Comparison of the HIV LIA vs WB on

Comparison of the HIV LIA vs WB on HIV-Negative Samples CDC-HIV Diagnostics Meeting “New Diagnostic Technologies” Dec 5 -7, 2007 Dr. John Kim National Laboratory for HIV Reference Services Public Health Agency of Canada Ottawa, Ontario
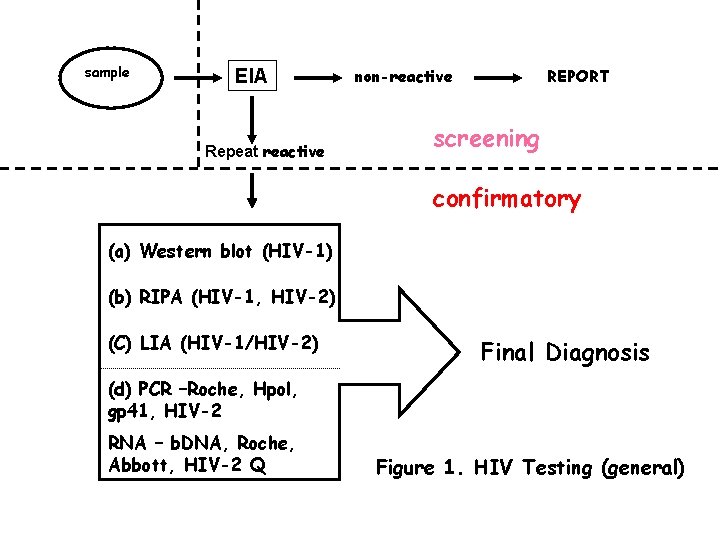
sample EIA Repeat reactive non-reactive REPORT screening confirmatory (a) Western blot (HIV-1) (b) RIPA (HIV-1, HIV-2) (C) LIA (HIV-1/HIV-2) Final Diagnosis (d) PCR –Roche, Hpol, gp 41, HIV-2 RNA – b. DNA, Roche, Abbott, HIV-2 Q Figure 1. HIV Testing (general)
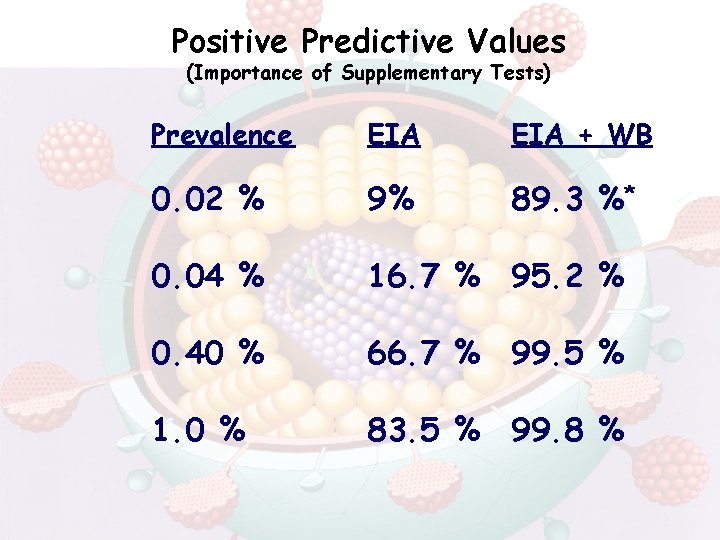
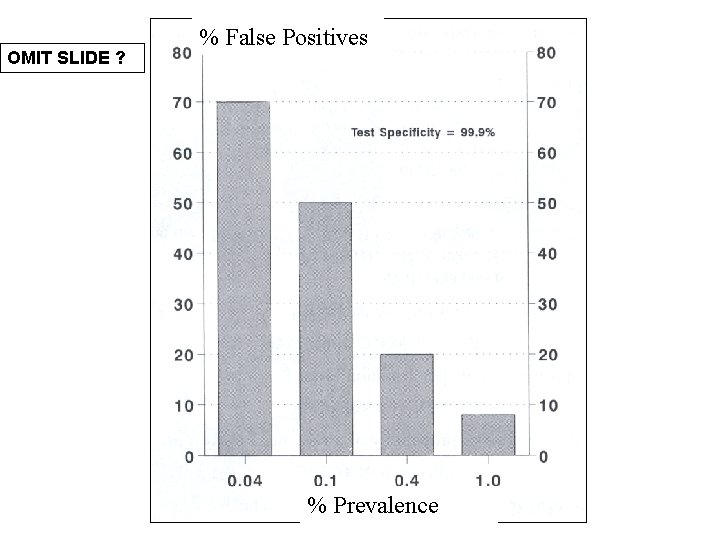
Positive Predictive Values (Importance of Supplementary Tests) Prevalence EIA + WB 0. 02 % 9% 89. 3 %* 0. 04 % 16. 7 % 95. 2 % 0. 40 % 66. 7 % 99. 5 % 1. 0 % 83. 5 % 99. 8 %

INNO-LIA HIV (new technology ? ) • New anti-human immunodeficiency virus immunoblot assays resolve non-specific Western blot results. (Zaaijer et al, Transfusion, 37: 193, 1997). • Validation of a new immunoblot assay for confirmation of human immunodeficiency virus infection (Zaaijer et al, Transfusion, 38: 776, 1998). • Ability of the Innogenetics Inno-Lia to distinguish HIV-1, HIV 2 and dually reactive sera. (Granade et al, Abstr. Gen Meet Am Soc Microbiol, 92: 416 [T-47[, 1992. • Confirmation of HIV infection with the line immune assay (INNO-LIA) using whole blood impregnated filter paper discs. (Kestens et al, Abstr. Int Conf Aids 5: 309 [TBP 135], 1989.
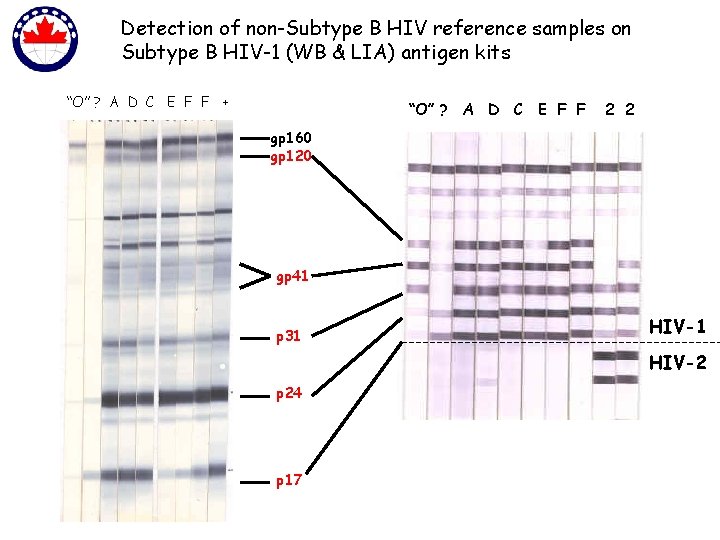
Detection of non-Subtype B HIV reference samples on Subtype B HIV-1 (WB & LIA) antigen kits “O” ? A D C E F F + “O” ? A D C E F F 2 2 gp 160 gp 120 gp 41 p 31 HIV-2 p 24 p 17
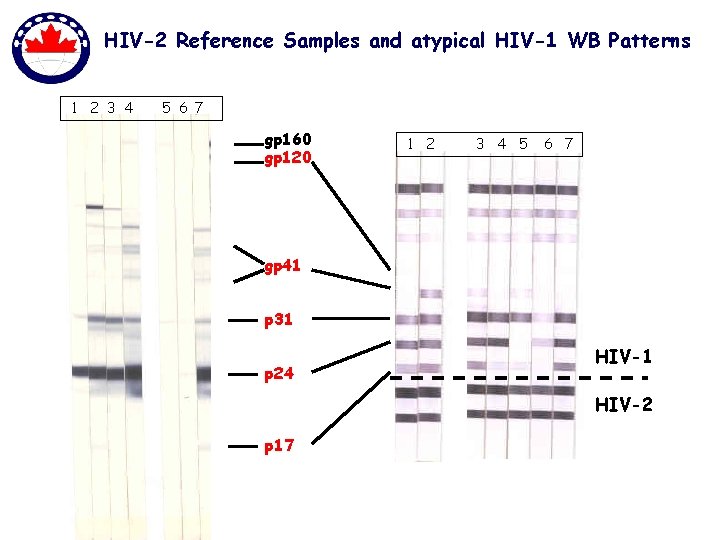
HIV-2 Reference Samples and atypical HIV-1 WB Patterns 1 2 3 4 5 6 7 gp 160 gp 120 1 2 3 4 5 6 7 gp 41 p 31 p 24 HIV-1 HIV-2 p 17

HIV WB Quality ISSUES-Canada Site Lot # Date Problems Sask 927304 Negative control had a few faint bands, slightly above where you would expect Sep 04 the gp 160 and slightly below p 18 Alberta 931422 Apr 05 Halifax 928718 & 928634 NFLD 929713 May 05 935405 Faint band at p 18 on the negative Jan 06 control Quebec Weak positive control strips of Biorad Mar-Apr Western Blot kit that the 120 band is not 05 visible after the maximum incubation time

Methods • BIO-RAD Wb vs INNOGenetics HIV 1/2 Score • > 1000 samples, (2002 -2007) • n=823 (77 -pos; 746 -neg), final status known/WBLIA results • Neg samples (746) n=472, LIA-neg/Wb-neg n=232*, LIA-neg/Wb-ind n=42, LIA-ind/Wb-ind [23], LIA-ind/Wb-neg [15], LIA-pos/Wb-pos/neg/ind [4]
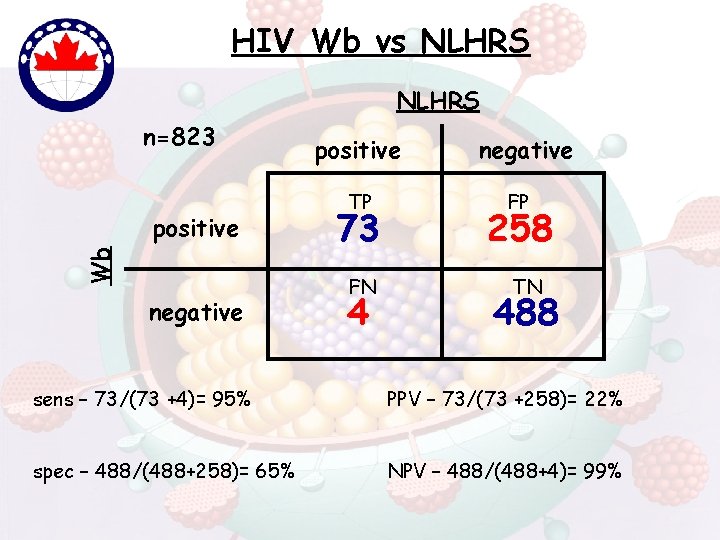
HIV Wb vs NLHRS n=823 Wb positive negative positive TP 73 FN 4 negative FP 258 TN 488 sens – 73/(73 +4)= 95% PPV – 73/(73 +258)= 22% spec – 488/(488+258)= 65% NPV – 488/(488+4)= 99%
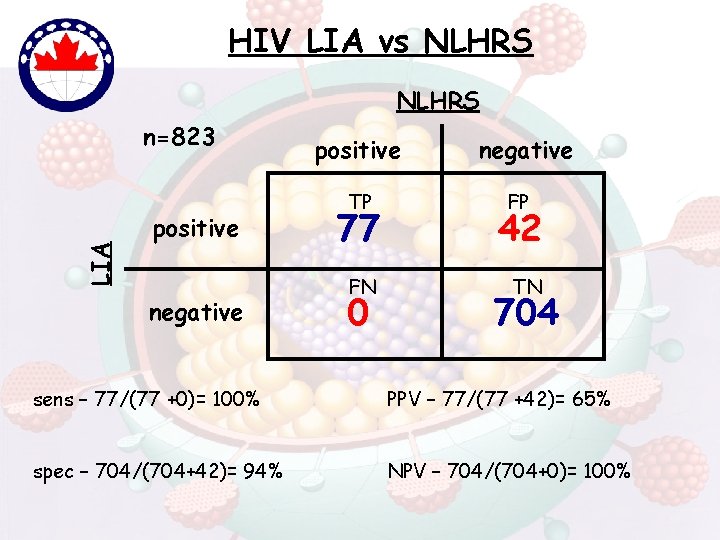
HIV LIA vs NLHRS LIA n=823 positive negative positive TP negative FP 77 42 FN TN 0 704 sens – 77/(77 +0)= 100% PPV – 77/(77 +42)= 65% spec – 704/(704+42)= 94% NPV – 704/(704+0)= 100%
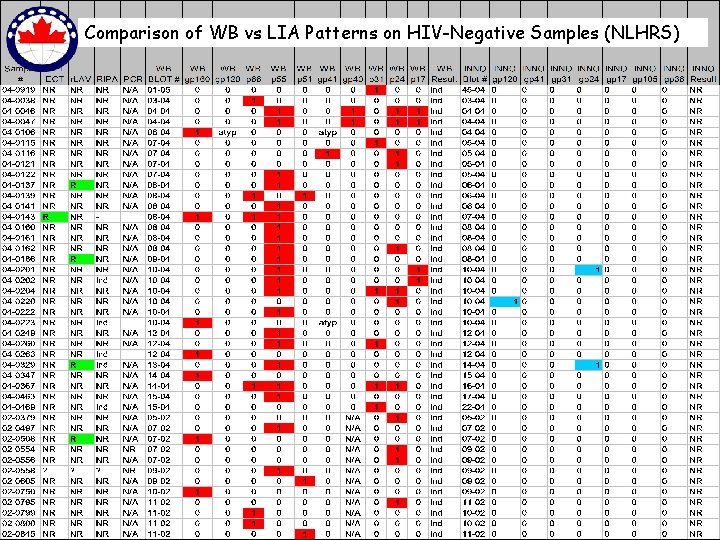
Comparison of WB vs LIA Patterns on HIV-Negative Samples (NLHRS)
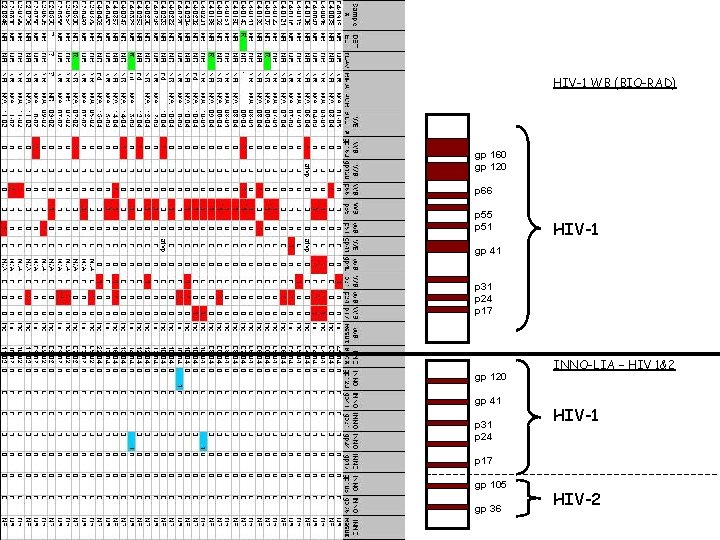
HIV-1 WB (BIO-RAD) gp 160 gp 120 p 66 p 55 p 51 HIV-1 gp 41 p 31 p 24 p 17 gp 120 gp 41 p 31 p 24 INNO-LIA – HIV 1&2 HIV-1 p 17 gp 105 gp 36 HIV-2
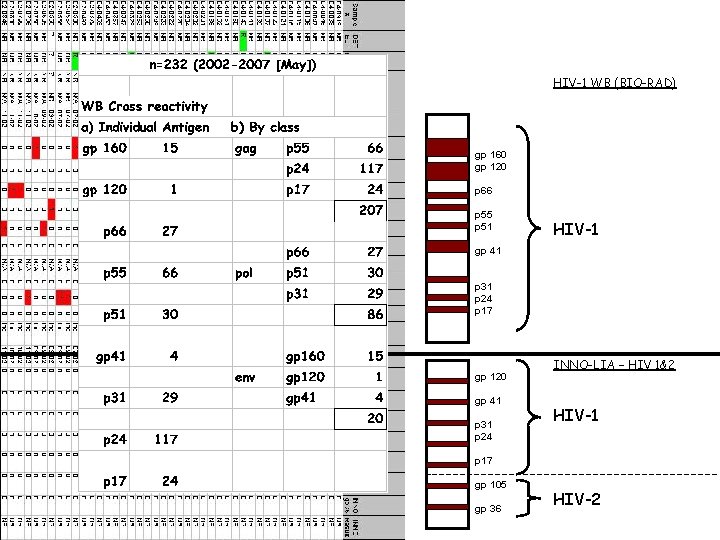
HIV-1 WB (BIO-RAD) gp 160 gp 120 p 66 p 55 p 51 HIV-1 gp 41 p 31 p 24 p 17 gp 120 gp 41 p 31 p 24 INNO-LIA – HIV 1&2 HIV-1 p 17 gp 105 gp 36 HIV-2

HIV-1 WB (BIO-RAD) gp 160 gp 120 p 66 p 55 p 51 HIV-1 gp 41 p 31 p 24 p 17 gp 120 gp 41 p 31 p 24 INNO-LIA – HIV 1&2 HIV-1 p 17 gp 105 gp 36 HIV-2


Living with the Wb-Improvements • Wb still most widely used (licensed) confirm tool • Known for years (89. 3% [266/298]; BIORad; p 20) • Review/Change interpretative criteria…Harmonize international criterias • Combine wb interpretative criteria eg: CDC/ASTPHLD + WHO [Tebourski et al, 2004] • License new tests (eg. LIA) – patent issues ? • LIA – test of choice in NLHRS sens (100% vs 95%), spec (94% vs 65%) PPV (65% vs 22%), NPV (100% vs 99%)
OMIT SLIDE ? % False Positives % Prevalence

OMIT SLIDE ? ? ? Predictive Values Positive predictive value (PPV) Negative predictive value (NPV) PPV = TP/TP +FP (x 100) NPV = TN/TN+FN (x 100) How accurately a test predicts the presence or absence of analyte (disease) ! (i) sensitivity - incidence of true positives in those with disease (ii) specificity - incidence of true of negatives in those without disease (iii) prevalence - number of actual cases (of disease) in the population. Galen RS and Gambino SR. Beyond Normality: The Predictive Value and Efficiency of Medical Diagnosis. New York: Wiley, 1975.
- Slides: 18