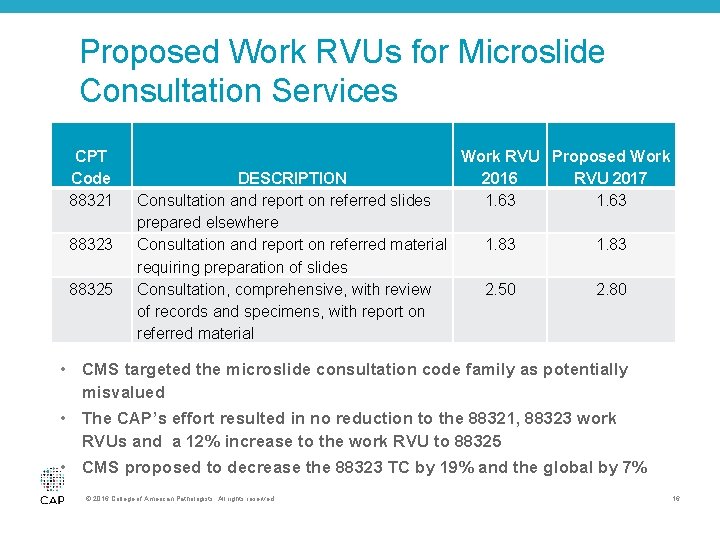
Proposed Work RVUs for Microslide Consultation Services CPT Code 88321 88323 88325 DESCRIPTION Consultation and report on referred slides prepared elsewhere Consultation and report on referred material requiring preparation of slides Consultation, comprehensive, with review of records and specimens, with report on referred material Work RVU Proposed Work 2016 RVU 2017 1. 63 1. 83 2. 50 2. 80 • CMS targeted the microslide consultation code family as potentially misvalued • The CAP’s effort resulted in no reduction to the 88321, 88323 work RVUs and a 12% increase to the work RVU to 88325 • CMS proposed to decrease the 88323 TC by 19% and the global by 7% © 2016 College of American Pathologists. All rights reserved. 16

Prostate Biopsy Services BACKGROUND • In 2009, CMS created four G-codes for the surgical pathology of prostate saturation biopsy services • CMS changed the set of descriptors in 2013, 2014, and in 2015 • 2015 changes eliminated codes G 0417, G 0418, and G 0419, and revised the descriptor for G 0416 – the code would apply to all prostate biopsy specimens regardless of the number of specimens or technique used to obtain the biopsy • CMS stated this simplified the coding and mitigated overutilization incentives © 2016 College of American Pathologists. All rights reserved. 17

Prostate Biopsy Services BACKGROUND • For 2015, G 0416 was identified by CMS as a potentially misvalued service • The AMA RUC and CMS evaluated the TC direct practice expense inputs and the PC in 2015 because they were based on outdated information and a coding structure that no longer exists © 2016 College of American Pathologists. All rights reserved. 18

Prostate Biopsy Services (PC) 2016 -2017 proposed Percentage Change 2015 Payment 2016 Payment 2017 Proposed Payment Prostate biopsy, any method $182. 90 $157. 90 $184. 96 17% TC Prostate biopsy, any method $466. 42 $376. 30 $303. 37 -19% Prostate biopsy, any method $649. 32 $534. 20 $488. 33 -9% CPT Code MOD G 0416 26 G 0416 DESCRIPTOR • CAP sought increases to the professional component of the prostate G-code • As a result of this effort, the CMS proposed to increase the physician work component of G 0416 from 3. 09 to 3. 60 o This represents a 17% increase © 2016 College of American Pathologists. All rights reserved. 19

Prostate Biopsy Services (TC) 2016 -2017 proposed Percentage Change 2015 Payment 2016 Payment 2017 Proposed Payment Prostate biopsy, any method $182. 90 $157. 90 $184. 96 17% TC Prostate biopsy, any method $466. 42 $376. 30 $303. 37 -19% Prostate biopsy, any method $649. 32 $534. 20 $488. 33 -9% CPT Code MOD G 0416 26 G 0416 DESCRIPTOR • The CMS also proposes further reductions to the TC payment for prostate services • Due to the TC review and shifts in medical service volume from the change in the description of service, the payment for these services decreased significantly • Changes in total RVUs will be phased-in over a 2 year period © 2016 College of American Pathologists. All rights reserved. 20

Payment for Flow Cytometry • In its 2016 rulemaking, CMS included cuts to flow cytometry codes TC codes 88184 and 88185 as the agency previously identified the services an overvalued • The CMS is phasing in further TC reductions over the next two years • CMS also targeted the professional component of flow cytometry • The 2017 proposed changes includes additional reductions to the professional and technical component valuations of the flow cytometry codes • The CAP engaged with a variety of stakeholders, the AMA RUC, and CMS in an attempt to reinstate some of the previously identified reductions to these codes © 2016 College of American Pathologists. All rights reserved. 21
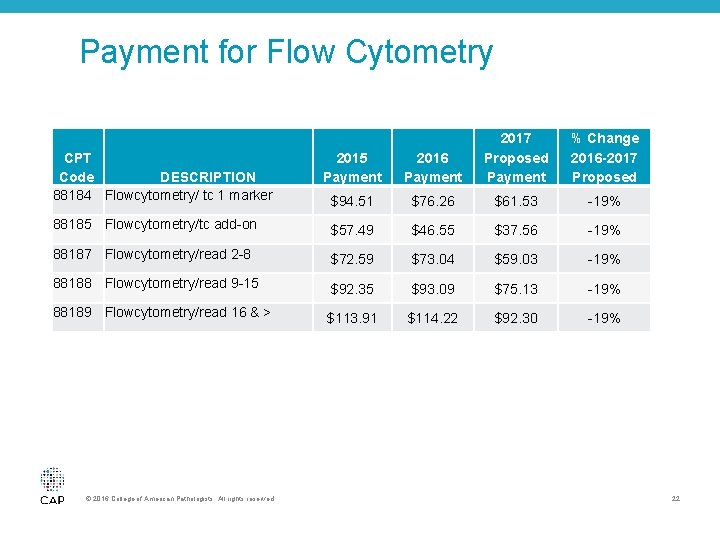
Payment for Flow Cytometry 2015 Payment 2016 Payment 2017 Proposed Payment $94. 51 $76. 26 $61. 53 -19% 88185 Flowcytometry/tc add-on $57. 49 $46. 55 $37. 56 -19% 88187 Flowcytometry/read 2 -8 $72. 59 $73. 04 $59. 03 -19% 88188 Flowcytometry/read 9 -15 $92. 35 $93. 09 $75. 13 -19% 88189 Flowcytometry/read 16 & > $113. 91 $114. 22 $92. 30 -19% CPT DESCRIPTION Code 88184 Flowcytometry/ tc 1 marker © 2016 College of American Pathologists. All rights reserved. % Change 2016 -2017 Proposed 22

Payment for Tumor Immunohistochemistry • In 2016, the CMS identified 88360 as potentially misvalued on the High Expenditure by Specialty Screen • Advocacy from the CAP and others assisted in proposed increases to the TC of 88360 and 88361 • These services are still under CMS’ review at this time and more changes in reimbursement are expected in the future © 2016 College of American Pathologists. All rights reserved. 23
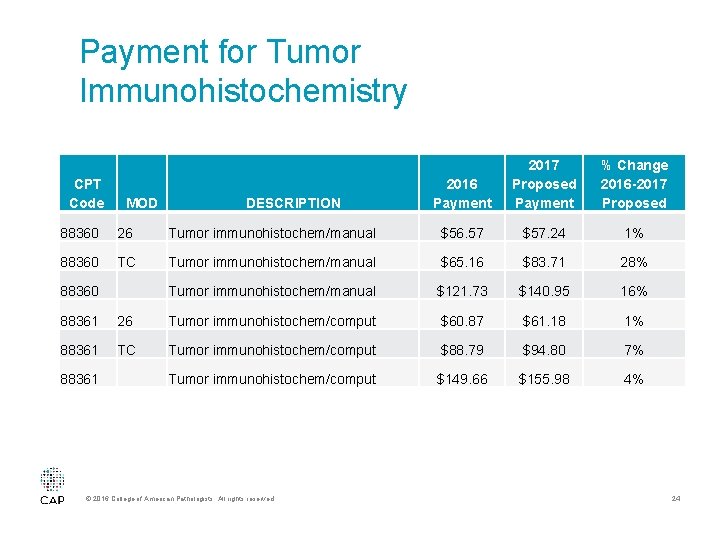
Payment for Tumor Immunohistochemistry CPT Code MOD DESCRIPTION 2016 Payment 2017 Proposed Payment % Change 2016 -2017 Proposed 88360 26 Tumor immunohistochem/manual $56. 57 $57. 24 1% 88360 TC Tumor immunohistochem/manual $65. 16 $83. 71 28% Tumor immunohistochem/manual $121. 73 $140. 95 16% 88360 88361 26 Tumor immunohistochem/comput $60. 87 $61. 18 1% 88361 TC Tumor immunohistochem/comput $88. 79 $94. 80 7% Tumor immunohistochem/comput $149. 66 $155. 98 4% 88361 © 2016 College of American Pathologists. All rights reserved. 24

Payment for Surgical Pathology • CMS’ Misvalued Code Initiatives continue to scrutinize these services – Proposed changes to the type, quantity, and price of eosin stain, as a direct practice expense item, resulted in proposed decreases to anatomic pathology TC payment – The CAP will engage with the CMS and other stakeholders to address this proposal © 2016 College of American Pathologists. All rights reserved. 25
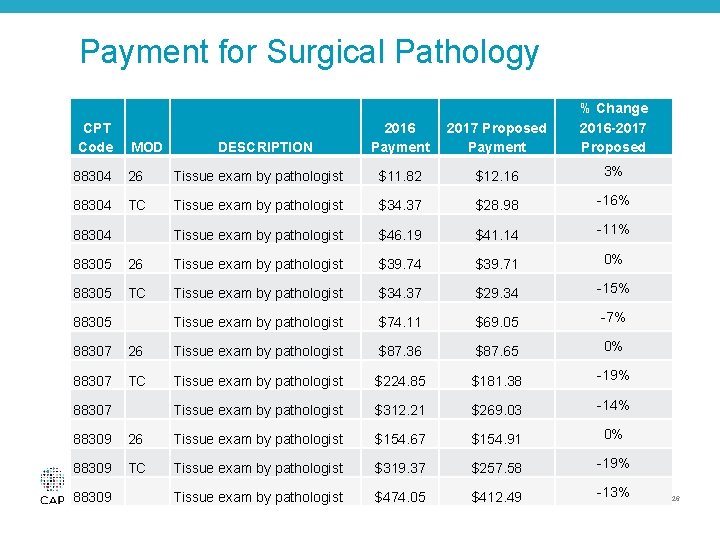
Payment for Surgical Pathology CPT Code MOD DESCRIPTION 2016 Payment 2017 Proposed Payment % Change 2016 -2017 Proposed 88304 26 Tissue exam by pathologist $11. 82 $12. 16 3% 88304 TC Tissue exam by pathologist $34. 37 $28. 98 -16% 88304 Tissue exam by pathologist $46. 19 $41. 14 -11% 88305 26 Tissue exam by pathologist $39. 74 $39. 71 0% 88305 TC Tissue exam by pathologist $34. 37 $29. 34 -15% 88305 Tissue exam by pathologist $74. 11 $69. 05 -7% 88307 26 Tissue exam by pathologist $87. 36 $87. 65 0% 88307 TC Tissue exam by pathologist $224. 85 $181. 38 -19% 88307 Tissue exam by pathologist $312. 21 $269. 03 -14% 88309 26 Tissue exam by pathologist $154. 67 $154. 91 0% 88309 TC Tissue exam by pathologist $319. 37 $257. 58 -19% © 2016 College of American Pathologists. All rights reserved. 88309 Tissue exam by pathologist $474. 05 $412. 49 -13% 26

Payment for New Skin Reflectance Confocal Microscopy CPT codes CPT Description Code 96931 Reflectance confocal microscopy (RCM) for cellular and sub-cellular imaging of skin; image acquisition and interpretation and report, first lesion 96932 Reflectance confocal microscopy (RCM) for cellular and sub-cellular imaging of skin; image acquisition only, first lesion 96933 Reflectance confocal microscopy (RCM) for cellular and sub-cellular imaging of skin; interpretation and report only, first lesion © 2016 College of American Pathologists. All rights reserved. Work Total Non- Proposed RVU Facility RVU Payment 0. 75 4. 40 $157. 41 0. 00 2. 90 $103. 75 0. 75 NA $42. 93 27

Payment for New Skin Reflectance Confocal Microscopy CPT codes CPT Description Code 96934 Reflectance confocal microscopy (RCM) for cellular and sub-cellular imaging of skin; image acquisition and interpretation and report, each additional lesion (List separately in addition to code for primary procedure) 96935 Reflectance confocal microscopy (RCM) for cellular and sub-cellular imaging of skin; image acquisition only, each additional lesion (List separately in addition to code for primary procedure) 96936 Reflectance confocal microscopy (RCM) for cellular and sub-cellular imaging of skin; interpretation and report only, each additional lesion (List separately in addition to code for primary procedure) © 2016 College of American Pathologists. All rights reserved. Work Total Non- Proposed RVU Facility RVU Payment 0. 71 2. 23 $79. 78 0. 00 0. 97 $34. 70 0. 71 NA $40. 78 28
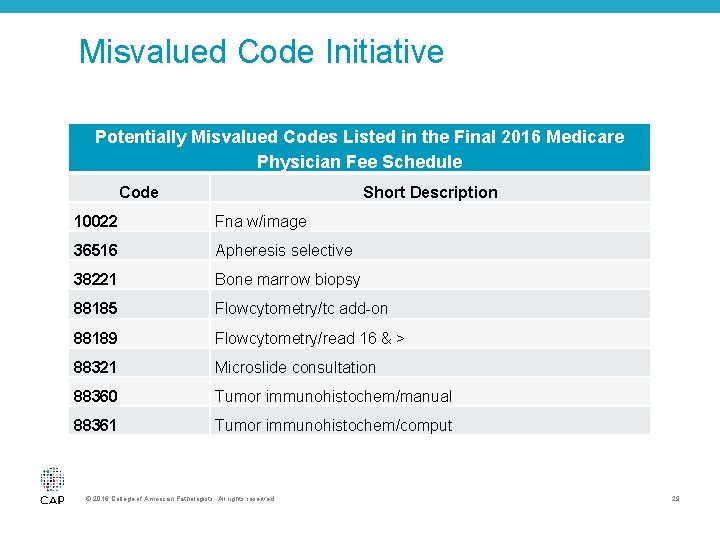
Misvalued Code Initiative Potentially Misvalued Codes Listed in the Final 2016 Medicare Physician Fee Schedule Code Short Description 10022 Fna w/image 36516 Apheresis selective 38221 Bone marrow biopsy 88185 Flowcytometry/tc add-on 88189 Flowcytometry/read 16 & > 88321 Microslide consultation 88360 Tumor immunohistochem/manual 88361 Tumor immunohistochem/comput © 2016 College of American Pathologists. All rights reserved. 29

Medicare Access & CHIP Reauthorization Act (MACRA) © 2016 College of American Pathologists. All rights reserved. 30

Background • How did we (and our public, payers and regulators) get to MACRA, and where is it taking us? • Our system of healthcare financing structurally increases expenses © 2016 College of American Pathologists. All rights reserved. 31

Background, continued • Inflexion points in US health care finance – Business: World War II => Wage Freeze => Fringe Benefits – Government: Social Security => Title XVIII Medicare © 2016 College of American Pathologists. All rights reserved. 32

Background, continued US Healthcare Expenditures $10, 000, 000 1, 000, 000 $1, 000, 000 $100, 000, 000 $10, 000, 000 Medicare Fringes $1, 000, 000 100, 000 1934 1938 1942 1946 1950 1954 1958 1962 1966 1970 1974 1978 1982 1986 1990 1994 1998 2002 2006 2010 2014 $100, 000 Healthcare Population http: //www. bea. gov/national/index. htm https: //www. gpo. gov/fdsys/browse/collection. GPO. action? collection. Code=BUDGET © 2016 College of American Pathologists. All rights reserved. 33

Background, continued • Inflexion points in US healthcare finance – Business: World War II => Wage Freeze =>Fringe Benefits – Government: Social Security => Title XVIII Medicare & Title XIX Medicaid – Accelerating Medicare Expenditures => Balanced Budget Act => Sustainable Growth Rate (SGR) © 2016 College of American Pathologists. All rights reserved. 34
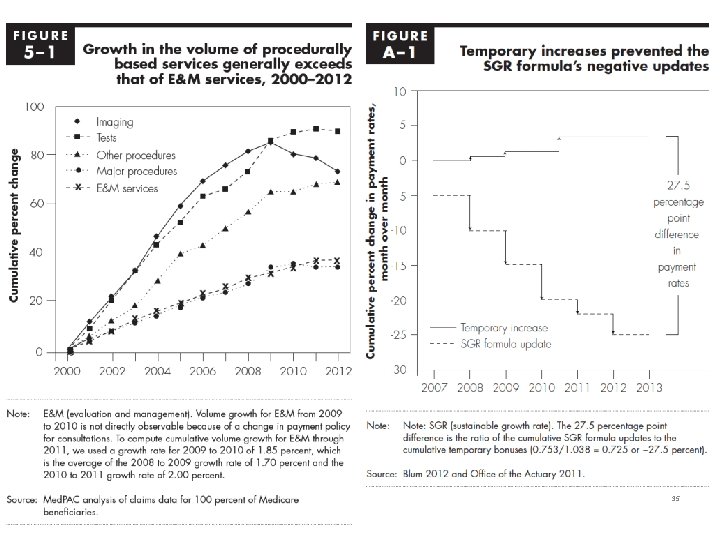
35

Medicare Access and CHIP Reauthorization Act of 2015 (MACRA) • Ended the broken Sustainable Growth Rate (SGR) formula • Established a new framework for rewarding health care providers for giving better care not more just more care • Combined existing Medicare quality reporting programs into one new system (Merit-Based Incentive Payment System, or MIPS) © 2016 College of American Pathologists. All rights reserved. 36

MACRA Implementation • Final MACRA rule published on October 14 – Additional flexibility • For example, avoid penalty by reporting one quality measure in 2017 – All eight CAP quality measures approved for MIPS – Changes to non-patient-facing physicians definition – Pathologists, including those practicing at independent laboratories, are eligible for MIPS – Reduced risk criteria for alternative payment models © 2016 College of American Pathologists. All rights reserved. 37
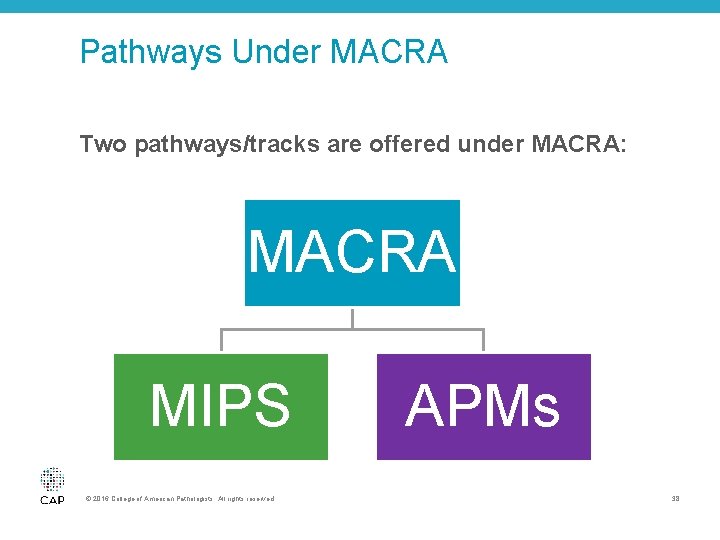
Pathways Under MACRA Two pathways/tracks are offered under MACRA: MACRA MIPS © 2016 College of American Pathologists. All rights reserved. APMs 38

Additional Options Possible under MIPS On September 8, the CMS administrator announced plans to provide easier options under MIPS for 2017: MIPS In 2017 Test the quality programs © 2016 College of American Pathologists. All rights reserved. Participate for part of 2017 Participate for full year 39

2017 Transition Year for MIPS • The 2017 performance year affects 2019 Medicare reimbursement – Test MIPS by submitting minimum amount of data in 2017 will stop pay penalty – Do not participate in MIPS leads to -4% pay penalty in 2019 – Submit quality data to Medicare for 90 days in 2017 and may earn a small bonus in 2019 – Submit full year of data in 2017 and may earn a moderate bonus in 2019 © 2016 College of American Pathologists. All rights reserved. 40

Merit-Based Incentive Payment System (MIPS) • System to assess the total performance of each MIPS eligible professional (EP) according to performance standards for a year • A composite performance score (CPS) for each professional will be calculated for each performance period • The composite performance score determines a MIPS adjustment factor applied to the EP’s Medicare reimbursement for the year © 2016 College of American Pathologists. All rights reserved. 41
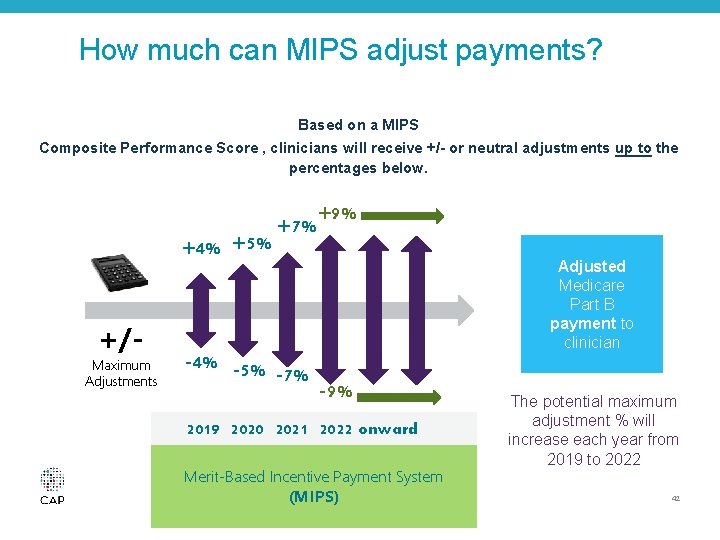
How much can MIPS adjust payments? Based on a MIPS Composite Performance Score , clinicians will receive +/- or neutral adjustments up to the percentages below. +4% +5% +/- Maximum Adjustments +7% -4% -5% -7% +9% Adjusted Medicare Part B payment to clinician -9% 2019 2020 2021 2022 onward Merit-Based Incentive Payment System (MIPS) The potential maximum adjustment % will increase each year from 2019 to 2022 42
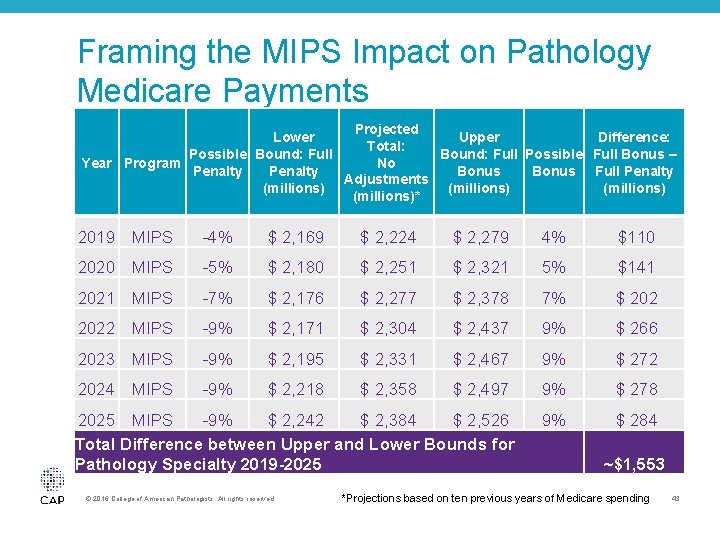
Framing the MIPS Impact on Pathology Medicare Payments Projected Lower Upper Difference: Total: Possible Bound: Full Possible Full Bonus – Year Program No Penalty Bonus Full Penalty Adjustments (millions)* 2019 MIPS -4% $ 2, 169 $ 2, 224 $ 2, 279 4% $110 2020 MIPS -5% $ 2, 180 $ 2, 251 $ 2, 321 5% $141 2021 MIPS -7% $ 2, 176 $ 2, 277 $ 2, 378 7% $ 2022 MIPS -9% $ 2, 171 $ 2, 304 $ 2, 437 9% $ 266 2023 MIPS -9% $ 2, 195 $ 2, 331 $ 2, 467 9% $ 272 2024 MIPS -9% $ 2, 218 $ 2, 358 $ 2, 497 9% $ 278 2025 MIPS -9% $ 2, 242 $ 2, 384 $ 2, 526 Total Difference between Upper and Lower Bounds for Pathology Specialty 2019 -2025 9% $ 284 © 2016 College of American Pathologists. All rights reserved. ~$1, 553 *Projections based on ten previous years of Medicare spending 43
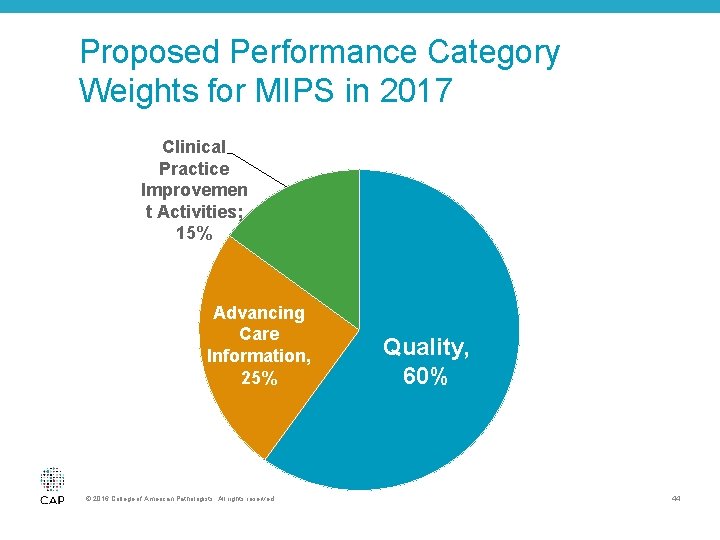
Proposed Performance Category Weights for MIPS in 2017 Clinical Practice Improvemen t Activities; 15% Advancing Care Information, 25% © 2016 College of American Pathologists. All rights reserved. Quality, 60% 44

MIPS Category Weights for 2017 60% Quality • Replaces Physician Quality Reporting System (PQRS); report up to six quality measures Clinical Practice • New category; attest that you complete up 15% Improvement to four improvement activities Advancing Care • Replaces EHR Incentive Program; non 25% patient-facing clinicians do not need to Information report this category 0% Cost • Replaces value-based modifier; not a factor for 2017, will start to count in 2018 45 © 2016 College of American Pathologists. All rights reserved.

Alternative Payment Models (APMs) • Per CMS, APMs are new approaches to paying for medical care through Medicare that incentivize quality & value • Only “Advanced APMs” qualify for MACRA APM payment incentives © 2016 College of American Pathologists. All rights reserved. 46
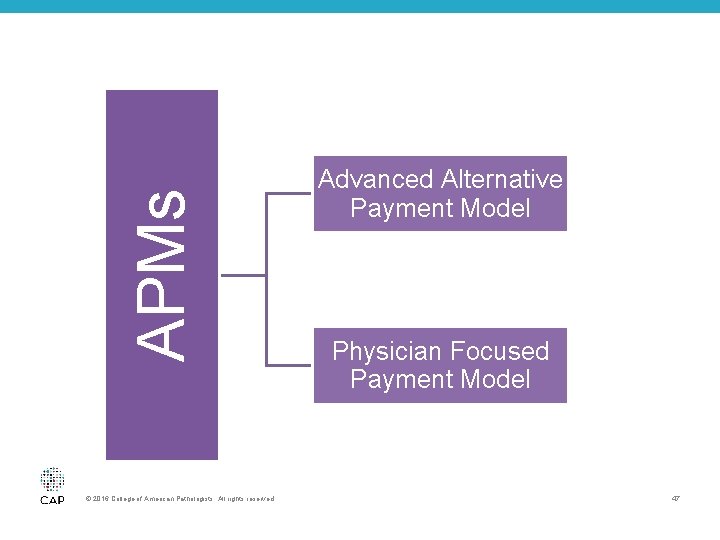
APMs © 2016 College of American Pathologists. All rights reserved. Advanced Alternative Payment Model Physician Focused Payment Model 47

APMs and Pathology Challenges • Advanced APMs not likely to be available for participation by a broad number of physicians • The five models that presently qualify as advanced APMs are: – Oncology care model (2 -sided risk – starts in 2018) – Next Generation ACO (2 -sided risk) – MSSP – Tracks 2 and 3 (2 -sided risk) – Comprehensive Primary Care Plus (primary care organizations) – Comprehensive ESRD Care Model (large dialysis organizations) © 2016 College of American Pathologists. All rights reserved. 48
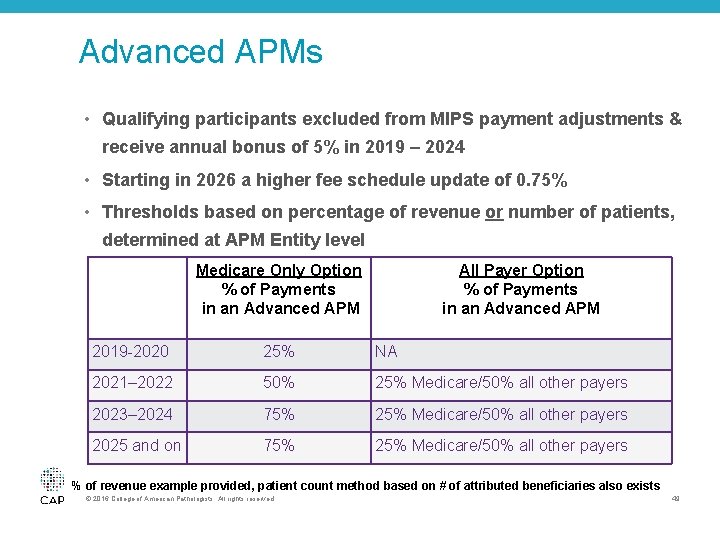
Advanced APMs • Qualifying participants excluded from MIPS payment adjustments & receive annual bonus of 5% in 2019 – 2024 • Starting in 2026 a higher fee schedule update of 0. 75% • Thresholds based on percentage of revenue or number of patients, determined at APM Entity level Medicare Only Option % of Payments in an Advanced APM All Payer Option % of Payments in an Advanced APM 2019 -2020 25% NA 2021– 2022 50% 25% Medicare/50% all other payers 2023– 2024 75% 25% Medicare/50% all other payers 2025 and on 75% 25% Medicare/50% all other payers % of revenue example provided, patient count method based on # of attributed beneficiaries also exists © 2016 College of American Pathologists. All rights reserved. 49
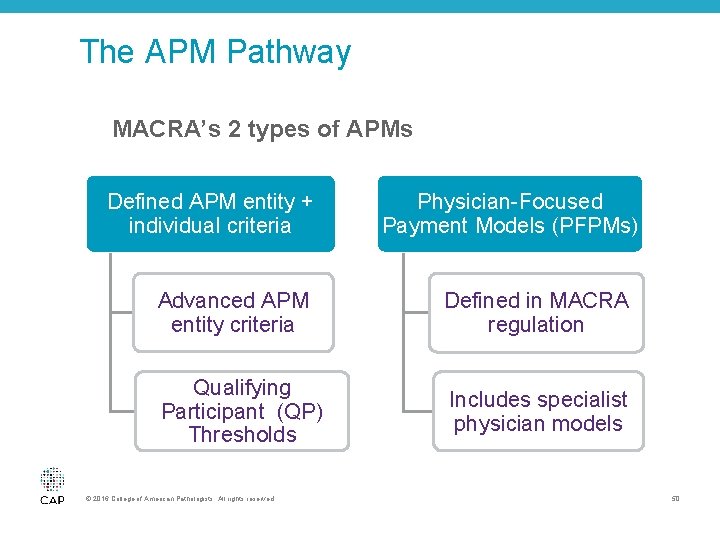
The APM Pathway MACRA’s 2 types of APMs Defined APM entity + individual criteria Physician-Focused Payment Models (PFPMs) Advanced APM entity criteria Defined in MACRA regulation Qualifying Participant (QP) Thresholds Includes specialist physician models © 2016 College of American Pathologists. All rights reserved. 50

APMs and Pathology Challenges • Pathology not as readily adapted to PFPM development as more episodic specialties which have attributed patients • No shortcuts to developing a PFPM that will benefit the specialty financially & improve patient care • Pathologists do influence a large % of spending but do not control, and therefore cannot be held responsible for, total cost of care in an APM (or readily in a PFPM) • Potential opportunities, but also uncertainty & potential risk (including downside financial risk) © 2016 College of American Pathologists. All rights reserved. 51

APM Models and the CAP’s Focus • Education and information to enable pathologists compliance with the APM pathway, as applicable • Assessment of feasibility of the PFPM option for pathologists that will fulfill MACRA requirements – If feasible, development of model for PTAC submission © 2016 College of American Pathologists. All rights reserved. 52

Medicare Regulatory and Payment Update Timeline © 2016 College of American Pathologists. All rights reserved. 53

Advocacy on MACRA for CAP Members • Prevent pathologists from being unfairly penalized under MIPS rules: o MIPS categories must accommodate non-patient-facing clinicians o When categories don’t fit pathology practice, pathologists should receive a median score o Identify pathologists as non-patient-facing clinicians instead of using an arbitrary formula © 2016 College of American Pathologists. All rights reserved. 54

Advocacy on MACRA for CAP Members Going forward, the CAP is … • Developing new quality measures • Engaging with CMS on clinical practice improvement activities • Evaluating APM opportunities for pathologists • Evaluating QCDR implementation © 2016 College of American Pathologists. All rights reserved. 55

The RUC • Resource Based Relative Value System Update Committee (1991) • The RUC: – 31 members (including CAP) including four rotating seats (1 PC, 2 IM, 1 open), AMA and CPT members (28 voting members) • Advisory Committee: 122 representatives from each specialty society seated in the AMA HOD • Functions of the RUC – Value new and revised codes put forth by CPT – Review inputs on established but potentially misvalued codes (Continuous process) – Practice expense refinements © 2016 College of American Pathologists. All rights reserved. 56
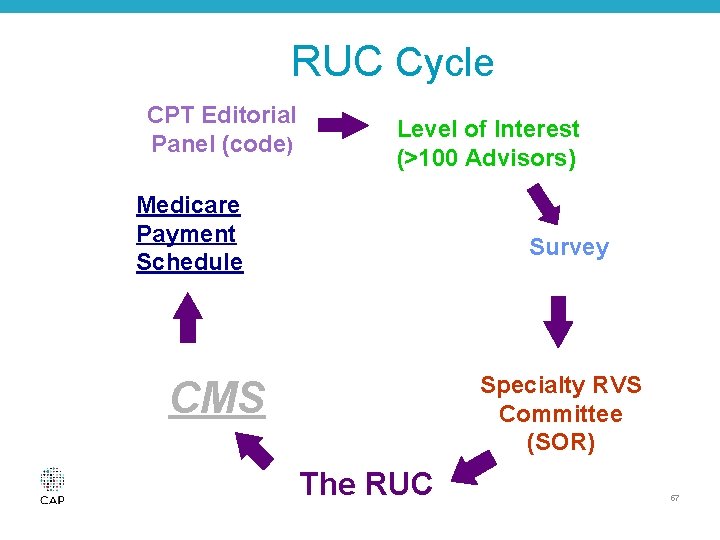
RUC Cycle CPT Editorial Panel (code) Level of Interest (>100 Advisors) Medicare Payment Schedule Survey Specialty RVS Committee (SOR) CMS The RUC 57

WHAT’S in an RVU? • PHYSICIAN WORK - based on member surveys – time, skill, effort, stress • PRACTICE EXPENSE excludes Indirect practice expenses – overhead • PLI (malpractice liability) – based on formula derived from actuarial data © 2016 College of American Pathologists. All rights reserved. 58

RVU Final Formula Physician Work RVU x Work (GPCI*) + Practice Expense RVU x PE (GPCI) + Malpractice (PLI) RVU x PLI (GPCI) = Total RVU X CY Conversion Factor = Payment CF for CY 2016 $35. 8043 Geographic Practice Cost Index is variable across regions based on prevailing rates for supplies and support © 2016 College of American Pathologists. All rights reserved. 59

Patient Access to Medicare Act (PAMA) © 2016 College of American Pathologists. All rights reserved. 60
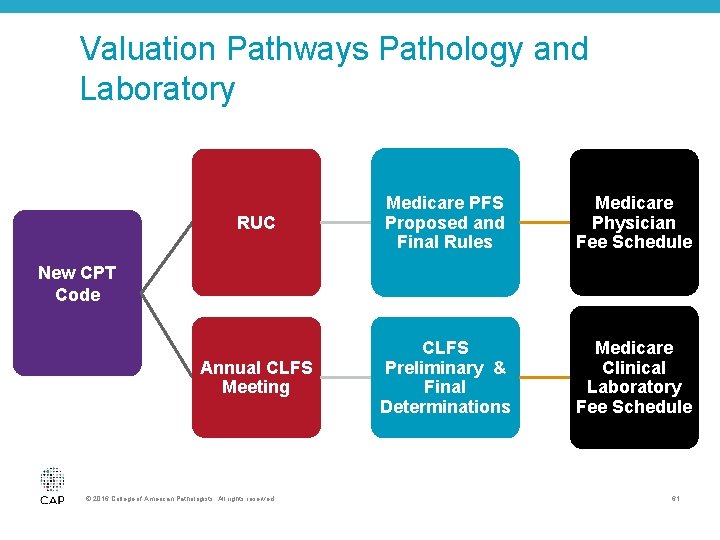
Valuation Pathways Pathology and Laboratory RUC Medicare PFS Proposed and Final Rules Medicare Physician Fee Schedule Annual CLFS Meeting CLFS Preliminary & Final Determinations Medicare Clinical Laboratory Fee Schedule New CPT Code © 2016 College of American Pathologists. All rights reserved. 61

Protecting Access to Medicare Act (PAMA) Implementation of PAMA • Final rule release in June 2016 • Establishes reporting requirements for labs, data used to set reduced Clinical Diagnostic Laboratory Test Payments o 10% reduction cap 2017– 2019 o 15% reduction cap 2020– 2022 © 2016 College of American Pathologists. All rights reserved. 62

PAMA Final Rule Results Overall: Progress made on each of the 6 policy objectives over proposed rule. • Implementation of the new CLFS and data collection period delayed a year • At least some hospital outreach laboratories included • TIN level reporting & certification even though applicable lab determined at the NPI level • Language that tempers civil monetary penalty liability • Coding structure and processes for CLFS services preserved • No consolidation of coverage and/or claims to from 1 – 4 MACs. © 2016 College of American Pathologists. All rights reserved. 63

FDA Laboratory. Developed Test Oversight © 2016 College of American Pathologists. All rights reserved. 64

FDA Guidance on Laboratory-Developed Tests • Final FDA guidance expected in 2016 • CAP remains engaged with FDA and Congress on issues concerning LDTs • CAP advocates for: – Patient access to LDTs – Continued innovation of tests – Guidance that does not overburden laboratories with administrative requirements © 2016 College of American Pathologists. All rights reserved. 65

CAP Recommendations to the FDA The CAP recommended significant changes to the FDA draft LDT guidance, including: • Removal of duplicative and unnecessary requirements for laboratories • Changing its definition of an LDT • Continued enforcement discretion for low risk tests • Limiting the breadth of the high risk category so that it does not include all companion diagnostics © 2016 College of American Pathologists. All rights reserved. 66

Congressional LDT Activities • The CAP is actively engaged with Congress; however, release of FDA guidance likely prior to Congressional resolution • House: E&C Committee released a discussion draft • House: Appropriations Committee passed report language to deter the release of the FDA LDT guidance • Senate: HELP Committee passed its “Innovation Bill”; but it does not include any LDT language 67 © 2016 College of American Pathologists. All rights reserved.

Thank You Jonathan L. Myles, M. D. The Cleveland Clinic 9500 Euclid Ave L 25 Cleveland, OH 44195 216 -444 -2842 mylesj@ccf. org © 2016 College of American Pathologists. All rights reserved. 68
Part A/B MAC Current Jurisdictions © 2016 College of American Pathologists. All rights reserved. 69
- Slides: 69