Commonly encountered attacks on aromatic ring substituents that
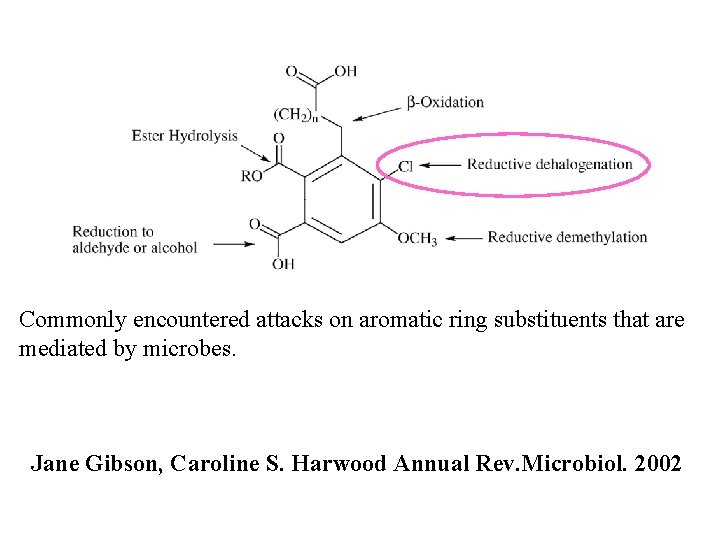
Commonly encountered attacks on aromatic ring substituents that are mediated by microbes. Jane Gibson, Caroline S. Harwood Annual Rev. Microbiol. 2002

Vinyl chloride The largest application of organochlorine chemistry is the production of vinyl chloride, to be converted into polyvinylchloride (PVC). Production of the order of 10’ 000 tons/year Chloromethanes Most low molecular weight chlorinated hydrocarbons are useful solvents. Production of the order of 1’ 000 tons/year Polychloroalkanes (PCA), polychlorobyphenyls (PCB), polybrominated diphenyl ethers (PBDE), etc. A large variety of applications from plastic production to electrical insulation, flame retardation, etc. Production of the order of 100’ 000 tons/year
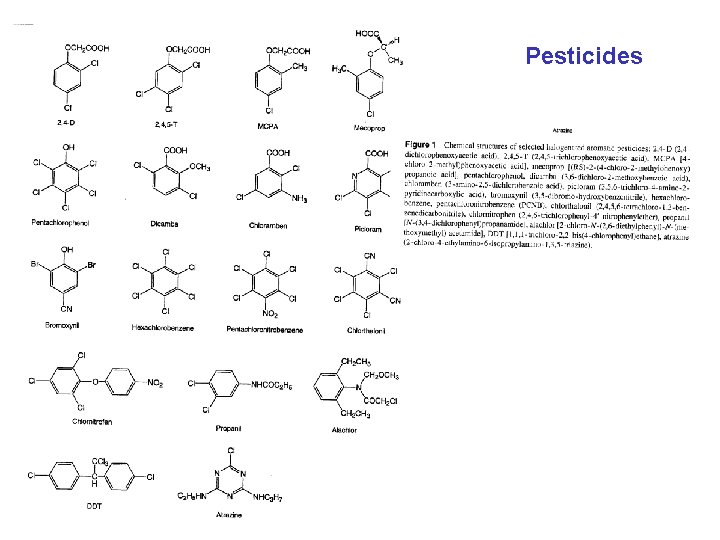
Pesticides
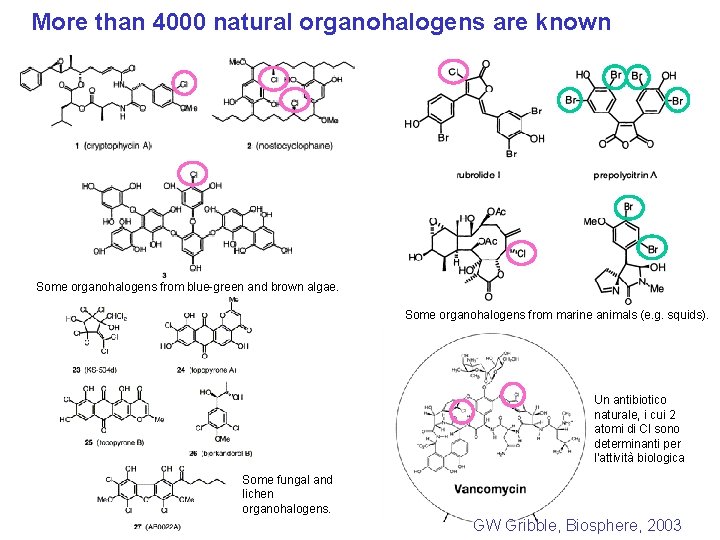
More than 4000 natural organohalogens are known Some organohalogens from blue-green and brown algae. Some organohalogens from marine animals (e. g. squids). Un antibiotico naturale, i cui 2 atomi di Cl sono determinanti per l’attività biologica Some fungal and lichen organohalogens. GW Gribble, Biosphere, 2003

Abiogenic sources Natural combustion sources such as biomass fires, volcanoes, and other geothermal processes account for a wide range of organohalogens. The early studies of volcanic gases and the presence of organohalogens therein are well documented. In 2000, a study of the volcanoes Kuju, Satsuma Iwojima, Mt. Etna, and Vulcano has revealed an extraordinarily large array of organohalogens, including 100 organochlorines, 25 organobromines, five organofluorines, and four organoiodines, most of which were new compounds. Haloalkanes have been found entombed in rocks, minerals, and shales. Thus, when rocks are crushed, as during mining operations, or as a result of weathering processes, small quantities of CH 3 Cl, CH 2 Cl 2, CHCl 3, CCl 4, CH 3 CHCl 2, Cl. CH 2 Cl, Cl 2 C=CH 2, CH 3 CH 2 Br, CF 2 Cl 2, CFCl 3, CHF 3, chlorobenzene, 1 chloronaphthalene, and other organohalogens are released (Isidorov et al. , 1993). For example, 1000 tons of silvinite ore yields 50 g of chloroform. The authors estimate that the potassium salt mining industry alone accounts for the annual liberation of 10– 15 000 tons of CHCl 3 and 100– 150 tons each of CCl 4 and CFCl 3. While there is no dispute about the emissions of chloromethane and bromomethane from biomass burning and other natural sources (Rhew et al. , 2000; Yokouchi et al. , 2000), the evidence regarding larger organohalogens, such as dioxins, has been more difficult to obtain and quantify ( Martínez et al. , 2000). However, various studies suggest that the dioxins in sediments and clays have originated from natural sources ( Fiedler et al. , 1996; Ferrario et al. , 2000), and one such obvious source is biomass burning and subsequent deposition ( Gaus et al. , 2001; Green et al. , 2001). Moreover, other studies indicate that dioxins are formed in peat and forest soil, presumably via the enzymatic oxidative dimerization of natural chlorophenols ( Silk et al. , 1997; Hoekstra et al. , 1999).
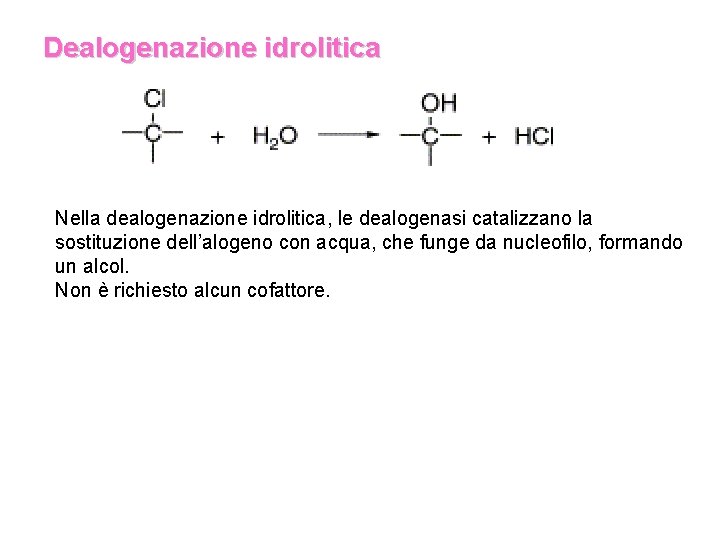
Dealogenazione idrolitica Nella dealogenazione idrolitica, le dealogenasi catalizzano la sostituzione dell’alogeno con acqua, che funge da nucleofilo, formando un alcol. Non è richiesto alcun cofattore.
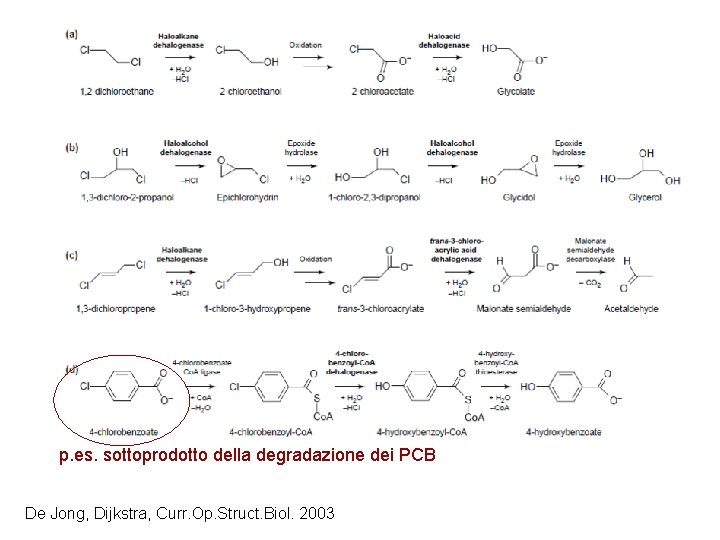
p. es. sottoprodotto della degradazione dei PCB De Jong, Dijkstra, Curr. Op. Struct. Biol. 2003
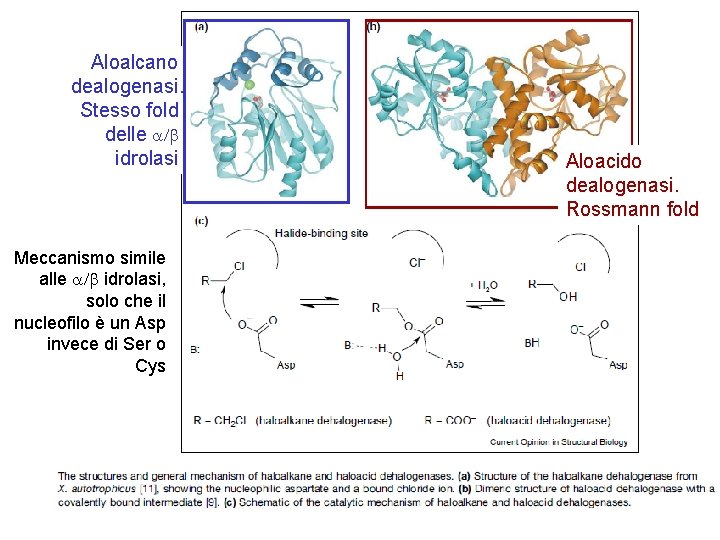
Aloalcano dealogenasi. Stesso fold delle a/b idrolasi Meccanismo simile alle a/b idrolasi, solo che il nucleofilo è un Asp invece di Ser o Cys Aloacido dealogenasi. Rossmann fold

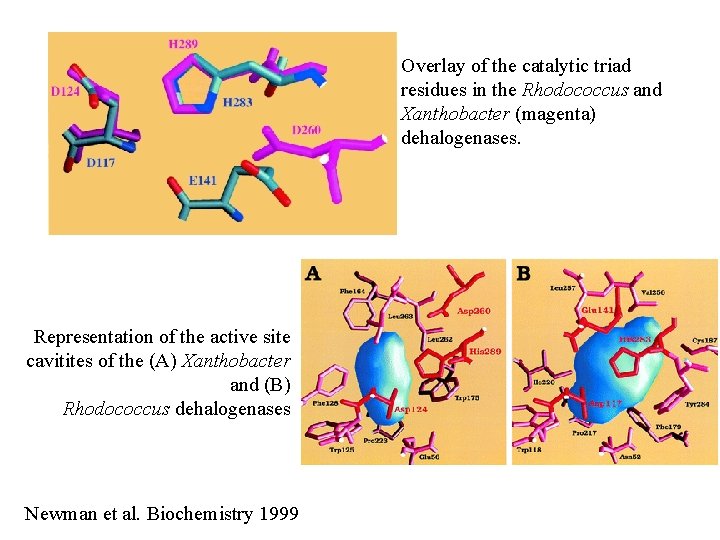
Overlay of the catalytic triad residues in the Rhodococcus and Xanthobacter (magenta) dehalogenases. Representation of the active site cavitites of the (A) Xanthobacter and (B) Rhodococcus dehalogenases Newman et al. Biochemistry 1999

Alla fine il cloroalcano va a CO 2 + HCl (c. d. mineralizzazione) policloroalcano A parità di atomi di cloro si libera maggiore energia maggiore il numero di atomi di carbonio A parità di atomi di carbonio si libera minore energia maggiore il numero di atomi di cloro La quantità di biomassa che si produce è proporzionale all’energia liberata Heath et al. J. Industrial Microbiol. Biotech. 2004
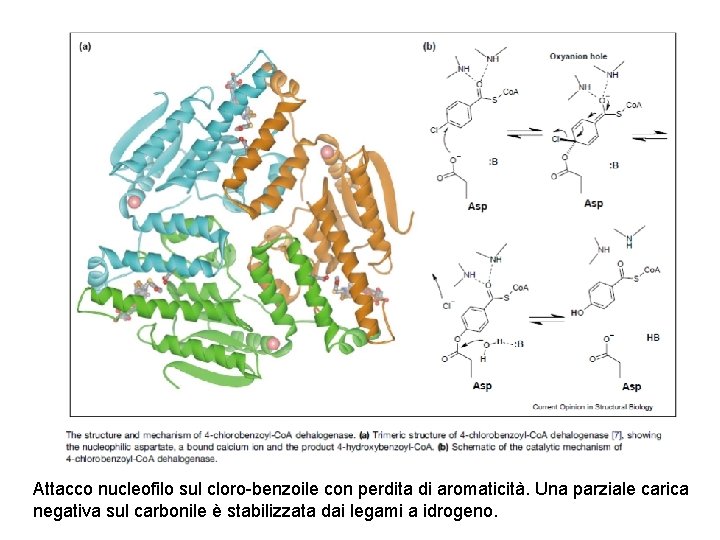
Attacco nucleofilo sul cloro-benzoile con perdita di aromaticità. Una parziale carica negativa sul carbonile è stabilizzata dai legami a idrogeno.
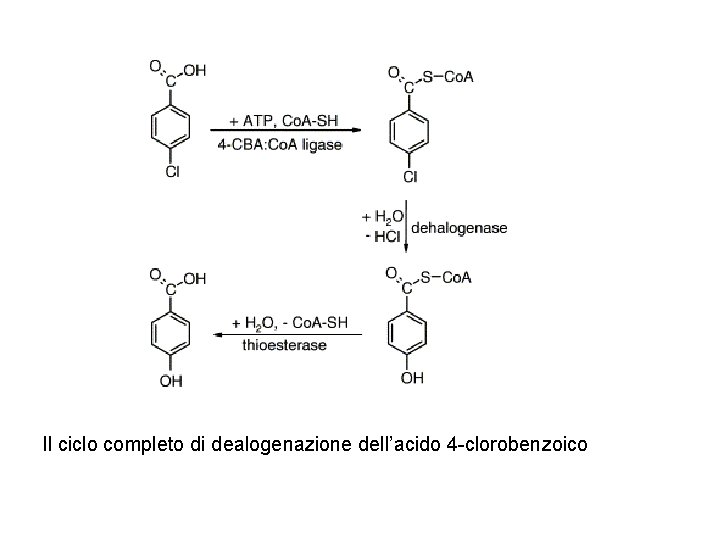
Il ciclo completo di dealogenazione dell’acido 4 -clorobenzoico
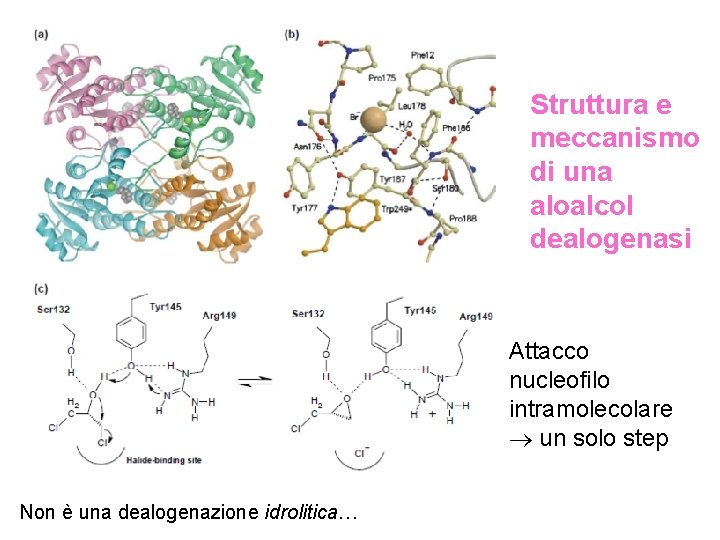
Struttura e meccanismo di una aloalcol dealogenasi Attacco nucleofilo intramolecolare un solo step Non è una dealogenazione idrolitica…
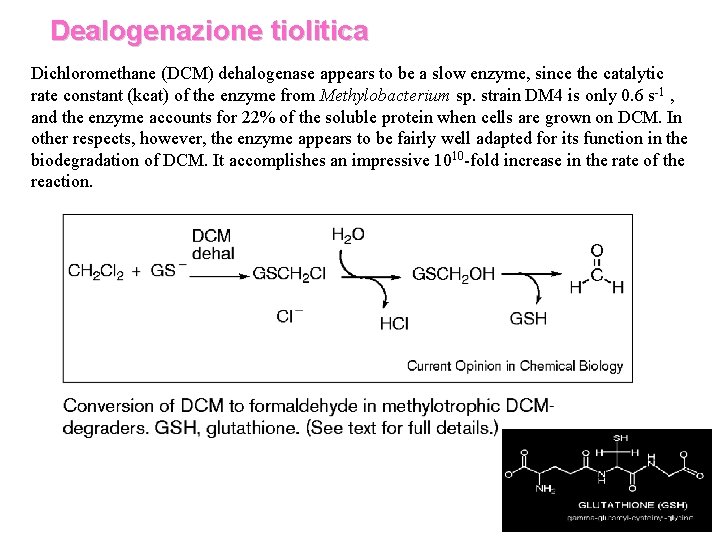
Dealogenazione tiolitica Dichloromethane (DCM) dehalogenase appears to be a slow enzyme, since the catalytic rate constant (kcat) of the enzyme from Methylobacterium sp. strain DM 4 is only 0. 6 s-1 , and the enzyme accounts for 22% of the soluble protein when cells are grown on DCM. In other respects, however, the enzyme appears to be fairly well adapted for its function in the biodegradation of DCM. It accomplishes an impressive 1010 -fold increase in the rate of the reaction.
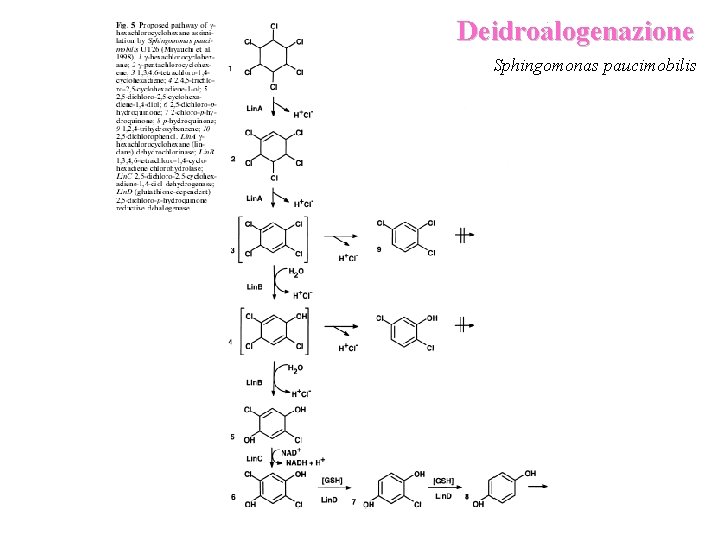
Deidroalogenazione Sphingomonas paucimobilis

Dealogenazione ossidativa – tanto migliore quanto meno atomi di alogeno sono presenti Aerobic environment: halocompounds can be substrates of mono- and dioxygenases, peroxidases, etc.
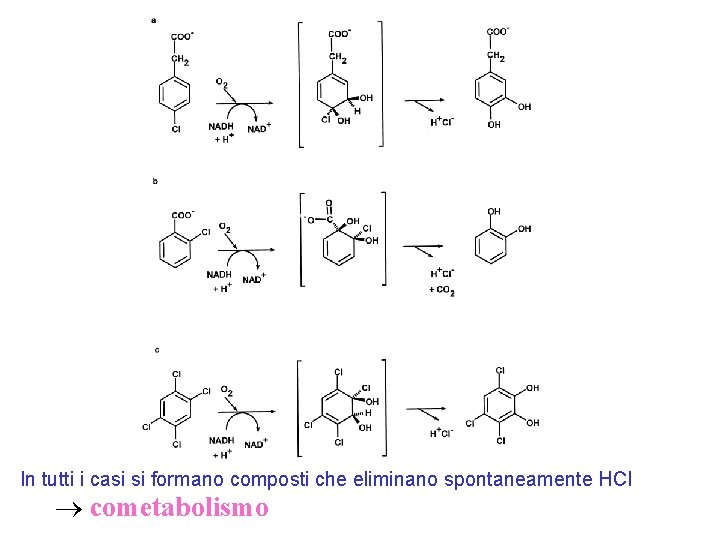
In tutti i casi si formano composti che eliminano spontaneamente HCl cometabolismo
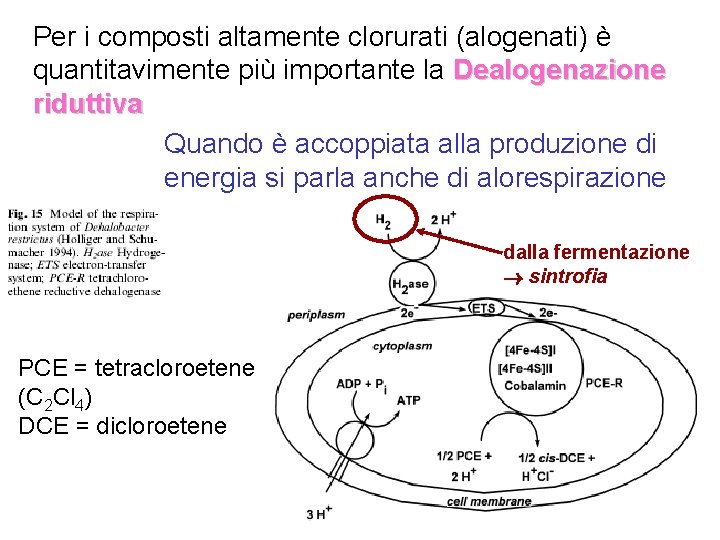
Per i composti altamente clorurati (alogenati) è quantitavimente più importante la Dealogenazione riduttiva Quando è accoppiata alla produzione di energia si parla anche di alorespirazione dalla fermentazione sintrofia PCE = tetracloroetene (C 2 Cl 4) DCE = dicloroetene

Poiché la dealogenazione riduttiva consuma H 2, essa compete con altri processi anaerobici quali • Metanogenesi • Acetogenesi • Riduzione di accettori quali nitrato, solfato, etc. Il potenziale di riduzione dipende dal numero di atomi di cloro. Si può arrivare a potenziali dell’ordine di quello del nitrato Dolfing J, Janssen DB. Biodegradation 1994
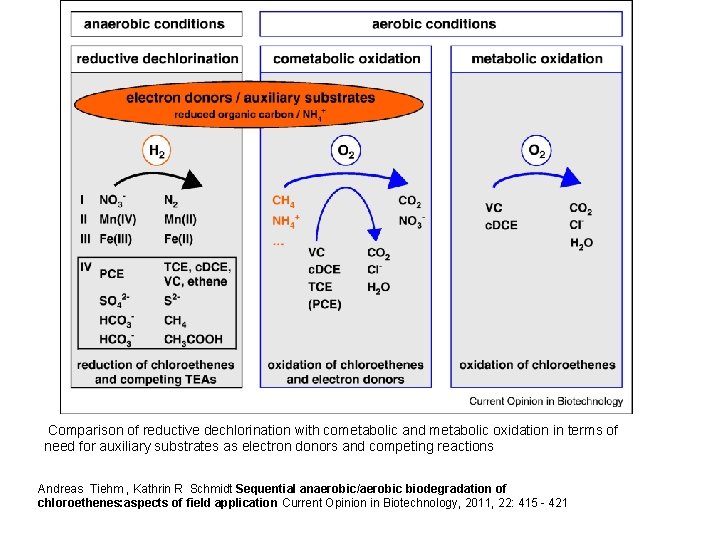
Comparison of reductive dechlorination with cometabolic and metabolic oxidation in terms of need for auxiliary substrates as electron donors and competing reactions Andreas Tiehm , Kathrin R Schmidt Sequential anaerobic/aerobic biodegradation of chloroethenes: aspects of field application Current Opinion in Biotechnology, 2011, 22: 415 - 421
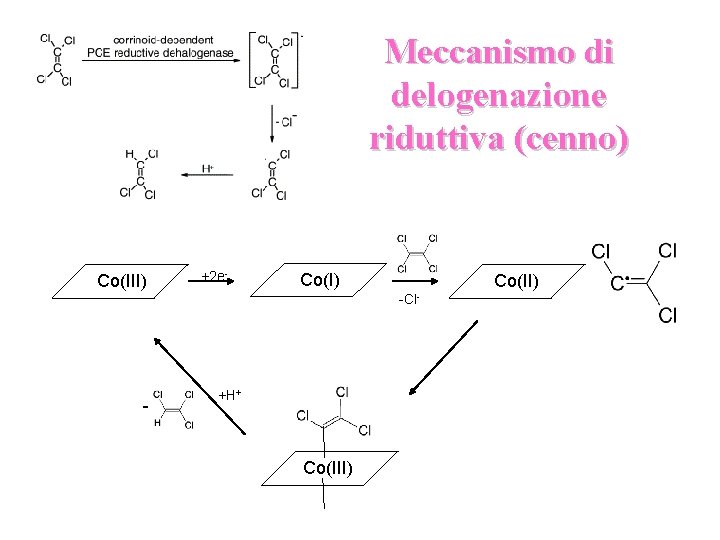
Meccanismo di delogenazione riduttiva (cenno) Co(III) +2 e- Co(I) Co(II) -Cl- - +H+ Co(III)
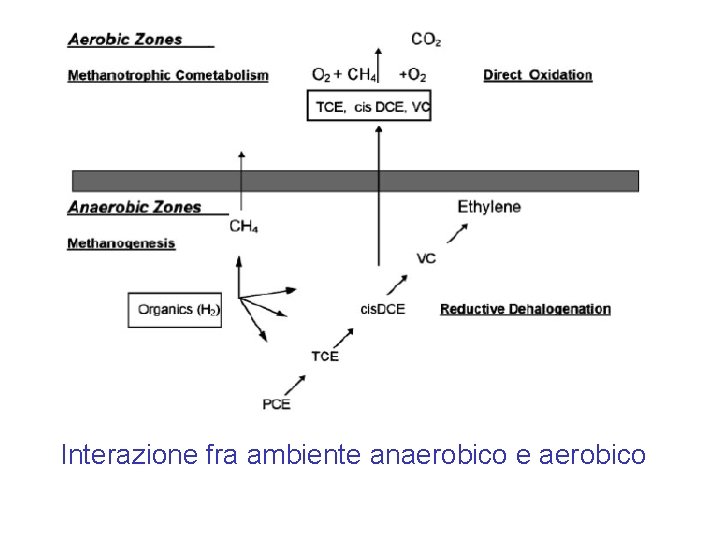
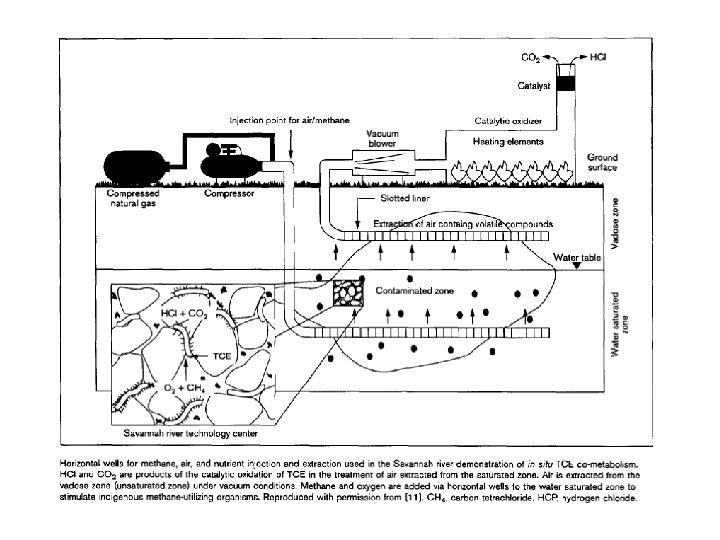
Interazione fra ambiente anaerobico e aerobico
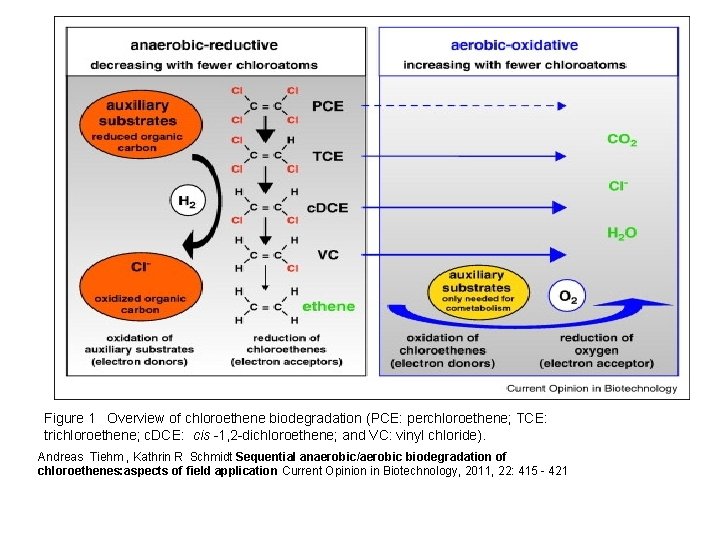
Figure 1 Overview of chloroethene biodegradation (PCE: perchloroethene; TCE: trichloroethene; c. DCE: cis -1, 2 -dichloroethene; and VC: vinyl chloride). Andreas Tiehm , Kathrin R Schmidt Sequential anaerobic/aerobic biodegradation of chloroethenes: aspects of field application Current Opinion in Biotechnology, 2011, 22: 415 - 421
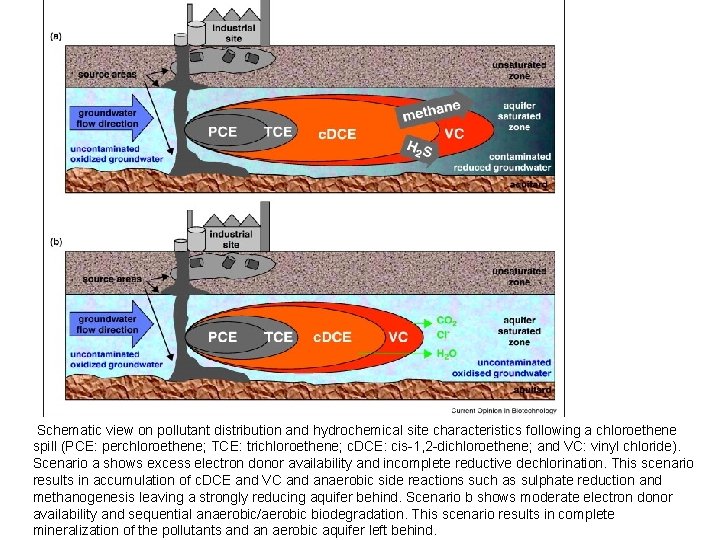
Schematic view on pollutant distribution and hydrochemical site characteristics following a chloroethene spill (PCE: perchloroethene; TCE: trichloroethene; c. DCE: cis-1, 2 -dichloroethene; and VC: vinyl chloride). Scenario a shows excess electron donor availability and incomplete reductive dechlorination. This scenario results in accumulation of c. DCE and VC and anaerobic side reactions such as sulphate reduction and methanogenesis leaving a strongly reducing aquifer behind. Scenario b shows moderate electron donor availability and sequential anaerobic/aerobic biodegradation. This scenario results in complete mineralization of the pollutants and an aerobic aquifer left behind.
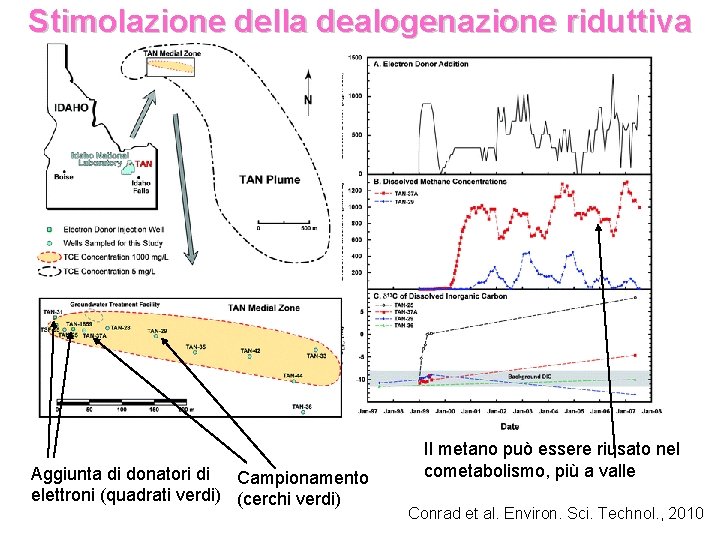
Stimolazione della dealogenazione riduttiva Aggiunta di donatori di Campionamento elettroni (quadrati verdi) (cerchi verdi) Il metano può essere riusato nel cometabolismo, più a valle Conrad et al. Environ. Sci. Technol. , 2010
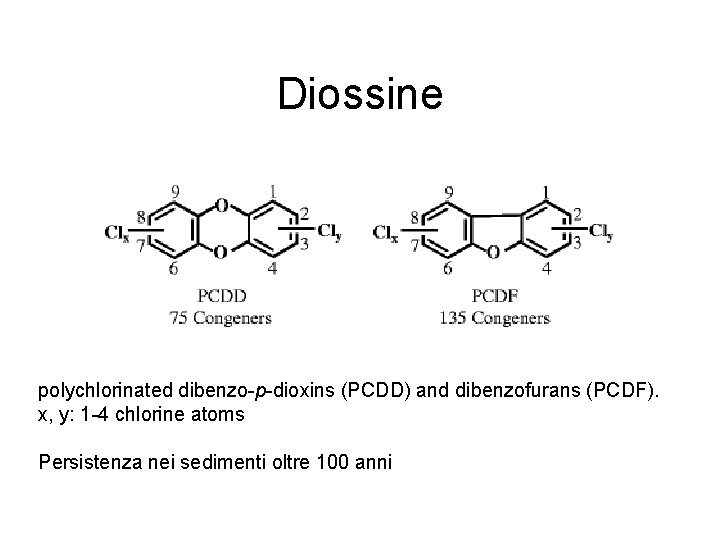
Diossine polychlorinated dibenzo-p-dioxins (PCDD) and dibenzofurans (PCDF). x, y: 1 -4 chlorine atoms Persistenza nei sedimenti oltre 100 anni
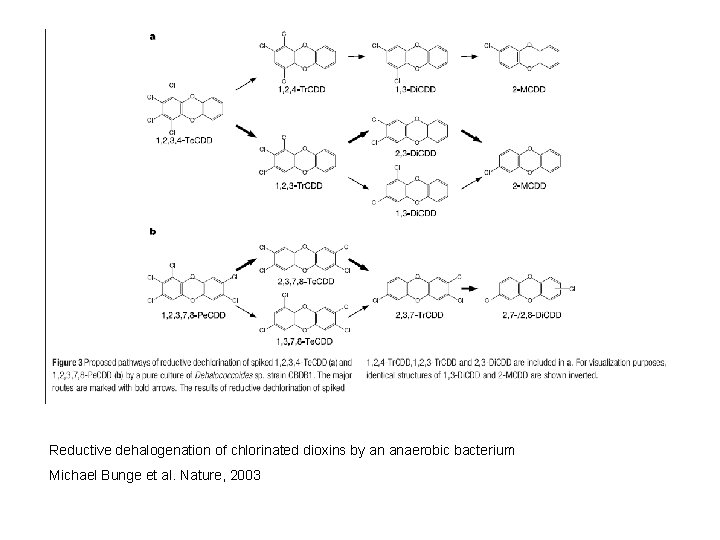
Reductive dehalogenation of chlorinated dioxins by an anaerobic bacterium Michael Bunge et al. Nature, 2003
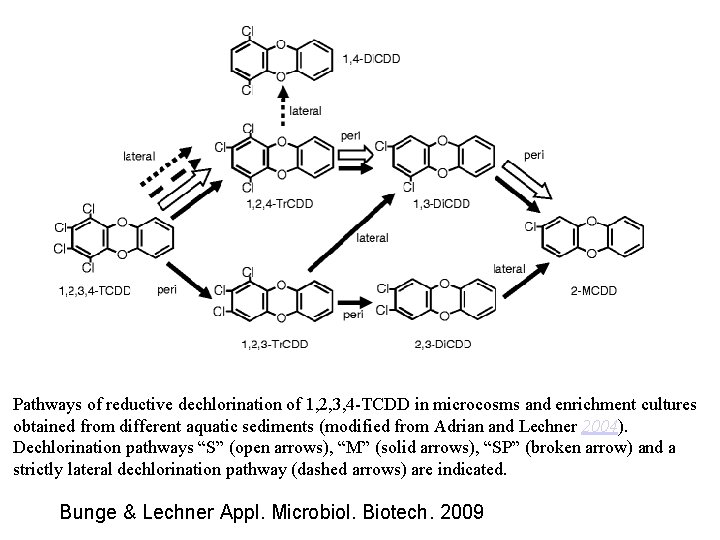
Pathways of reductive dechlorination of 1, 2, 3, 4 -TCDD in microcosms and enrichment cultures obtained from different aquatic sediments (modified from Adrian and Lechner 2004). Dechlorination pathways “S” (open arrows), “M” (solid arrows), “SP” (broken arrow) and a strictly lateral dechlorination pathway (dashed arrows) are indicated. Bunge & Lechner Appl. Microbiol. Biotech. 2009
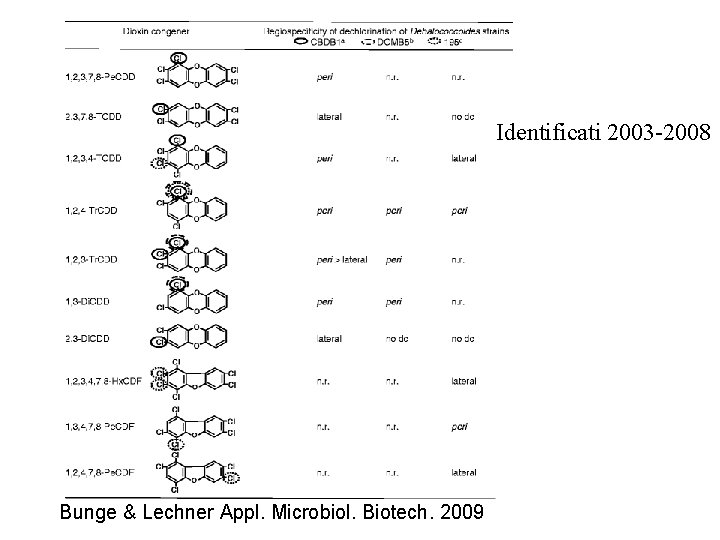
Identificati 2003 -2008 Bunge & Lechner Appl. Microbiol. Biotech. 2009
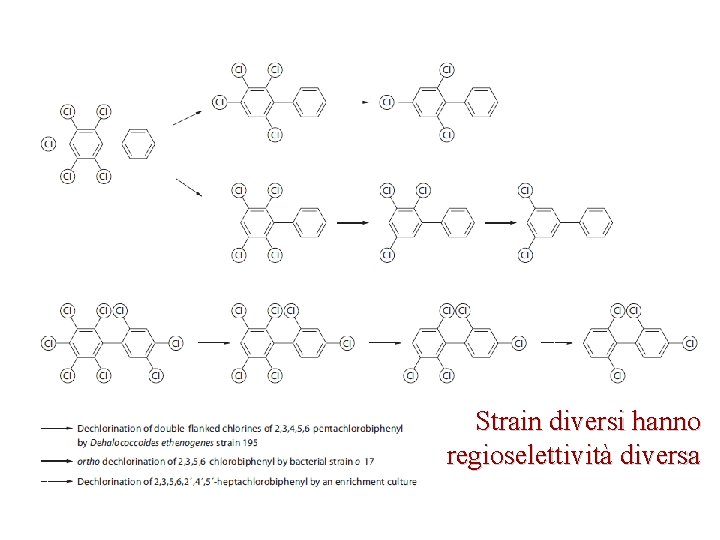
Strain diversi hanno regioselettività diversa
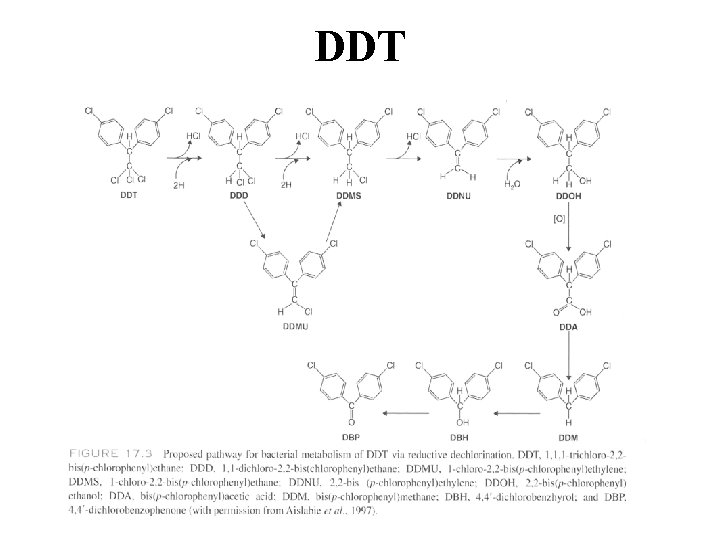
DDT
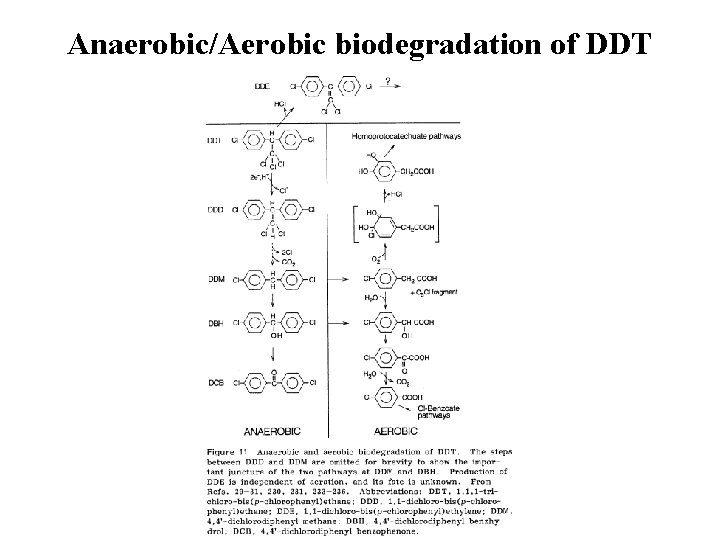
Anaerobic/Aerobic biodegradation of DDT


http: //pubs. acs. org/doi/full/10. 1021/es 903535 j
- Slides: 36