Comparative Effectiveness of AngiotensinConverting Enzyme Inhibitors andor Angiotensin

Comparative Effectiveness of Angiotensin-Converting Enzyme Inhibitors and/or Angiotensin II-Receptor Blockers Added to Standard Medical Therapy for Treating Patients With Stable Ischemic Heart Disease and Preserved Left Ventricular Systolic Function Prepared for: Agency for Healthcare Research and Quality (AHRQ) www. ahrq. gov

Outline of Material Background Process for developing the Comparative Effectiveness Review (CER) Questions addressed in the CER Results for each question in the CER Informed decisionmaking for physicians and patients

Health Impact of Cardiovascular Disease in the United States (1) An estimated 80 million American adults (1 in 3) have one or more forms of cardiovascular disease. 38. 1 million are estimated to be age 60 or older. 16. 8 million adults have ischemic heart disease, also known as coronary heart disease. Miniño AM, et al. Natl Vital Stat Rep 2006; 54(19): 1 -49; Lloyd-Jones D, et al. Circulation 2009; 119: e 21 -181.

Health Impact of Cardiovascular Disease in the United States (2) Every year, cardiovascular disease has accounted for more deaths than any other single cause or group of causes of death in the United States since 1900 (excluding 1918). Nearly 2, 400 Americans die of cardiovascular disease each day, an average of one death every 37 seconds. Miniño AM, et al. Natl Vital Stat Rep 2006; 54(19): 1 -49; Lloyd-Jones D, et al. Circulation 2009; 119: e 21 -181.

Characteristics of Stable Ischemic Heart Disease Atherosclerosis reduces the supply of blood and oxygen to the myocardium. Symptoms range from asymptomatic ischemic episodes to severely debilitating symptoms. Disease can manifest in large vessels or as diffuse microvascular disease. There is an increased risk of acute coronary syndrome. Gibbons RJ, et al. J Am Coll Cardiol 2003; 41: 159 -68; Fraker TD, Fihn SD. J Am Coll Cardiol 2007; 50: 2264 -74; Coleman CI, et al. AHRQ Comparative Effectiveness Review No. 18. October 2009.

Standard Therapy for Stable Ischemic Heart Disease Standard therapy that can reduce cardiovascular events: Single or dual antiplatelet therapy Statins β-blockers Aggressive modification of risk factors Standard therapy that can help with symptoms: Fast-acting nitrates Negative chronotropic agents (β-blockers; nondihydropyridine calcium channel blockers) Vasodilators (calcium channel blockers; long-acting nitrates) Gibbons RJ, et al. J Am Coll Cardiol 2002; 41: 159 -68; Fraker TD, Fihn SD. J Am Coll Cardiol 2007; 50: 2264 -74.

Rationale for Additional Therapies for Patients With Stable Ischemic Heart Disease and Preserved Left Ventricular Systolic Function Despite standard medical therapy, these patients continue to experience considerable morbidity and mortality. ACEIs and ARBs have established benefit in patients with heart failure and left ventricular dysfunction. The evidence for prophylactic use of ACEIs and ARBs in patients without heart failure and with preserved left ventricular systolic function is less clear. ACEI = angiotensin-converting enzyme inhibitor; ARB = angiotensin II-receptor blocker.

Guidelines for the Use of ACEIs, ARBs, or Both to Treat Patients With Cardiac Disease (1) American College of Cardiology and American Heart Association guidelines state that ACEIs can be used in addition to standard therapy in patients who have: Chronic heart failure. Myocardial infarction and left ventricular dysfunction (defined as a left ventricular ejection fraction (LVEF) ≤ 40%). ARBs are reserved for patients who cannot tolerate ACEIs. In patients with heart failure, combining an ACEI with an ARB may provide additional benefit over an ACEI alone. Baker WL, et al. Ann Intern Med 2009 Oct 19. [Epub ahead of print]; Hunt SA, et al. Circulation 2005; 112: e 154 -235; Pfeffer MA, et al. N Engl J Med 2003; 149: 1893 -906; Smith SC, et al. Circulation 2006; 113: 2363 -72.

Guidelines for the Use of ACEIs, ARBs, or Both to Treat Patients With Cardiac Disease (2) Clinical trials have been conducted to evaluate the use of ACEIs, ARBs, or both in patients with stable ischemic heart disease but without heart failure or left ventricular systolic dysfunction (patients with an LVEF >40%). Baker WL, et al. Ann Intern Med 2009 Oct 19. [Epub ahead of print]; Hunt SA, et al. Circulation 2005; 112: e 154 -235; Pfeffer MA, et al. N Engl J Med 2003; 149: 1893 -906; Smith SC, et al. Circulation 2006; 113: 2363 -72.
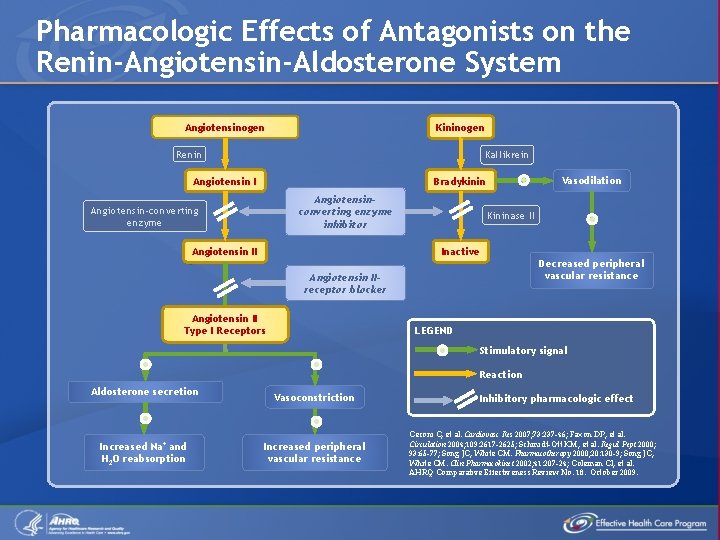
Pharmacologic Effects of Antagonists on the Renin-Angiotensin-Aldosterone System Angiotensinogen Kininogen Renin Kallikrein Angiotensin I Vasodilation Bradykinin Angiotensinconverting enzyme inhibitor Angiotensin-converting enzyme Angiotensin II Kininase II Inactive Angiotensin IIreceptor blocker Angiotensin II Type I Receptors Decreased peripheral vascular resistance LEGEND Stimulatory signal Reaction Aldosterone secretion Increased Na+ and H 2 O reabsorption Vasoconstriction Increased peripheral vascular resistance Inhibitory pharmacologic effect Ceconi C, et al. Cardiovasc Res 2007; 73: 237 -46; Faxon DP, et al. Circulation 2004; 109: 2617 -2625; Schmidt-Ott KM, et al. Regul Pept 2000; 93: 65 -77; Song JC, White CM. Pharmacotherapy 2000; 20: 130 -9; Song JC, White CM. Clin Pharmacokinet 2002; 41: 207 -24; Coleman CI, et al. AHRQ Comparative Effectiveness Review No. 18. October 2009.

The CER Development Process The topic was nominated in a public process. A specialized Technical Expert Panel guided selection of the clinical questions that the CER would address. The research for the CER was based on a well-defined systematic literature review process. The methods used for data collection and meta-analysis followed the Methods Reference Guide for Effectiveness and Comparative Effectiveness Reviews. The draft CER was made available for public comment and underwent a rigorous peer-review process to improve the final product. The complete final report is available online at http: //effectivehealthcare. ahrq. gov/ehc/products/57/335/ bodyfinal. pdf.

Rating the Strength of Evidence From the CER: A Modification of the GRADE Methodology The GRADE system of the Cochrane Collaboration was used to rate the strength of evidence resulting from the CER but with a slight modification. The modified system uses four domains — risk of bias, consistency, directness, and precision — for assessment. For the purposes of the CER, the strength of evidence pertaining to each key question was classified into three broad categories or grades: High There are consistent results from good quality clinical trials. Further research is very unlikely to change the conclusions. Moderate Findings are supported, but further research could change the conclusions. Low There are very few clinical trials, or existing trials are flawed. AHRQ. Methods Reference Guide for Effectiveness and Comparative Effectiveness Reviews, Version 1. 0; Brozek J, et al. GRADEpro Version 3. 2 for Windows. Coleman CI, et al. AHRQ Comparative Effectiveness Review No. 18. October 2009.

Comparative Effectiveness Review: Outcomes of Interest End Points: Benefits End Points: Harms Total mortality Hyperkalemia Cardiovascular (CV) death Cough Nonfatal myocardial infarction (MI) Angioedema Stroke Rash Composite endpoint (CV death, nonfatal MI, stroke) Blood dyscrasias Revascularization Quality-of-life measures Hypotension Syncope Withdrawal from trial Coleman CI, et al. AHRQ Comparative Effectiveness Review No. 18. October 2009.

Clinical Questions Addressed by the Comparative Effectiveness Review for Stable Ischemic Heart Disease and Preserved Left Ventricular Systolic Function The comparative effectiveness of different combination treatments: ACEI or ARB + Standard Therapy Versus Standard Therapy Alone ACEI + ARB + Standard Therapy Versus ACEI + Standard Therapy ACEI or ARB + Standard Therapy Versus Standard Therapy Alone Close to a Revascularization Procedure The benefits and harms associated with each treatment modality. The differences in the benefits or harms between various subpopulations of patients. Coleman CI, et al. AHRQ Comparative Effectiveness Review No. 18. October 2009.

Results of Trials Evaluating the Addition of an ACEI or an ARB to Standard Therapy for Stable Ischemic Heart Disease and Preserved Left Ventricular Systolic Function Adding an ACEI or an ARB can provide additional clinical benefits for some patients. Adding an ACEI may increase the risk of cough, syncope, or hyperkalemia. Adding an ARB may increase the risk of hyperkalemia. Adding an ACEI does not impact cardiovascular mortality in patients with end-stage renal disease and left ventricular hypertrophy. Coleman CI, et al. AHRQ Comparative Effectiveness Review No. 18. October 2009
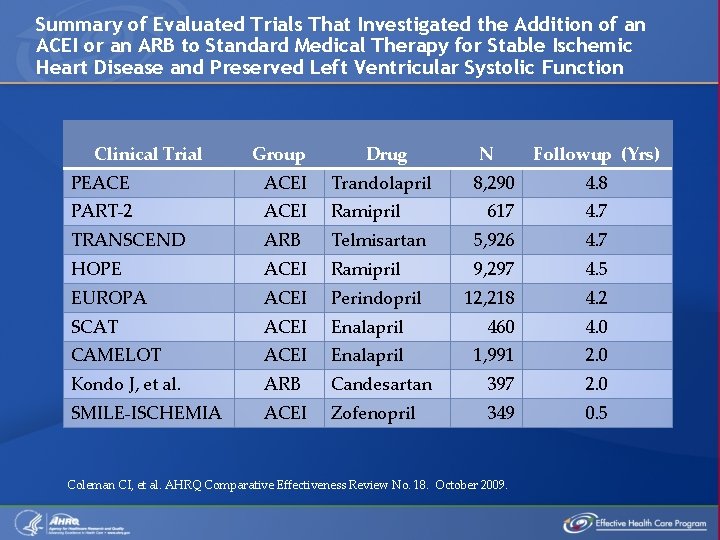
Summary of Evaluated Trials That Investigated the Addition of an ACEI or an ARB to Standard Medical Therapy for Stable Ischemic Heart Disease and Preserved Left Ventricular Systolic Function Clinical Trial Group Drug PEACE ACEI Trandolapril PART-2 ACEI Ramipril TRANSCEND ARB HOPE N Followup (Yrs) 8, 290 4. 8 617 4. 7 Telmisartan 5, 926 4. 7 ACEI Ramipril 9, 297 4. 5 EUROPA ACEI Perindopril 12, 218 4. 2 SCAT ACEI Enalapril 460 4. 0 CAMELOT ACEI Enalapril 1, 991 2. 0 Kondo J, et al. ARB Candesartan 397 2. 0 SMILE-ISCHEMIA ACEI Zofenopril 349 0. 5 Coleman CI, et al. AHRQ Comparative Effectiveness Review No. 18. October 2009.
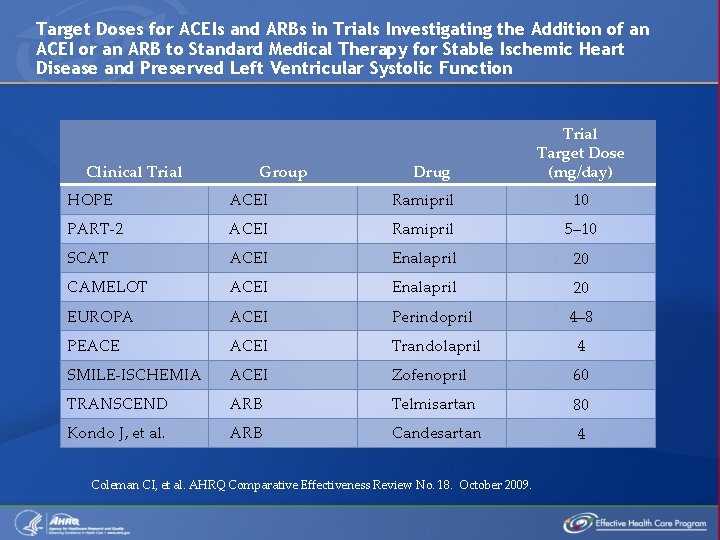
Target Doses for ACEIs and ARBs in Trials Investigating the Addition of an ACEI or an ARB to Standard Medical Therapy for Stable Ischemic Heart Disease and Preserved Left Ventricular Systolic Function Clinical Trial Group Drug Trial Target Dose (mg/day) HOPE ACEI Ramipril 10 PART-2 ACEI Ramipril 5– 10 SCAT ACEI Enalapril 20 CAMELOT ACEI Enalapril 20 EUROPA ACEI Perindopril 4– 8 PEACE ACEI Trandolapril 4 SMILE-ISCHEMIA ACEI Zofenopril 60 TRANSCEND ARB Telmisartan 80 Kondo J, et al. ARB Candesartan 4 Coleman CI, et al. AHRQ Comparative Effectiveness Review No. 18. October 2009.
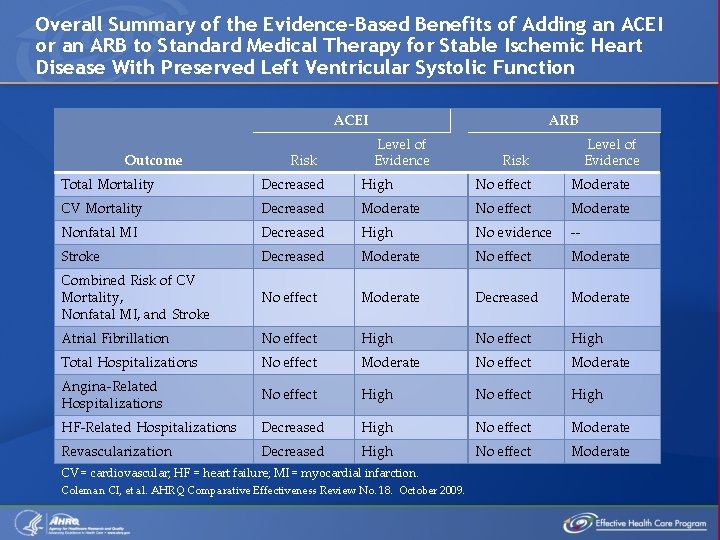
Overall Summary of the Evidence-Based Benefits of Adding an ACEI or an ARB to Standard Medical Therapy for Stable Ischemic Heart Disease With Preserved Left Ventricular Systolic Function ACEI Outcome Risk ARB Level of Evidence Risk Total Mortality Decreased High No effect Moderate CV Mortality Decreased Moderate No effect Moderate Nonfatal MI Decreased High No evidence -- Stroke Decreased Moderate No effect Moderate Combined Risk of CV Mortality, Nonfatal MI, and Stroke No effect Moderate Decreased Moderate Atrial Fibrillation No effect High Total Hospitalizations No effect Moderate Angina-Related Hospitalizations No effect High HF-Related Hospitalizations Decreased High No effect Moderate Revascularization Decreased High No effect Moderate CV = cardiovascular; HF = heart failure; MI = myocardial infarction. Coleman CI, et al. AHRQ Comparative Effectiveness Review No. 18. October 2009.
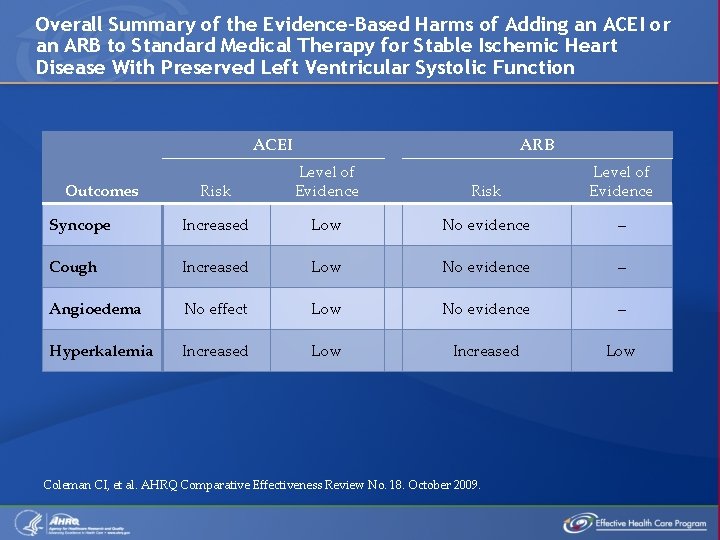
Overall Summary of the Evidence-Based Harms of Adding an ACEI or an ARB to Standard Medical Therapy for Stable Ischemic Heart Disease With Preserved Left Ventricular Systolic Function ACEI ARB Risk Level of Evidence Syncope Increased Low No evidence – Cough Increased Low No evidence – Angioedema No effect Low No evidence – Hyperkalemia Increased Low Outcomes Coleman CI, et al. AHRQ Comparative Effectiveness Review No. 18. October 2009.
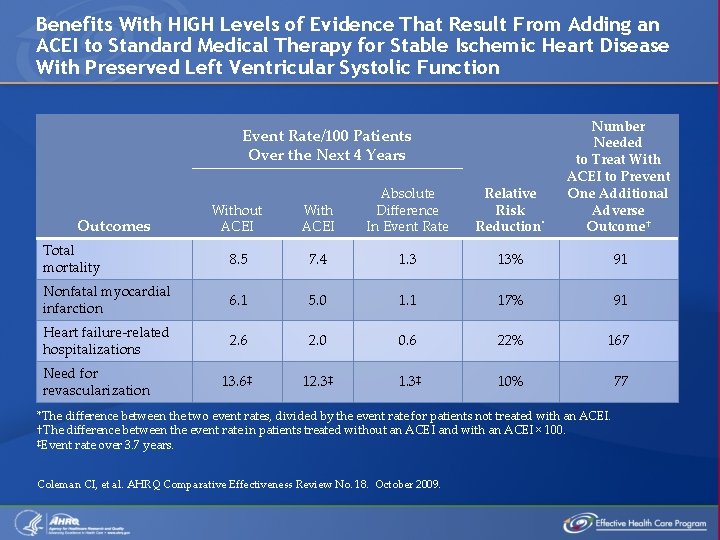
Benefits With HIGH Levels of Evidence That Result From Adding an ACEI to Standard Medical Therapy for Stable Ischemic Heart Disease With Preserved Left Ventricular Systolic Function Relative Risk Reduction* Number Needed to Treat With ACEI to Prevent One Additional Adverse Outcome† Event Rate/100 Patients Over the Next 4 Years Without ACEI With ACEI Absolute Difference In Event Rate Total mortality 8. 5 7. 4 1. 3 13% 91 Nonfatal myocardial infarction 6. 1 5. 0 1. 1 17% 91 Heart failure-related hospitalizations 2. 6 2. 0 0. 6 22% 167 13. 6‡ 12. 3‡ 10% 77 Outcomes Need for revascularization *The difference between the two event rates, divided by the event rate for patients not treated with an ACEI. †The difference between the event rate in patients treated without an ACEI and with an ACEI × 100. ‡Event rate over 3. 7 years. Coleman CI, et al. AHRQ Comparative Effectiveness Review No. 18. October 2009.

Benefits With HIGH Levels of Evidence That Result From Adding an ARB to Standard Medical Therapy for Stable Ischemic Heart Disease With Preserved Left Ventricular Systolic Function* Event Rate/100 Patients Over the Next 5 Years Outcomes Combined risk of death from heartrelated cause, suffering a nonfatal myocardial infarction, or having a stroke Without ARB With ARB Absolute Difference In Event Rate 14. 8 13. 0 1. 8 Relative Risk Reduction† Number Needed to Treat With an ARB to Prevent One Additional Adverse Outcome ‡ 12% 56 * Only the data from the Telmisartan Randomized Assessment Study in ACE Intolerant Subjects with Cardiovascular Disease (TRANSCEND) trial were used in the analysis. †The difference between the two event rates, divided by the event rate for patients not treated with an ARB. ‡The difference between the event rate in patients treated without an ARB and with an ARB × 100. Coleman CI, et al. AHRQ Comparative Effectiveness Review No. 18. October 2009.

Results of Trials That Evaluated the Addition of an ACEI/ARB Combination Versus an ACEI Alone to Standard Medical Therapy for Stable Ischemic Heart Disease With Preserved Left Ventricular Systolic Function The Ongoing Telmisartan Alone and in Combination with Ramipril Global Endpoint Trial (ONTARGET) was the only trial that investigated the addition of an ACEI/ARB combination to standard medical therapy versus standard medical therapy plus an ACEI alone. There was no evidence of any greater clinical benefit with the addition of the ACEI/ARB combination as opposed to an ACEI alone. There was evidence that patients who received the ACEI/ARB combination were at increased risk for adverse events. Coleman CI, et al. AHRQ Comparative Effectiveness Review No. 18. October 2009.

Overall Summary of the Evidence-Based Benefits and Harms of Adding an ACEI/ARB Combination Versus an ACEI Alone to Standard Medical Therapy for Stable Ischemic Heart Disease With Preserved Left Ventricular Systolic Function There were no clinical benefits for the ACEI/ARB (ramipril + telmisartan) combination (Moderate Level of Evidence). The risk of harms was higher in the ACEI/ARB combination treatment group (Moderate Level of Evidence). ACEI Alone (N = 8, 576) ACEI/ARB Combination (N = 8, 502) ACEI/ARB Combination Versus ACEI Alone n n (P value) 2, 099 2, 495 < 0. 001 149 406 < 0. 001 Syncope 15 29 0. 03 Renal impairment 60 94 < 0. 001 Diarrhea 12 39 < 0. 001 Outcomes Total number of discontinuations Hypotension Modified from Yusuf S, et al. New Engl J Med 2008; 358: 1547 -59.

Results of Trials Evaluating the Addition of an ACEI or an ARB to Standard Medical Therapy (SMT) Versus SMT Alone Close to a Revascularization Procedure Seven trials met the inclusion criteria for this analysis. There was no clinical benefit from adding an ACEI or an ARB to standard medical therapy in close proximity to a revascularization procedure. There was an increased risk of adverse events. Coleman CI, et al. AHRQ Comparative Effectiveness Review No. 18. October 2009.

Characteristics of Trials Evaluating the Addition of an ACEI or an ARB to Standard Medical Therapy (SMT) Versus SMT Alone Close to a Revascularization Procedure Trial Treatment Group Drug MARCATOR ACEI given the same day post-PTCA cilazapril APRES ACEI given 5 to 7 days after CABG or 1 to 2 days after PTCA ramipril Kondo J, et al. ACEI given after coronary stenting quinapril PARIS ACEI given ≤ 48 hours after coronary stenting quinapril QUIET ACEI given 12 to 72 hours after angioplasty quinapril IMAGINE ACEI given ≤ 7 days after CABG quinapril AACHEN ARB given 7 to 14 days before coronary stenting candesartan CABG = coronary artery bypass grafting surgery; PTCA = percutaneous transluminal coronary angioplasty. Coleman CI, et al. AHRQ Comparative Effectiveness Review No. 18. October 2009.

Analysis of Trials That Tested the Addition of an ACEI or an ARB to Standard Medical Therapy (SMT) Versus SMT Alone Close to a Revascularization Procedure Overall, there were no clinical benefits to adding ACEIs or ARBs to standard medical therapy close to a revascularization procedure. There was an increased risk for these harms: Increased Risk of Harms Drug Level of Evidence Hypotension ACEI Moderate Need for subsequent revascularization ACEI or ARB High Coleman CI, et al. AHRQ Comparative Effectiveness Review No. 18. October 2009.
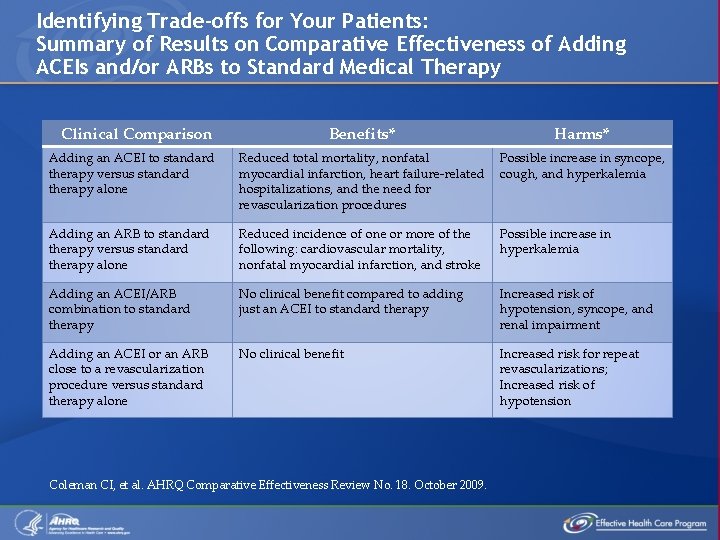
Identifying Trade-offs for Your Patients: Summary of Results on Comparative Effectiveness of Adding ACEIs and/or ARBs to Standard Medical Therapy Clinical Comparison Benefits* Harms* Adding an ACEI to standard therapy versus standard therapy alone Reduced total mortality, nonfatal myocardial infarction, heart failure-related hospitalizations, and the need for revascularization procedures Possible increase in syncope, cough, and hyperkalemia Adding an ARB to standard therapy versus standard therapy alone Reduced incidence of one or more of the following: cardiovascular mortality, nonfatal myocardial infarction, and stroke Possible increase in hyperkalemia Adding an ACEI/ARB combination to standard therapy No clinical benefit compared to adding just an ACEI to standard therapy Increased risk of hypotension, syncope, and renal impairment Adding an ACEI or an ARB close to a revascularization procedure versus standard therapy alone No clinical benefit Increased risk for repeat revascularizations; Increased risk of hypotension Coleman CI, et al. AHRQ Comparative Effectiveness Review No. 18. October 2009.

Gaps in Knowledge About ACEIs and ARBs as Treatment for Stable Ischemic Heart Disease With Preserved Left Ventricular Systolic Function Meta-analyses or future clinical trials that evaluate the use of ACEIs or ARBs to treat patients who have stable ischemic heart disease and preserved left ventricular systolic function are needed to address the benefits and harms in the following patient subpopulations: Patients who are receiving antiplatelet therapy or who have a history of revascularization. Patients of different ethnicities (especially African Americans and Latinos). Patients with stenosis in the left anterior descending artery, as compared to other coronary arteries. Patients with single-vessel versus multi-vessel disease. Patients who have genetic polymorphisms of the angiotensin-converting enzyme gene or the angiotensin II type I receptor gene. Patients on different dosing intensities of ACE inhibitors or ARBs. Coleman CI, et al. AHRQ Comparative Effectiveness Review No. 18. October 2009.

Steps in the Informed Decisionmaking Process for Adding an ACEI or an ARB to Standard Medical Therapy for Stable Ischemic Heart Disease With Preserved Left Ventricular Systolic Function 1. Review the critical evidence to help your patients understand: • Relevance of risk reductions after adding an ACEI to their regimen. • Risks for cough, syncope, and hyperkalemia after adding an ACEI and what they could mean. • The option of using an ARB if intolerant to ACEIs. • The harms of adding an ACEI or an ARB too close to a revascularization procedure. 2. Explore each patient’s values by asking: 3. Encourage your patients to be involved in their care: • What worries you most about taking these types of medications? • Do you have concerns about the cost of your medicines? • Do you have any problems remembering to take your medicines? • Discuss the benefits and risks of each choice. • Talk about the impact their comorbidities will have on the decision to add an ACEI or ARB. • Discuss other things they can do to help reduce their risk of heart attack and stroke.
- Slides: 29