Comparison of RTV vs Cobi GSUS216 0114 Study

Comparison of RTV vs Cobi § GS-US-216 -0114
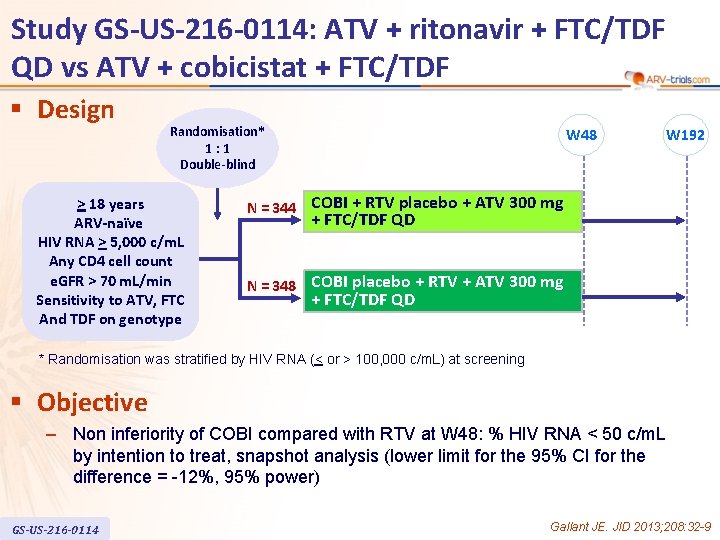
Study GS-US-216 -0114: ATV + ritonavir + FTC/TDF QD vs ATV + cobicistat + FTC/TDF § Design Randomisation* 1: 1 Double-blind > 18 years ARV-naïve HIV RNA > 5, 000 c/m. L Any CD 4 cell count e. GFR > 70 m. L/min Sensitivity to ATV, FTC And TDF on genotype W 48 W 192 N = 344 COBI + RTV placebo + ATV 300 mg + FTC/TDF QD N = 348 COBI placebo + RTV + ATV 300 mg + FTC/TDF QD * Randomisation was stratified by HIV RNA (< or > 100, 000 c/m. L) at screening § Objective – Non inferiority of COBI compared with RTV at W 48: % HIV RNA < 50 c/m. L by intention to treat, snapshot analysis (lower limit for the 95% CI for the difference = -12%, 95% power) GS-US-216 -0114 Gallant JE. JID 2013; 208: 32 -9
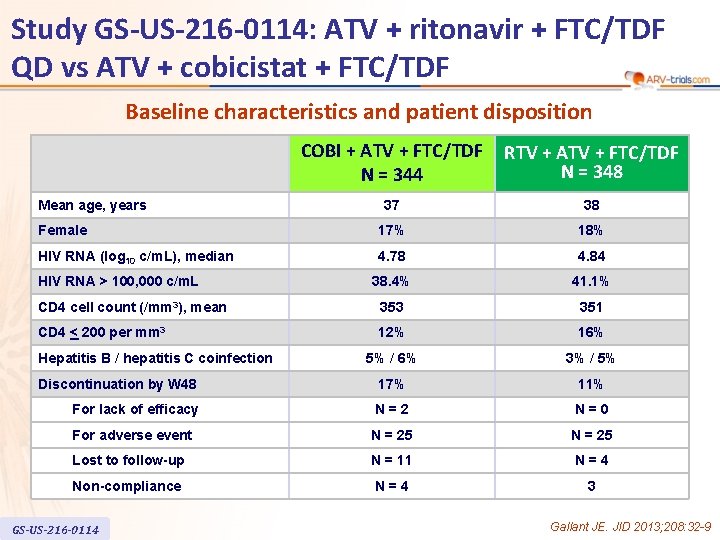
Study GS-US-216 -0114: ATV + ritonavir + FTC/TDF QD vs ATV + cobicistat + FTC/TDF Baseline characteristics and patient disposition COBI + ATV + FTC/TDF N = 344 RTV + ATV + FTC/TDF N = 348 37 38 Female 17% 18% HIV RNA (log 10 c/m. L), median 4. 78 4. 84 38. 4% 41. 1% CD 4 cell count (/mm 3), mean 353 351 CD 4 < 200 per mm 3 12% 16% 5% / 6% 3% / 5% Discontinuation by W 48 17% 11% For lack of efficacy N=2 N=0 For adverse event N = 25 Lost to follow-up N = 11 N=4 Non-compliance N=4 3 Mean age, years HIV RNA > 100, 000 c/m. L Hepatitis B / hepatitis C coinfection GS-US-216 -0114 Gallant JE. JID 2013; 208: 32 -9
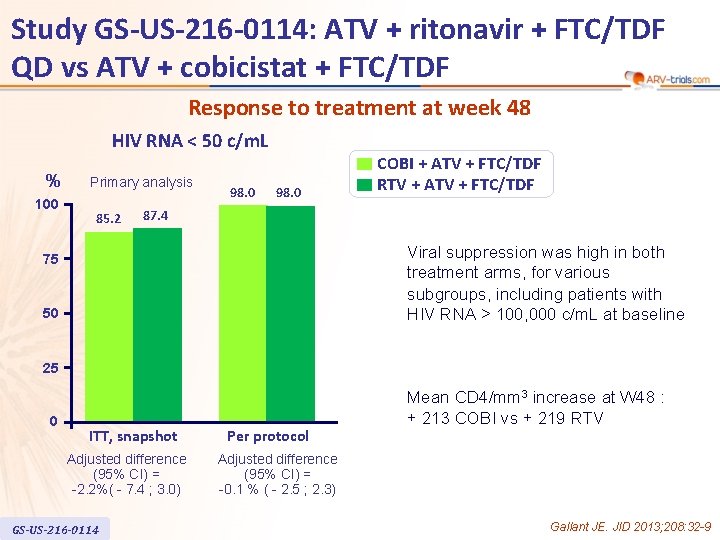
Study GS-US-216 -0114: ATV + ritonavir + FTC/TDF QD vs ATV + cobicistat + FTC/TDF Response to treatment at week 48 HIV RNA < 50 c/m. L % 100 Primary analysis 85. 2 98. 0 COBI + ATV + FTC/TDF RTV + ATV + FTC/TDF 87. 4 Viral suppression was high in both treatment arms, for various subgroups, including patients with HIV RNA > 100, 000 c/m. L at baseline 75 50 25 0 ITT, snapshot Adjusted difference (95% CI) = -2. 2%( - 7. 4 ; 3. 0) GS-US-216 -0114 Per protocol Mean CD 4/mm 3 increase at W 48 : + 213 COBI vs + 219 RTV Adjusted difference (95% CI) = -0. 1 % ( - 2. 5 ; 2. 3) Gallant JE. JID 2013; 208: 32 -9
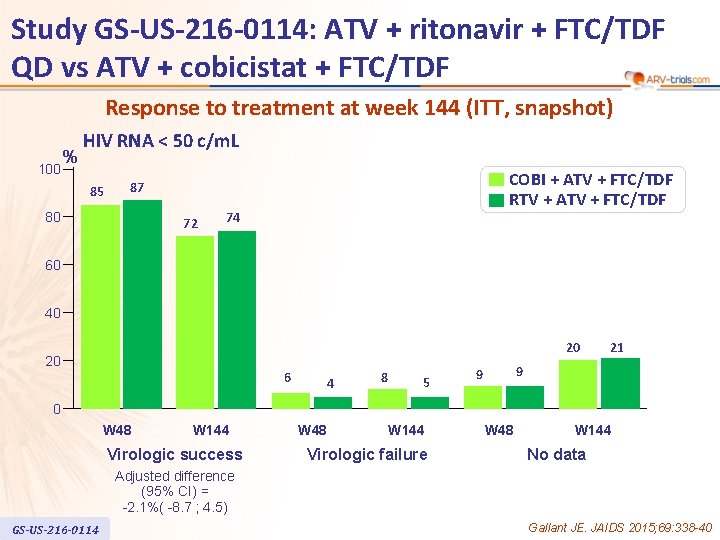
Study GS-US-216 -0114: ATV + ritonavir + FTC/TDF QD vs ATV + cobicistat + FTC/TDF Response to treatment at week 144 (ITT, snapshot) 100 % HIV RNA < 50 c/m. L 85 COBI + ATV + FTC/TDF RTV + ATV + FTC/TDF 87 80 72 74 60 40 20 20 6 4 8 5 21 9 9 0 W 48 W 144 Virologic success W 48 W 144 Virologic failure W 48 W 144 No data Adjusted difference (95% CI) = -2. 1%( -8. 7 ; 4. 5) GS-US-216 -0114 Gallant JE. JAIDS 2015; 69: 338 -40
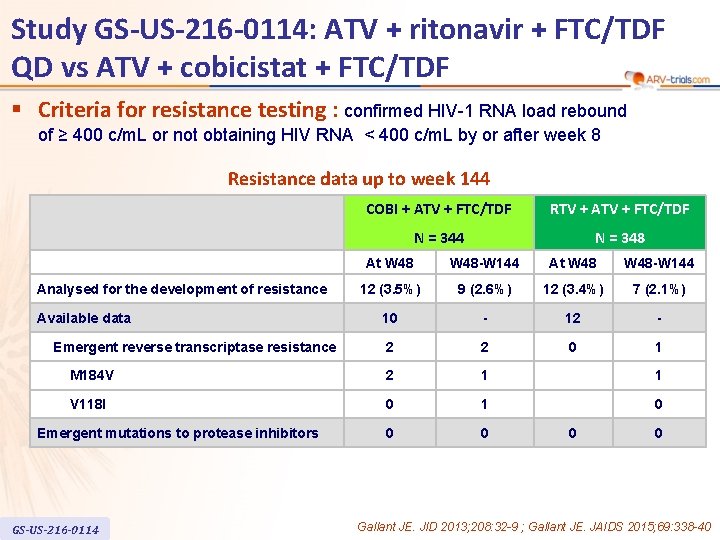
Study GS-US-216 -0114: ATV + ritonavir + FTC/TDF QD vs ATV + cobicistat + FTC/TDF § Criteria for resistance testing : confirmed HIV-1 RNA load rebound of ≥ 400 c/m. L or not obtaining HIV RNA < 400 c/m. L by or after week 8 Resistance data up to week 144 COBI + ATV + FTC/TDF RTV + ATV + FTC/TDF N = 344 N = 348 At W 48 -W 144 12 (3. 5%) 9 (2. 6%) 12 (3. 4%) 7 (2. 1%) 10 - 12 - 2 2 0 1 M 184 V 2 1 1 V 118 I 0 1 0 0 0 Analysed for the development of resistance Available data Emergent reverse transcriptase resistance Emergent mutations to protease inhibitors GS-US-216 -0114 0 0 Gallant JE. JID 2013; 208: 32 -9 ; Gallant JE. JAIDS 2015; 69: 338 -40
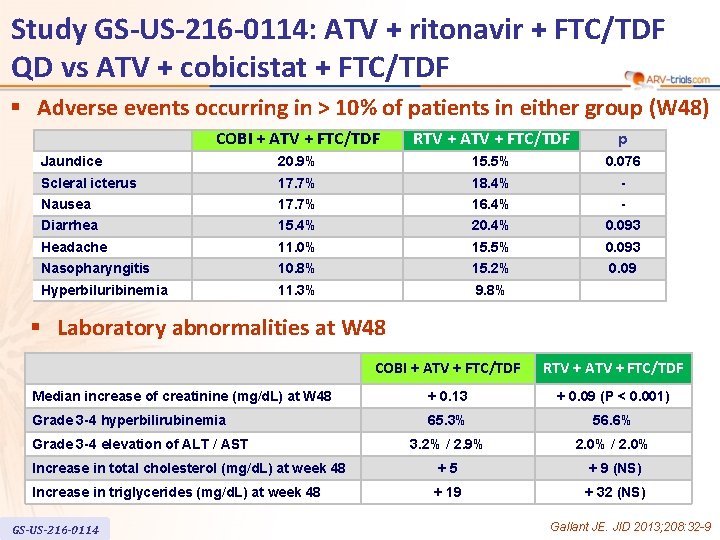
Study GS-US-216 -0114: ATV + ritonavir + FTC/TDF QD vs ATV + cobicistat + FTC/TDF § Adverse events occurring in > 10% of patients in either group (W 48) COBI + ATV + FTC/TDF RTV + ATV + FTC/TDF p Jaundice 20. 9% 15. 5% 0. 076 Scleral icterus 17. 7% 18. 4% - Nausea 17. 7% 16. 4% - Diarrhea 15. 4% 20. 4% 0. 093 Headache 11. 0% 15. 5% 0. 093 Nasopharyngitis 10. 8% 15. 2% 0. 09 Hyperbiluribinemia 11. 3% 9. 8% § Laboratory abnormalities at W 48 COBI + ATV + FTC/TDF RTV + ATV + FTC/TDF Median increase of creatinine (mg/d. L) at W 48 + 0. 13 + 0. 09 (P < 0. 001) Grade 3 -4 hyperbilirubinemia 65. 3% 56. 6% 3. 2% / 2. 9% 2. 0% / 2. 0% Increase in total cholesterol (mg/d. L) at week 48 +5 + 9 (NS) Increase in triglycerides (mg/d. L) at week 48 + 19 + 32 (NS) Grade 3 -4 elevation of ALT / AST GS-US-216 -0114 Gallant JE. JID 2013; 208: 32 -9
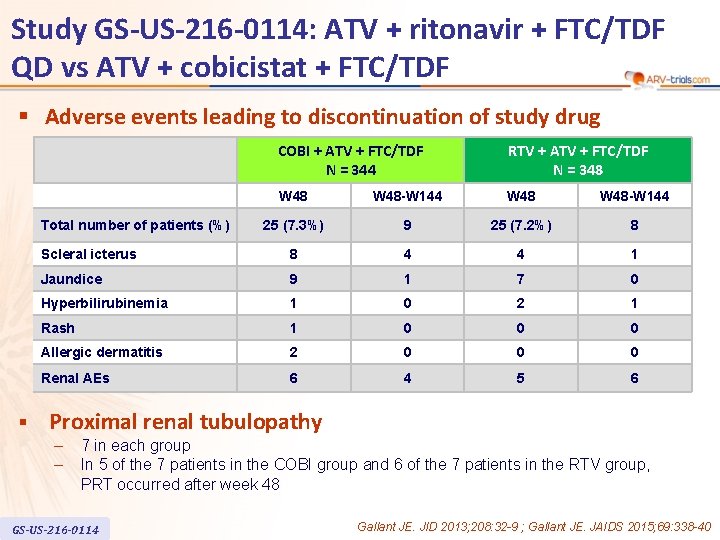
Study GS-US-216 -0114: ATV + ritonavir + FTC/TDF QD vs ATV + cobicistat + FTC/TDF § Adverse events leading to discontinuation of study drug COBI + ATV + FTC/TDF N = 344 RTV + ATV + FTC/TDF N = 348 W 48 -W 144 25 (7. 3%) 9 25 (7. 2%) 8 Scleral icterus 8 4 4 1 Jaundice 9 1 7 0 Hyperbilirubinemia 1 0 2 1 Rash 1 0 0 0 Allergic dermatitis 2 0 0 0 Renal AEs 6 4 5 6 Total number of patients (%) § Proximal renal tubulopathy – – 7 in each group In 5 of the 7 patients in the COBI group and 6 of the 7 patients in the RTV group, PRT occurred after week 48 GS-US-216 -0114 Gallant JE. JID 2013; 208: 32 -9 ; Gallant JE. JAIDS 2015; 69: 338 -40
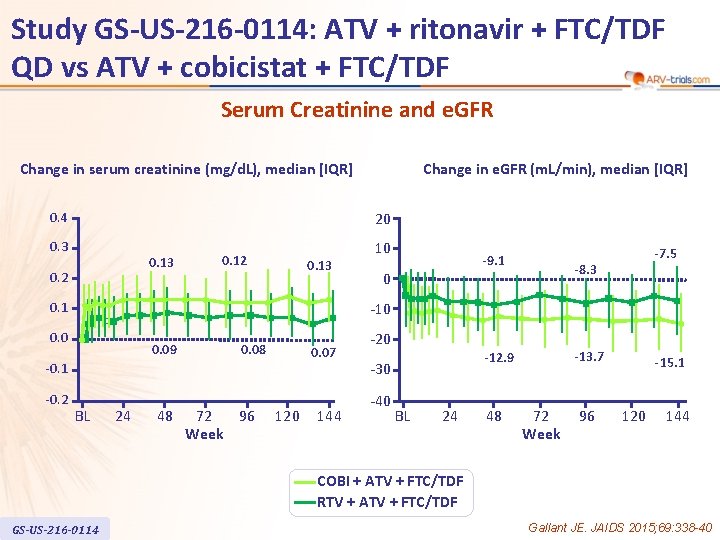
Study GS-US-216 -0114: ATV + ritonavir + FTC/TDF QD vs ATV + cobicistat + FTC/TDF Serum Creatinine and e. GFR Change in serum creatinine (mg/d. L), median [IQR] 20 0. 4 0. 3 0. 13 0. 2 0. 13 10 -9. 1 0 -7. 5 -8. 3 -10 0. 1 0. 09 0. 08 0. 07 -0. 1 -0. 2 Change in e. GFR (m. L/min), median [IQR] BL 24 48 72 96 Week 120 144 -20 -40 -13. 7 -12. 9 -30 BL 24 48 72 Week 96 -15. 1 120 144 COBI + ATV + FTC/TDF RTV + ATV + FTC/TDF GS-US-216 -0114 Gallant JE. JAIDS 2015; 69: 338 -40
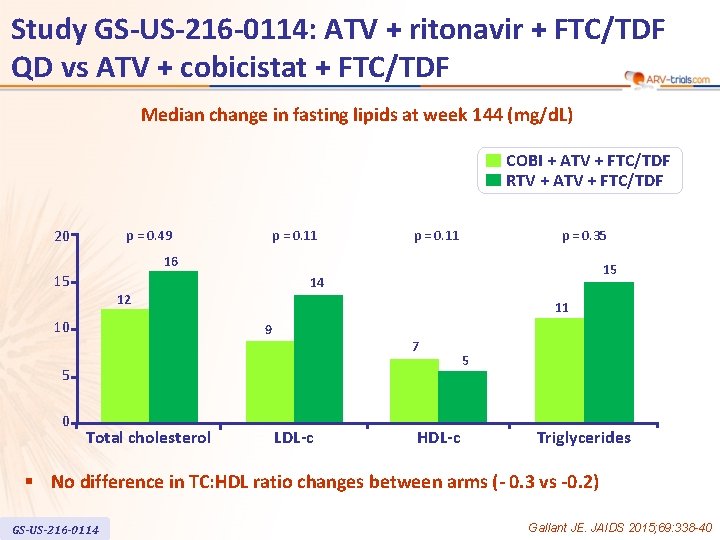
Study GS-US-216 -0114: ATV + ritonavir + FTC/TDF QD vs ATV + cobicistat + FTC/TDF Median change in fasting lipids at week 144 (mg/d. L) COBI + ATV + FTC/TDF RTV + ATV + FTC/TDF 20 p = 0. 49 p = 0. 11 p = 0. 35 16 15 15 14 12 10 11 9 7 5 0 Total cholesterol LDL-c HDL-c 5 Triglycerides § No difference in TC: HDL ratio changes between arms (- 0. 3 vs -0. 2) GS-US-216 -0114 Gallant JE. JAIDS 2015; 69: 338 -40

Study GS-US-216 -0114: ATV + ritonavir + FTC/TDF QD vs ATV + cobicistat + FTC/TDF § Summary – COBI was non inferior to RTV in combination with ATV plus FTC/TDF up to week 144 • Both regimens achieved high rates of virologic success – Safety and tolerability profiles of the 2 regimens were comparable – Once-daily COBI is a safe and effective pharmaco-enhancer of the protease inhibitor ATV – Renal safety was comparable between treatment arms • Discontinuation due to renal events was 2. 9% in the COBI group and 3. 2% in the RTV group at W 144 • Proximal renal tubulopathy occurred in 7 vs 7 patients (2. 0%) • A small, but significantly higher with COBI, increase in creatinine was seen in both groups, as early as week 2, with peak at week 8, and stabilization through 144 weeks GS-US-216 -0114 Gallant JE. JID 2013; 208: 32 -9 ; Gallant JE. JAIDS 2015; 69: 338 -40
- Slides: 11