Common Technical Document Basic General Information December 2005

Common Technical Document, Basic & General Information, December 2005 The Common Technical Document (CTD) Basic & General Information Miriam Titulaer, Regulatory Affairs EU Europe

Table of Content CTD Background and Objectives 1. 2. 3. 4. 5. Definition Scope Implementation Organisation of the CTD Information on the Structure and Content of the CTD 6. Genzyme Europe BV experience 2 Europe

1. Definition ICH Initiative § Agreement on the structure and format of a well-structured presentation for applications submitted to Regulatory Authorities in Europe, US and Japan § Benefits: Simplify dossier preparation(s) Save time & resources Facilitate Regulatory review & communication Simplify exchange of Regulatory information Easier preparation of e-submissions 3 Europe

1. Definition § Warnings - - Does not provide information on the content of dossier - Does not indicate which studies & data are required for successful approval Does not harmonise Assessment Procedures (Dossier approval & amendments) 4 Europe

2. Scope Application, Procedure, Product § Applicable for all types of marketing applications: full, abridged (including line extensions), and bibliographical applications § All types of procedures Centralised, Mutual Recognition or national § Applicable for all types of products NCE’s, generics, biologicals, radiopharmaceuticals, vaccines, herbal medicinal products 5 Europe

3. Implementation § Optional July 2001: EU, FDA, Japan (Canada, Switzerland) § Mandatory July 2003: EU, Japan (Canada, Switzerland) § Highly recommended July 2003: FDA 6 Europe

3. Implementation Variations and Line extensions § In Europe, after 1 July 2003, 2003 new CTD format mandatory for all variations & line extensions applications. § There is no obligation to reformat the dossier of already authorised products into the CTD format (cross-referencing to ‘’old’’ format documentation is acceptable). § Reformatting is allowed, but not recommended for the Non-clinical and Clinical parts of the documentation. § Reformatting is recommended for the Quality part to facilitate the handling of variations and line extensions. Such reformatting must involve the complete Quality parts, including Drug Master Files (if applicable) and also include all approved variations. A signed declaration must be provided stating that the content/data of the Quality Module is identical to the currently approved Quality part, with no changes to the dossier as a result of the re-formatting. 7 Europe
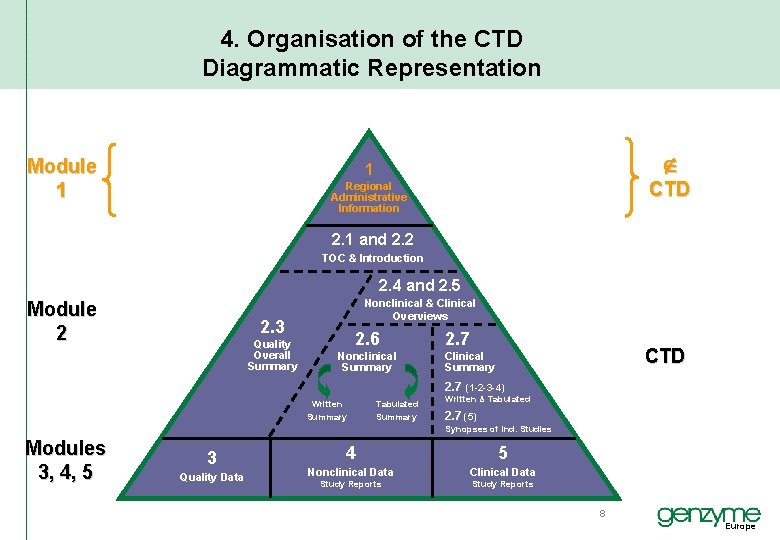
4. Organisation of the CTD Diagrammatic Representation Module 1 1 CTD Regional Administrative Information 2. 1 and 2. 2 TOC & Introduction 2. 4 and 2. 5 Module 2 Nonclinical & Clinical Overviews 2. 3 Quality Overall Summary 2. 6 Nonclinical Summary 2. 7 CTD Clinical Summary 2. 7 (1 -2 -3 -4) Written Summary Tabulated Summary Written & Tabulated 2. 7 (5) Synopses of Ind. Studies Modules 3, 4, 5 3 Quality Data 4 5 Nonclinical Data Clinical Data Study Reports 8 Europe

Common Technical Document? § The CTD defines the format of an application and not the content. Quote: “Neither the type nor extent of specific supporting data has been addressed in this guideline, and both may depend upon regional guidance. ” § For instance: Module 3, Quality information, in general EU requires less detailed equipment descriptions than US; Module 5, Clinical data, US requires inclusion of all clinical study report annexes. § Note: the implementation of the CTD is a compromise, resulting in a more elaborate dossier. 9 Europe

5. Information on the Structure and Content of the CTD Guidelines: standard format § Information on the structure and content of the CTD is provided in the rules governing medicinal products in the European Union: Notice to Applicants Volume 2 B: Presentation and content of the dossier, CTD, Edition June 2004. § It is supported by a Questions and Answers document (June 2004) which provides further information on the use of the CTD with respect to the different regulatory procedures/different forms of applications. § Additional information on the organisation of the CTD is provided in the CHMP guideline CPMP/ICH/2887/99, Topic M 4, Step 5: Common Technical Document for the Registration of Pharmaceuticals for Human Use - Organisation of CTD. 10 Europe

ICH Multidisciplinary - Adopted Guidelines § Topic M 4, Step 5 Common Technical Document for the Registration of Pharmaceuticals for Human Use: Organisation of Common Technical Document (CPMP/ICH/2887/99 - released for information February 2003): § M 4 - Organisation of the Common Technical Document Rev 2 Correction February 2004 Transmission to CPMP and Release for Information November 2003 § M 4 Quality - Quality overall Summary and CTD Quality Rev 1 § M 4 Safety - Nonclinical Summaries and Organisation of Module 4 Released for Information July 2003 § M 4 Efficacy - Clinical Overview, Clinical Summary, Sample Tables for Clinical Summary and Module V Rev 1 11 Europe

12 Europe

13 Europe

ICH Multidisciplinary - Adopted Guidelines CHMP Question and Answer Documents § M 4 Step 5 - General Questions and Answers. CPMP/ICH/5552/02, Rev 2, June 2004. § M 4 Quality - Question and Answer/Location issues For Common Technical Document for the Registration of Pharmaceuticals for Human Use - Quality CPMP/ICH/4680/02, July 2003. § M 4 Safety Step 5 - Questions and Answers. CPMP/ICH/5549/02, November 2003. § M 4 Efficacy Step 5 - Questions and Answers. CPMP/ICH/5551/02, Rev. 2, June 2004. 14 Europe

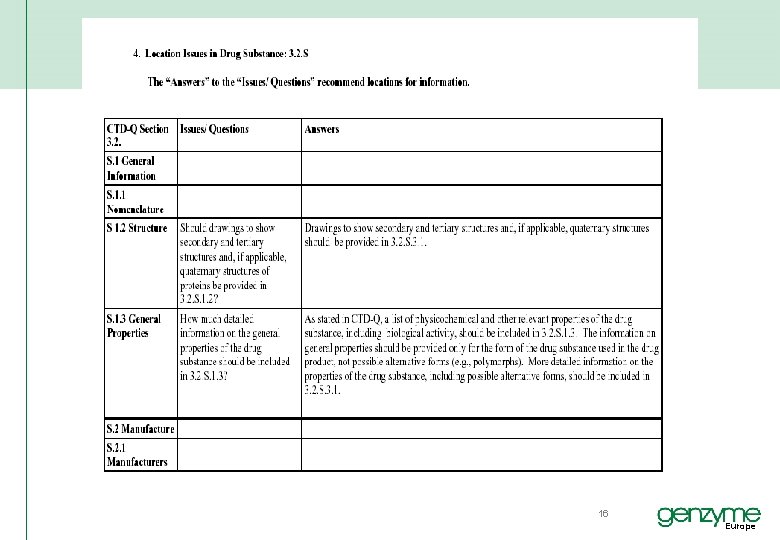
ICH M 4 QUALITY QUESTION AND ANSWER LOCATION ISSUES § Intended to provide additional guidance for the preparation of an application file in the CTD-Q format. § It should be read in conjunction with the CTD-Q guideline (Modules 2 and 3). § The document also addresses the relationship between linked CTD-Q sections for certain parameters, such as polymorphism, impurities, or particle size. § This document also clarifies location issues; that is, it indicates in which CTD-Q section(s), requested information should be placed (see section 4: Location Issues in Drug Substance, section 5: Location Issues in Drug Product, and section 6: Location Issues in Appendices). 15 Europe
16 Europe

Preparing and Organizing the CTD § Throughout the CTD, the display of information should be unambiguous and transparent, to facilitate the review of the basic data and to help a reviewer become quickly oriented to the application contents. - Text and tables should be prepared using margins that allow the document to be printed on A 4 paper. - The left-hand margin should be sufficiently large that information is not obscured through binding. - Font sizes for text and tables should be of a style and size that are large enough to be easily legible, even after photocopying. - Times New Roman, 12 -point font is recommended for narrative text. Acronyms and abbreviations should be defined the first time they are used in each module. 17 Europe

Preparing and Organizing the CTD § Guidance: M 4 Organisation of Common Technical Document, ANNEX: Granularity Document Provides guidance on: § headings in relation to document location and the section headings within those documents. § where in the CTD and e. CTD multiple documents can be located in the hierarchy. § how documents should be paginated and on what the module Table of Contents should therefore include. 18 Europe

7. Genzyme Europe BV Experience § Cholestagel®: 1 st CTD stand-alone, Application filed on August 30 th, 2002 § AT, filed on February 2004. § Myozyme (Pompe), filed on December, 2004 § Variations, line extensions, renewal applications 19 Europe

7. Genzyme Europe BV Experience CTD-format for Variations § Module 1: 1. 0 Cover Letter 1. 1 Comprehensive table of content 1. 2 Variation Application Form 1. 3 Product Information 1. 3. 1 Summary of Product Characteristics, Labelling and Package Leaflet – where appropriate 1. 4 Information about the experts: The relevant expert declaration(s) and signature(s) must be provided, corresponding to the Overview/Summary submitted in Module 2. § Module 2: As mentioned in the Variation Regulation any Type II variation should be accompanied by the relevant Overviews/Summaries updates or addenda (even if a variation is submitted at the request of the Competent Authority/CPMP). Expert details and signature are to be provided in Module 1. 4 separated from the actual Overview/Summary. Supportive data are to be included in Modules 3, 4 and/or 5 as appropriate and in accordance with the EU-CTD structure. § Documentations for Type IA/IB-Notifications also have to follow CTD-structure where applicable. 20 Europe

Genzyme Europe BV Experience CTD-format for Renewal Applications § Module 1: 1. 0 Cover Letter 1. 1 Comprehensive table of content 1. 2 Renewal Application Form with annexes 1. 3 Product Information: 1. 3. 1 SPC, Labelling and Package Leaflet 1. 3. 3 Specimen 1. 4 Information about the expert 1. 4. 1. Quality 1. 4. 2. Clinical § Module 2: 2. 3 Quality Overall Summary – (Quality Expert Statement) 2. 5 Clinical Overview (Clinical Expert Statement) § Module 5: § 5. 3. 6 Reports of Post-marketing experience. 21 Europe

7. Genzyme Europe BV Experience § Recommendation to use to the CTD format as early as possible in the global development process: - increase harmonization - save time and resources § EU: Clinical Trial Applications falling under the Clinical Trials Directive require submission of an Investigational Medicinal Product Dossier (IMPD). The IMPD contains CMC (chemistry, manufacturing control), Non-clinical and Clinical information which must be presented in the format of the CTD Module 2 Quality Overall Summary and Nonclinical and Clinical Overviews. § FDA: IND can be submitted in the CTD format, but this is not yet mandatory. 22 Europe
- Slides: 22