Common Pathway New Oral Agents Apixaban Rivaroxaban Xa
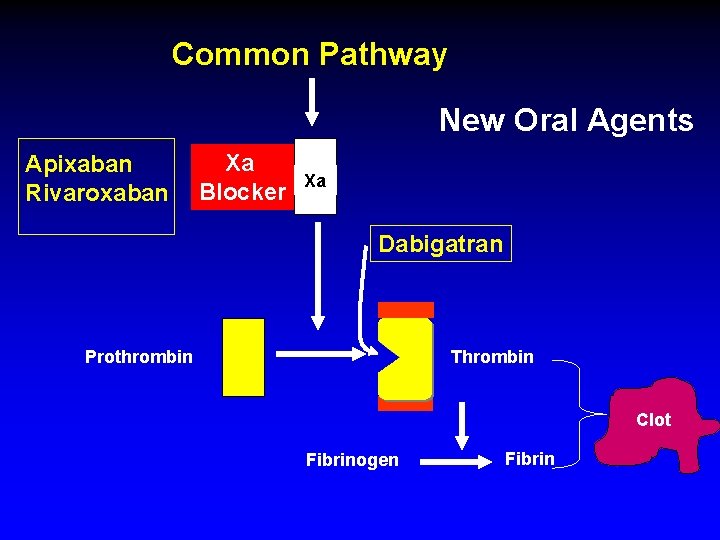
Common Pathway New Oral Agents Apixaban Rivaroxaban Xa Blocker Xa Dabigatran Prothrombin Thrombin Clot Fibrinogen Fibrin

Replacing Traditional Anticoagulants

Treatment VTE UFH, LMWH Bridge to Warfarin

RE-COVER Study Dabigatran 150 mg, BID for 6 months Double Blind, Double Dummy, Non-Inferiority Schulman S, et al NEJM 2009; 361: 2342 -2352
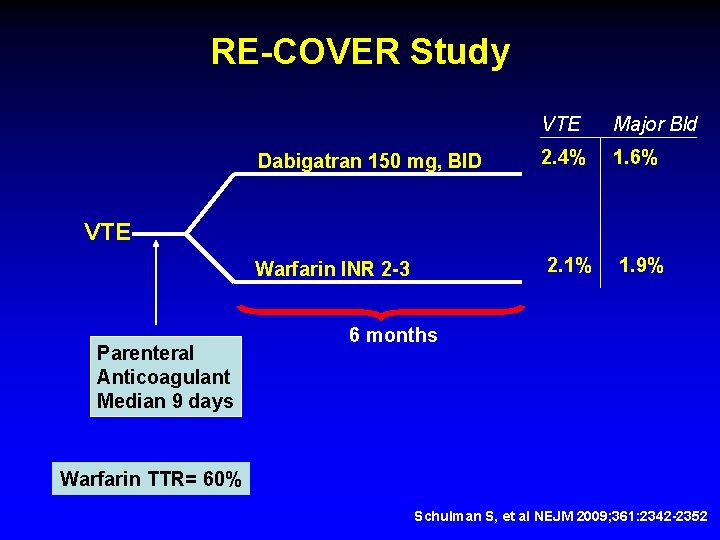
RE-COVER Study VTE Major Bld Dabigatran 150 mg, BID 2. 4% 1. 6% Warfarin INR 2 -3 2. 1% 1. 9% VTE Parenteral Anticoagulant Median 9 days 6 months Warfarin TTR= 60% Schulman S, et al NEJM 2009; 361: 2342 -2352
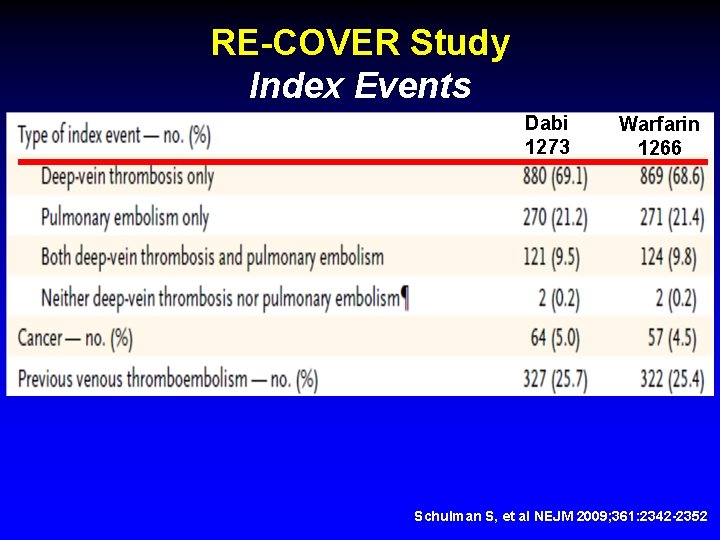
RE-COVER Study Index Events Dabi 1273 Warfarin 1266 Schulman S, et al NEJM 2009; 361: 2342 -2352
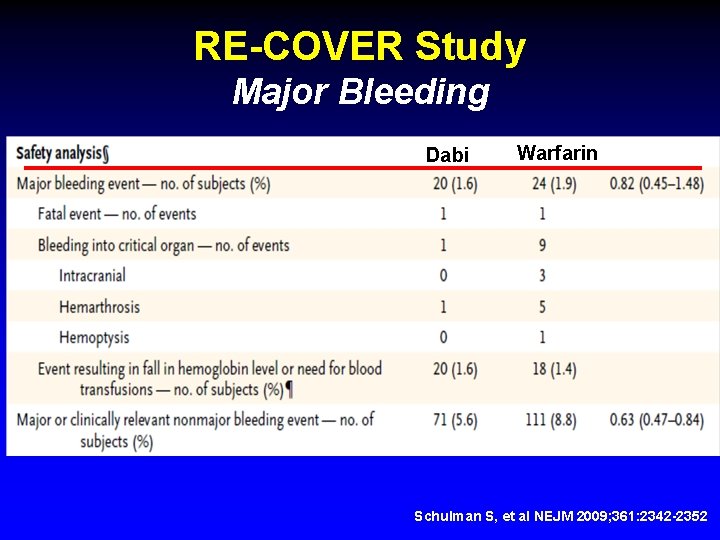
RE-COVER Study Major Bleeding Dabi Warfarin Schulman S, et al NEJM 2009; 361: 2342 -2352

RE-COVER § A limitation of the study is that the first dose of dabigatran, was given only after initial parenteral anticoagulation therapy had been administered for median of 9 days § “There is no data to support the use of dabigatran monotherapy for acute venous thromboembolism” Schulman S, et al NEJM 2009; 361: 2342 -2352

Rivaroxaban 15 mg, PO, BID x 3 weeks then 20 mg, Qday Enoxaparin 1 mg/kg/Q 12 hrs bridge to Warfarin INR 2 -3 Open Label, Non-Inferiority trial Einstein Investigators NEJM 2010; 363: 2499 -2510
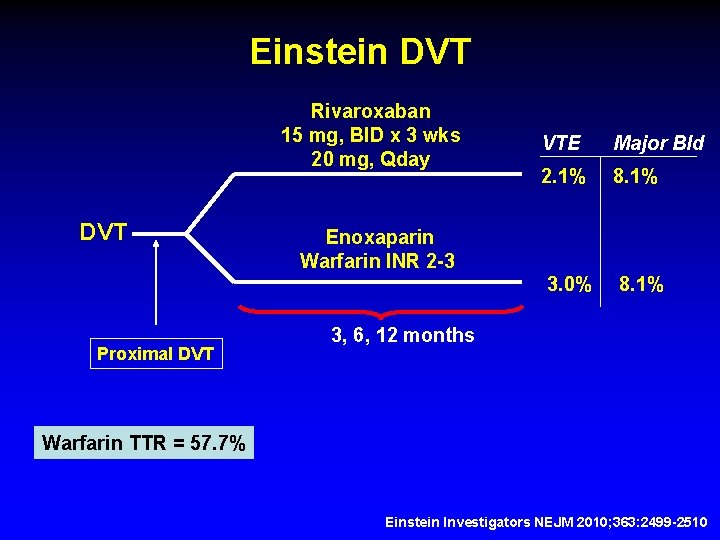
Einstein DVT Rivaroxaban 15 mg, BID x 3 wks 20 mg, Qday DVT Proximal DVT VTE Major Bld 2. 1% 8. 1% 3. 0% 8. 1% Enoxaparin Warfarin INR 2 -3 3, 6, 12 months Warfarin TTR = 57. 7% Einstein Investigators NEJM 2010; 363: 2499 -2510
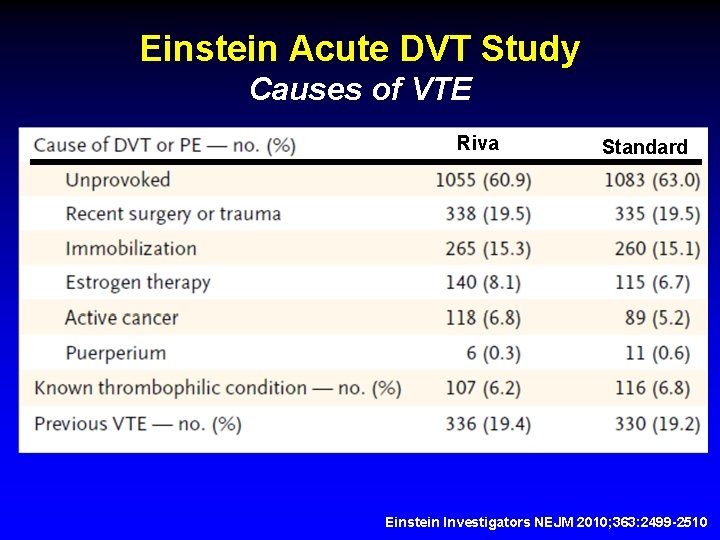
Einstein Acute DVT Study Causes of VTE Riva Standard Einstein Investigators NEJM 2010; 363: 2499 -2510

Einstein Acute DVT Study Safety Outcomes Riva Standard Einstein Investigators NEJM 2010; 363: 2499 -2510

Rivaroxaban 15 mg, PO, BID x 3 weeks then 20 mg, Qday Enoxaparin 1 mg/kg/Q 12 hrs bridge to Warfarin INR 2 -3 Open Label, Non-Inferiority Einstein Investigators NEJM 2012; 366: 1287 -1297

Einstein PE Rivaroxaban 15 mg, BID x 3 wks 20 mg, Qday PE Enoxaparin Warfarin INR 2 -3 VTE Major Bld 2. 1% 1. 1% Non-Inferior 1. 8% 2. 2% 3, 6, 12 months Warfarin TTR = 62. 7% Einstein-PE Investigators NEJM 2012; 366: 1287 -1297
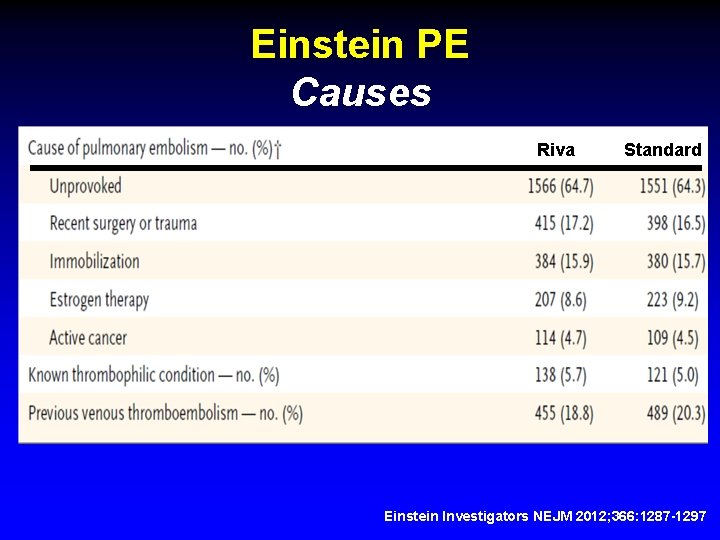
Einstein PE Causes Riva Standard Einstein Investigators NEJM 2012; 366: 1287 -1297
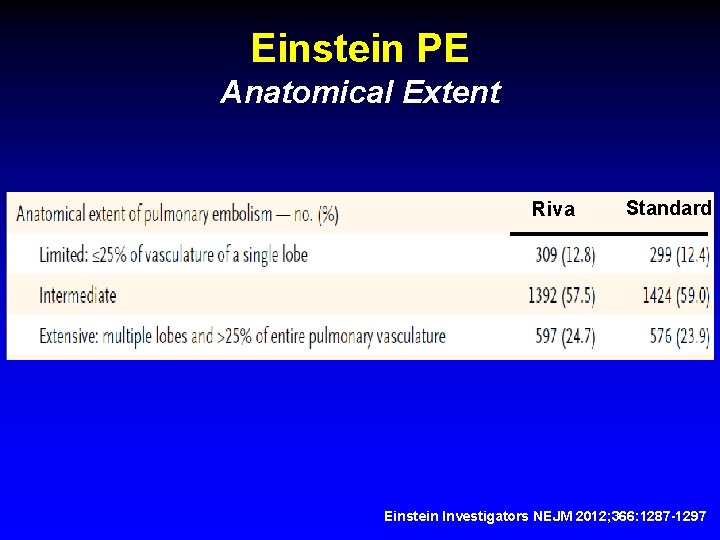
Einstein PE Anatomical Extent Riva Standard Einstein Investigators NEJM 2012; 366: 1287 -1297
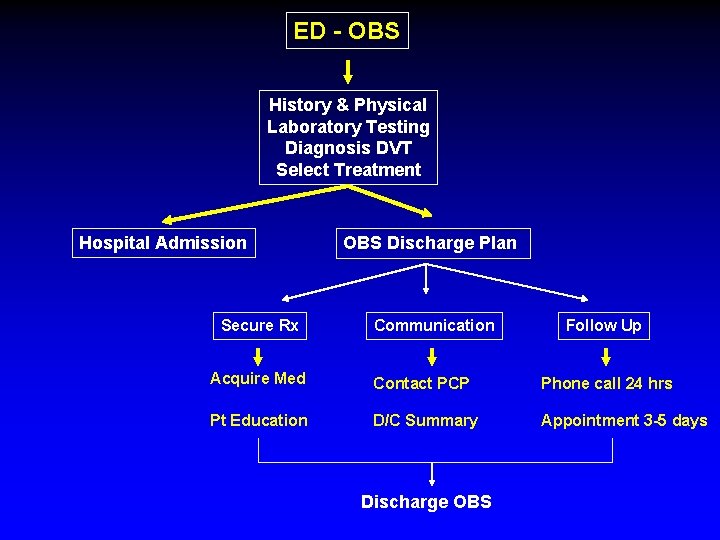
ED - OBS History & Physical Laboratory Testing Diagnosis DVT Select Treatment Hospital Admission Secure Rx OBS Discharge Plan Communication Follow Up Acquire Med Contact PCP Phone call 24 hrs Pt Education D/C Summary Appointment 3 -5 days Discharge OBS

Your patient who has been on long term warfarin would like to convert to one of the new oral anticoagulant.

Rivaroxaban 15 mg, PO, BID x 3 weeks then 20 mg, Qday Enoxaparin 1 mg/kg/Q 12 hrs bridge to Warfarin INR 2 -3 Open Label, Non-Inferiority trial Einstein Investigators NEJM 2010; 363: 2499 -2510

Einstein DVT-Extend Rivaroxaban 20 mg, Qday DVT VTE Major Bld 1. 3% 0. 7% 7. 1% 0% All Rxed Placebo 3, 6, 12 mo 6 -12 mo Einstein Investigators NEJM 2010; 363: 2499 -2510

Double Blind, Randomized Trial Schulman S, et al NEJM 2013; 368: 709 -718

RE-MEDY Dabigatran 150 mg, BID VTE Major Bld 1. 8% 0. 9% 1. 3% 1. 8% VTE Warfarin INR 2 -3 Patient Rx 3 to 12 months 6 months Schulman S, et al NEJM 2013; 368: 709 -718
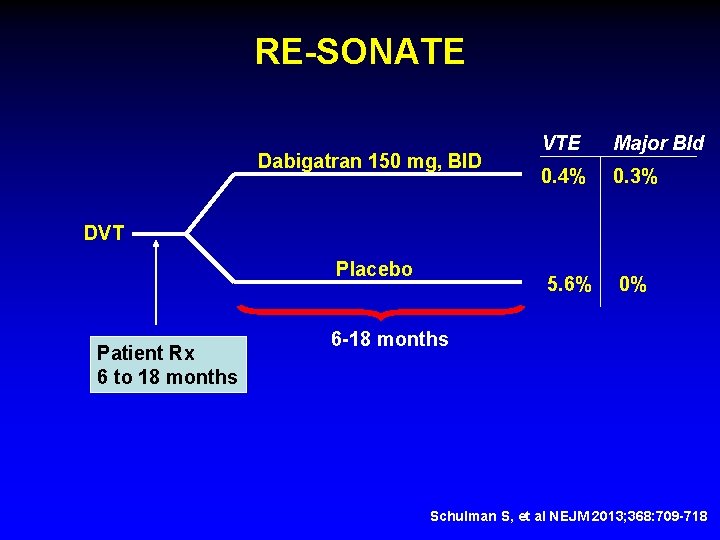
RE-SONATE Dabigatran 150 mg, BID VTE Major Bld 0. 4% 0. 3% 5. 6% 0% DVT Placebo Patient Rx 6 to 18 months 6 -18 months Schulman S, et al NEJM 2013; 368: 709 -718
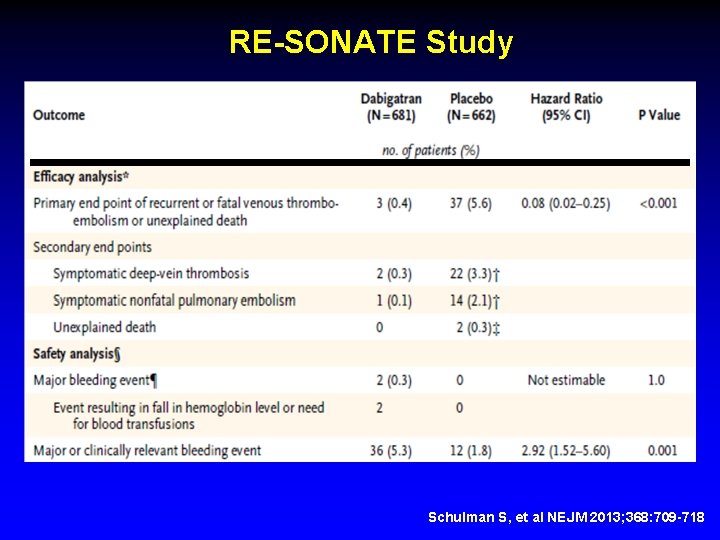
RE-SONATE Study Schulman S, et al NEJM 2013; 368: 709 -718

Agnelli G, et al NEJM 2012; 1 -10

AMPLIFY-EXT VTE Rx 6 -12 mo Major Bld Apixaban 2. 5 mg, BID 1. 7% 0. 2% Apixaban 5. 0 mg, BID 1. 7% 0. 1% Placebo 8. 8% 0. 5% 12 months Agnelli G, et al NEJM 2013; 368(8): 699 -708
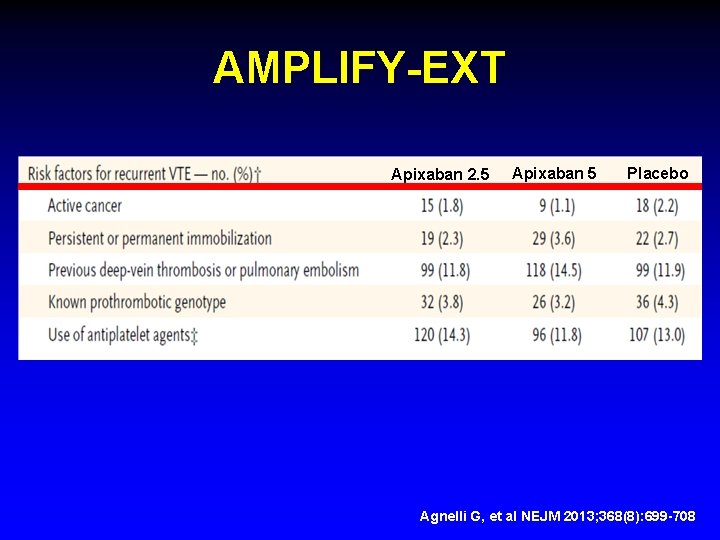
AMPLIFY-EXT Apixaban 2. 5 Apixaban 5 Placebo Agnelli G, et al NEJM 2013; 368(8): 699 -708

Warfarin to NOAC Agent Recommendation Rivaroxaban Start when INR < 3. 0 (we recommend < 2. 0) Apixaban Start when INR < 2. 0 Dabigatran Start when INR < 2. 0 NOAC= New Oral Anticoagulants

Non-Valvular Atrial Fibrillation
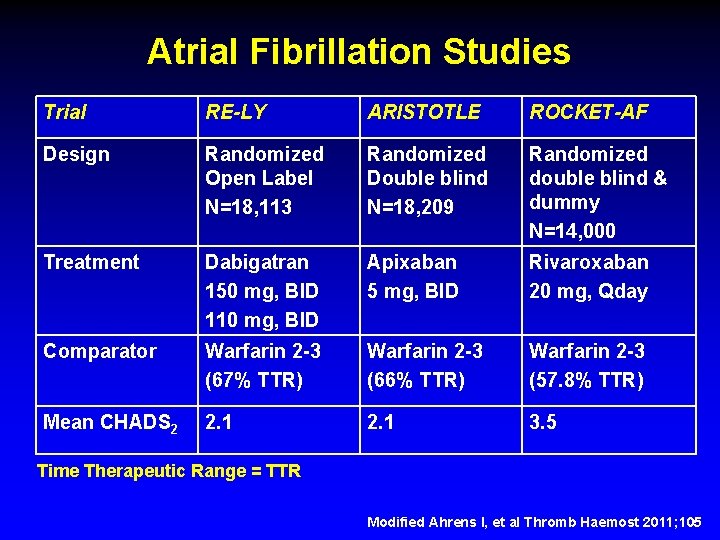
Atrial Fibrillation Studies Trial RE-LY ARISTOTLE ROCKET-AF Design Randomized Open Label N=18, 113 Randomized Double blind N=18, 209 Randomized double blind & dummy N=14, 000 Treatment Dabigatran 150 mg, BID 110 mg, BID Apixaban 5 mg, BID Rivaroxaban 20 mg, Qday Comparator Warfarin 2 -3 (67% TTR) Warfarin 2 -3 (66% TTR) Warfarin 2 -3 (57. 8% TTR) Mean CHADS 2 2. 1 3. 5 Time Therapeutic Range = TTR Modified Ahrens I, et al Thromb Haemost 2011; 105
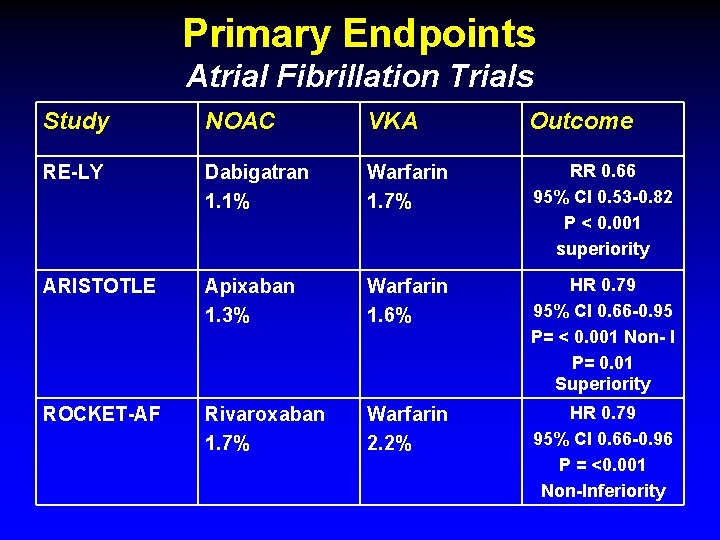
Primary Endpoints Atrial Fibrillation Trials Study NOAC VKA Outcome RE-LY Dabigatran 1. 1% Warfarin 1. 7% RR 0. 66 95% CI 0. 53 -0. 82 P < 0. 001 superiority ARISTOTLE Apixaban 1. 3% Warfarin 1. 6% HR 0. 79 95% CI 0. 66 -0. 95 P= < 0. 001 Non- I P= 0. 01 Superiority ROCKET-AF Rivaroxaban 1. 7% Warfarin 2. 2% HR 0. 79 95% CI 0. 66 -0. 96 P = <0. 001 Non-Inferiority
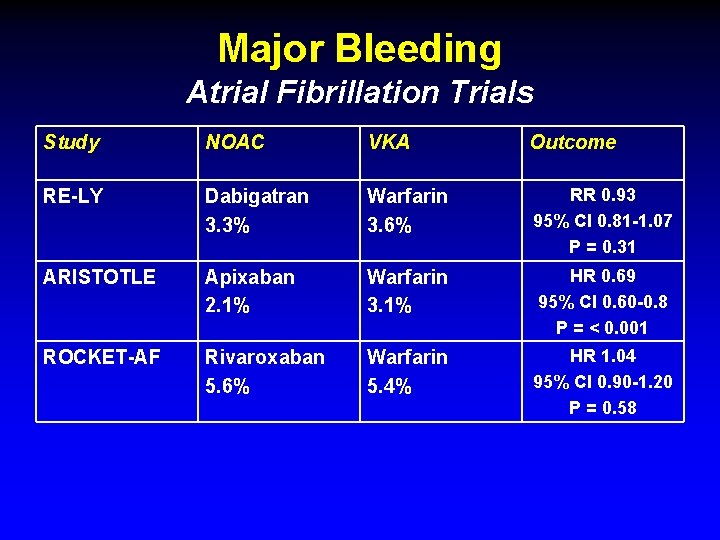
Major Bleeding Atrial Fibrillation Trials Study NOAC VKA Outcome RE-LY Dabigatran 3. 3% Warfarin 3. 6% RR 0. 93 95% CI 0. 81 -1. 07 P = 0. 31 ARISTOTLE Apixaban 2. 1% Warfarin 3. 1% HR 0. 69 95% CI 0. 60 -0. 8 P = < 0. 001 ROCKET-AF Rivaroxaban 5. 6% Warfarin 5. 4% HR 1. 04 95% CI 0. 90 -1. 20 P = 0. 58
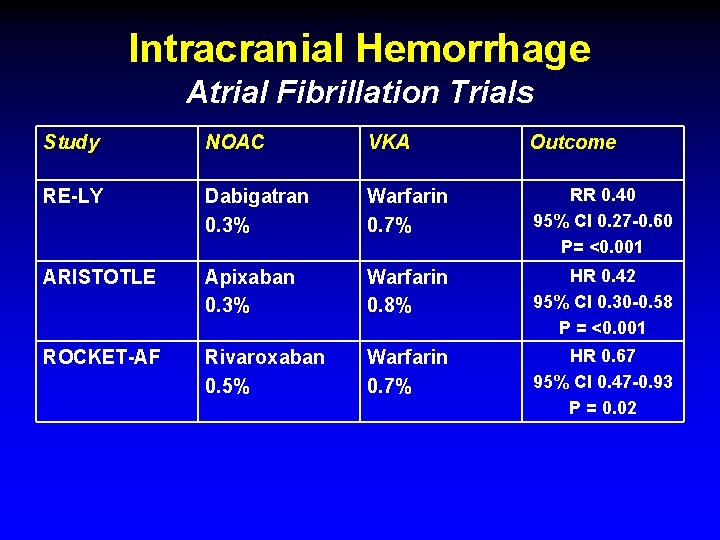
Intracranial Hemorrhage Atrial Fibrillation Trials Study NOAC VKA Outcome RE-LY Dabigatran 0. 3% Warfarin 0. 7% RR 0. 40 95% CI 0. 27 -0. 60 P= <0. 001 ARISTOTLE Apixaban 0. 3% Warfarin 0. 8% HR 0. 42 95% CI 0. 30 -0. 58 P = <0. 001 ROCKET-AF Rivaroxaban 0. 5% Warfarin 0. 7% HR 0. 67 95% CI 0. 47 -0. 93 P = 0. 02
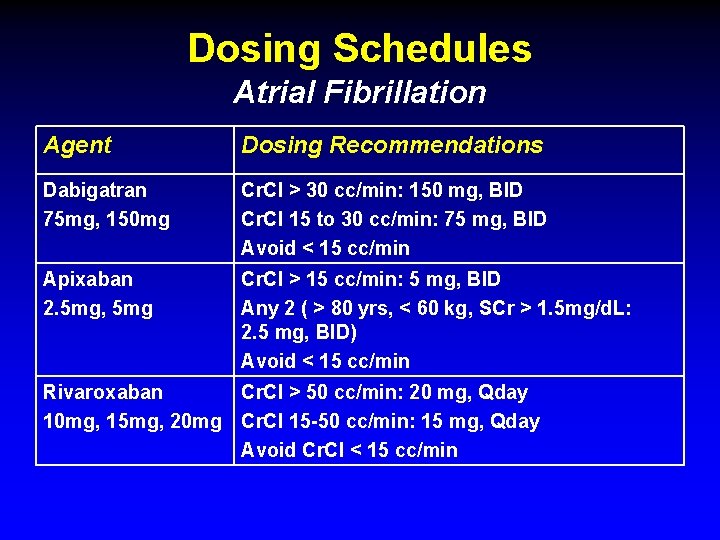
Dosing Schedules Atrial Fibrillation Agent Dosing Recommendations Dabigatran 75 mg, 150 mg Cr. Cl > 30 cc/min: 150 mg, BID Cr. Cl 15 to 30 cc/min: 75 mg, BID Avoid < 15 cc/min Apixaban 2. 5 mg, 5 mg Cr. Cl > 15 cc/min: 5 mg, BID Any 2 ( > 80 yrs, < 60 kg, SCr > 1. 5 mg/d. L: 2. 5 mg, BID) Avoid < 15 cc/min Rivaroxaban Cr. Cl > 50 cc/min: 20 mg, Qday 10 mg, 15 mg, 20 mg Cr. Cl 15 -50 cc/min: 15 mg, Qday Avoid Cr. Cl < 15 cc/min

Atrial Fibrillation Studies When should new orals be started? § RE-LY (Dabigatran) § Stroke within 14 days § Severe stroke within last 6 months § ARISTOTLE (Apixaban) § Stroke within 7 days § ROCKET-AF (Rivaroxaban) § Stroke within 14 days § Severe stroke within last 3 months Modified-Ahrens I, et al Thromb Haemost 2011; 105

Medically ill Patient
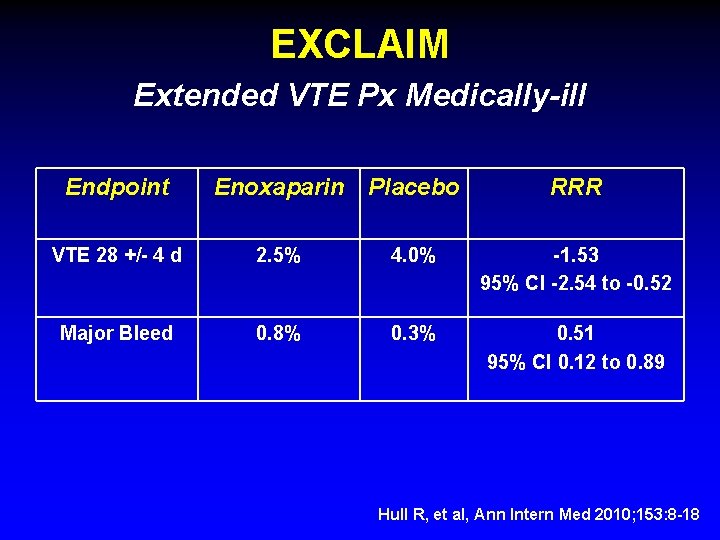
EXCLAIM Extended VTE Px Medically-ill Endpoint Enoxaparin Placebo RRR VTE 28 +/- 4 d 2. 5% 4. 0% -1. 53 95% CI -2. 54 to -0. 52 Major Bleed 0. 8% 0. 3% 0. 51 95% CI 0. 12 to 0. 89 Hull R, et al, Ann Intern Med 2010; 153: 8 -18

ADOPT Apixaban 2. 5 mg BID Enoxaparin 40 mg, Qday Goldhaber S, et al NEJM 2011; 365(23): 2167 -2177
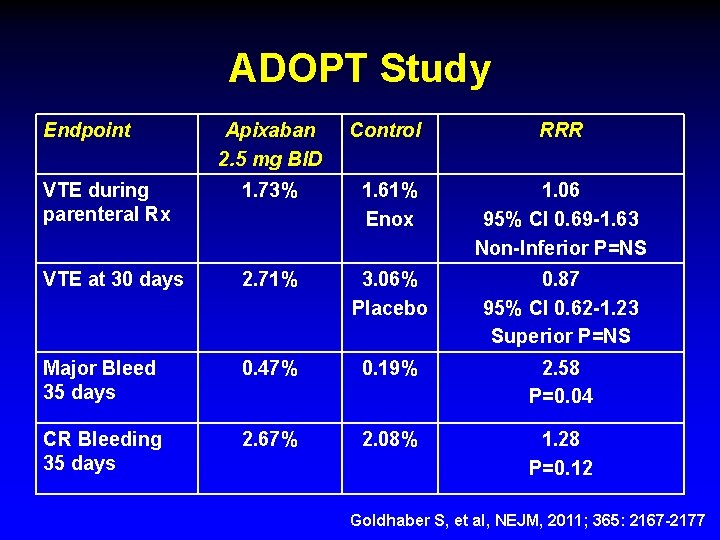
ADOPT Study Endpoint Apixaban 2. 5 mg BID Control RRR VTE during parenteral Rx 1. 73% 1. 61% Enox 1. 06 95% CI 0. 69 -1. 63 Non-Inferior P=NS VTE at 30 days 2. 71% 3. 06% Placebo 0. 87 95% CI 0. 62 -1. 23 Superior P=NS Major Bleed 35 days 0. 47% 0. 19% 2. 58 P=0. 04 CR Bleeding 35 days 2. 67% 2. 08% 1. 28 P=0. 12 Goldhaber S, et al, NEJM, 2011; 365: 2167 -2177

Cohen A, et al NEJM 2013; 368: 513 -523
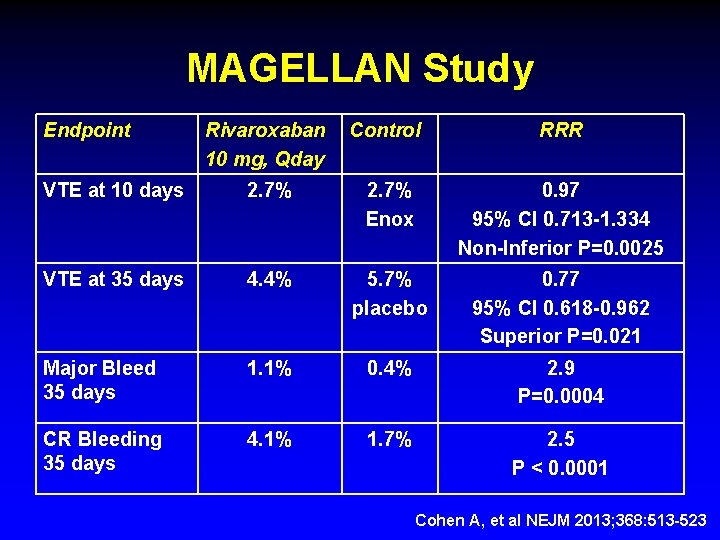
MAGELLAN Study Endpoint Rivaroxaban 10 mg, Qday Control RRR VTE at 10 days 2. 7% Enox 0. 97 95% CI 0. 713 -1. 334 Non-Inferior P=0. 0025 VTE at 35 days 4. 4% 5. 7% placebo 0. 77 95% CI 0. 618 -0. 962 Superior P=0. 021 Major Bleed 35 days 1. 1% 0. 4% 2. 9 P=0. 0004 CR Bleeding 35 days 4. 1% 1. 7% 2. 5 P < 0. 0001 Cohen A, et al NEJM 2013; 368: 513 -523
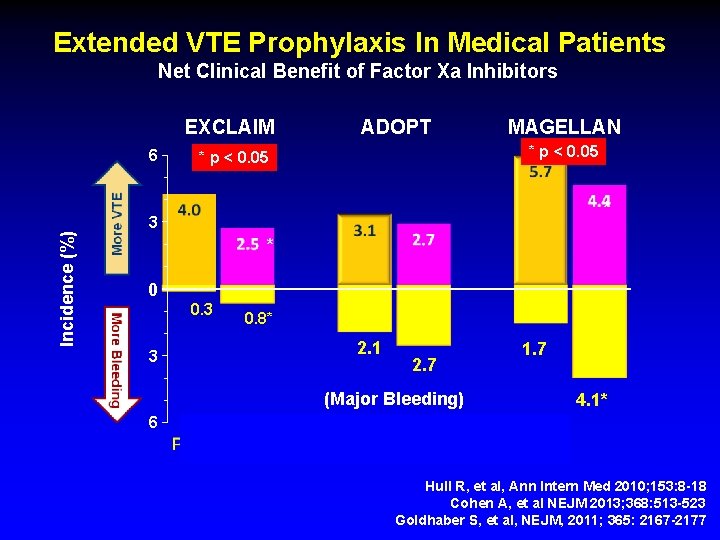
Extended VTE Prophylaxis In Medical Patients Net Clinical Benefit of Factor Xa Inhibitors EXCLAIM Incidence (%) 6 ADOPT MAGELLAN * p < 0. 05 * 3 * 0 0. 3 0. 8* 2. 1 3 2. 7 1. 7 (Major Bleeding) 4. 1* 6 (n = 5, 963) (n = 6, 528) (n = 8, 101) Hull R, et al, Ann Intern Med 2010; 153: 8 -18 Cohen A, et al NEJM 2013; 368: 513 -523 Goldhaber S, et al, NEJM, 2011; 365: 2167 -2177

Key Points Black Box Warnings, Stroke, MI Risk, Drug Interactions, Major Bleeding

Black Box Warning Rivaroxaban & Apixaban
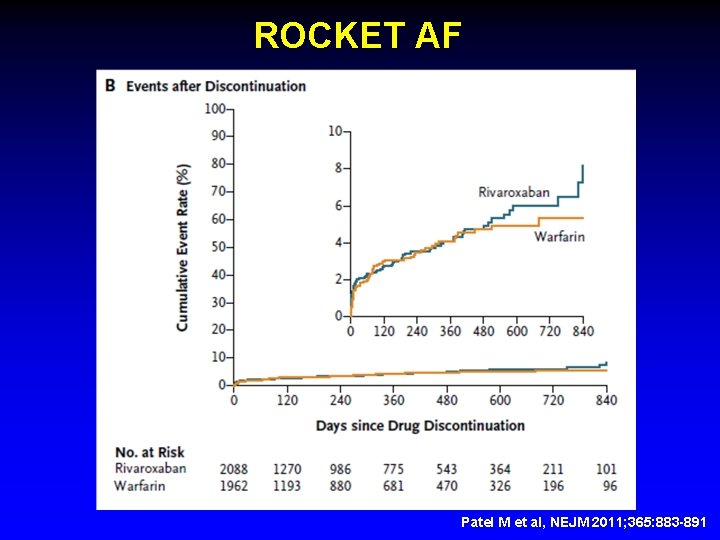
ROCKET AF Patel M et al, NEJM 2011; 365: 883 -891
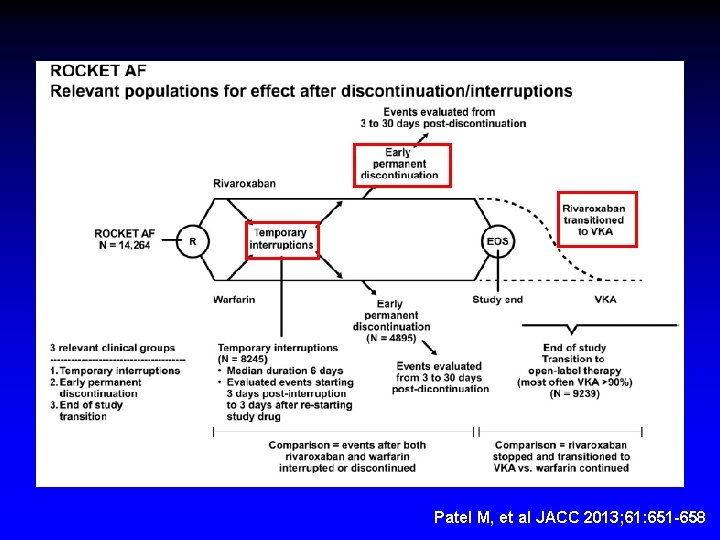
Patel M, et al JACC 2013; 61: 651 -658
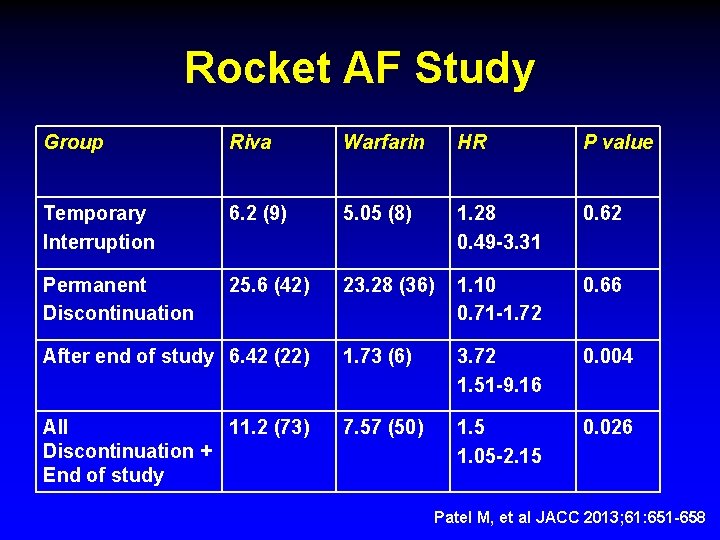
Rocket AF Study Group Riva Warfarin HR P value Temporary Interruption 6. 2 (9) 5. 05 (8) 1. 28 0. 49 -3. 31 0. 62 Permanent Discontinuation 25. 6 (42) 23. 28 (36) 1. 10 0. 71 -1. 72 0. 66 After end of study 6. 42 (22) 1. 73 (6) 3. 72 1. 51 -9. 16 0. 004 All 11. 2 (73) Discontinuation + End of study 7. 57 (50) 1. 5 1. 05 -2. 15 0. 026 Patel M, et al JACC 2013; 61: 651 -658
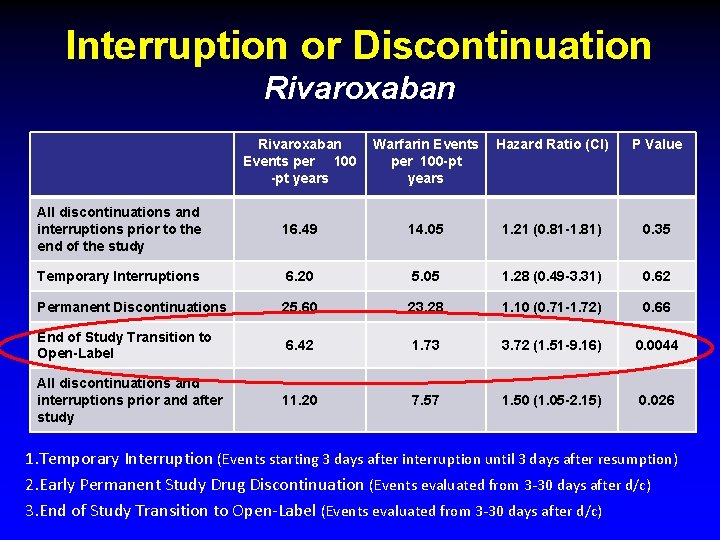
Interruption or Discontinuation Rivaroxaban Events per 100 -pt years Warfarin Events per 100 -pt years Hazard Ratio (CI) P Value All discontinuations and interruptions prior to the end of the study 16. 49 14. 05 1. 21 (0. 81 -1. 81) 0. 35 Temporary Interruptions 6. 20 5. 05 1. 28 (0. 49 -3. 31) 0. 62 Permanent Discontinuations 25. 60 23. 28 1. 10 (0. 71 -1. 72) 0. 66 End of Study Transition to Open-Label 6. 42 1. 73 3. 72 (1. 51 -9. 16) 0. 0044 All discontinuations and interruptions prior and after study 11. 20 7. 57 1. 50 (1. 05 -2. 15) 0. 026 1. Temporary Interruption (Events starting 3 days after interruption until 3 days after resumption) 2. Early Permanent Study Drug Discontinuation (Events evaluated from 3 -30 days after d/c) 3. End of Study Transition to Open-Label (Events evaluated from 3 -30 days after d/c)
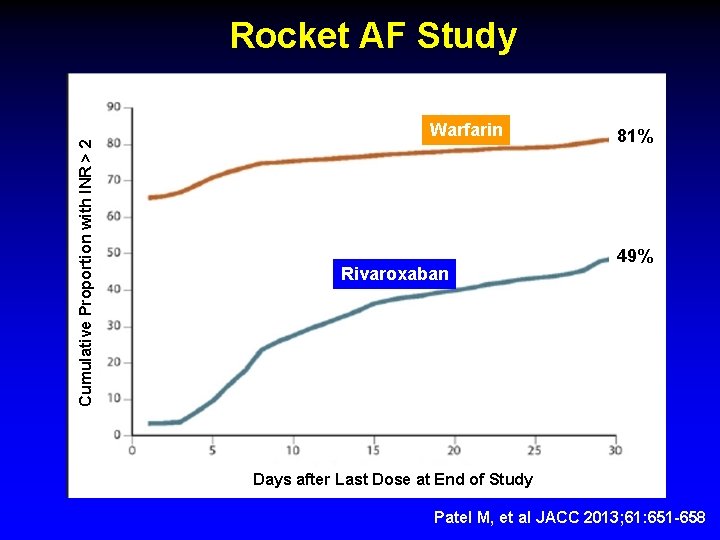
Cumulative Proportion with INR > 2 Rocket AF Study Warfarin Rivaroxaban 81% 49% Days after Last Dose at End of Study Patel M, et al JACC 2013; 61: 651 -658
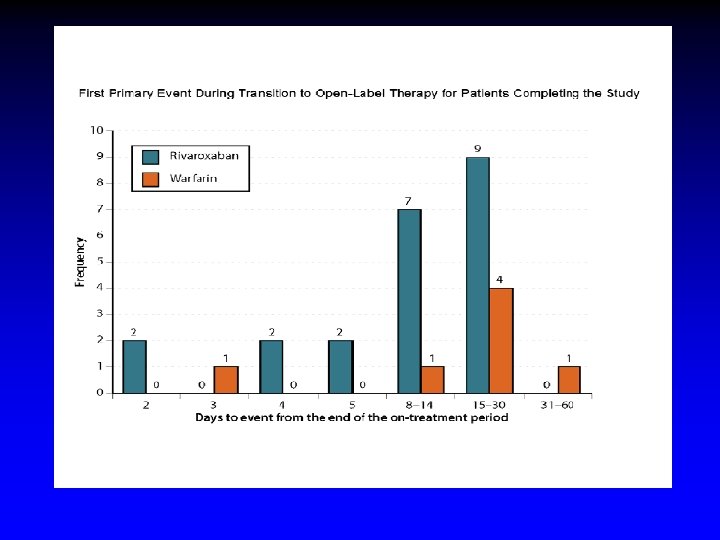

What happened in ROCKET AF ? § Warfarin patients continued warfarin § Rivaroxaban patients discontinued study drug and then began warfarin § Not anticoagulated during warfarin titration § No “Bridging” § Strokes during the 30 days post study § Warfarin group – 6 § Rivaroxaban group - 22

Black Box Warning Rivaroxaban § Epidural or Spinal Hematoma § Use of epidural catheter § Concomitant use of NSAID, Anti-platelet § Traumatic or repeated spinal puncture § History of spinal deformity
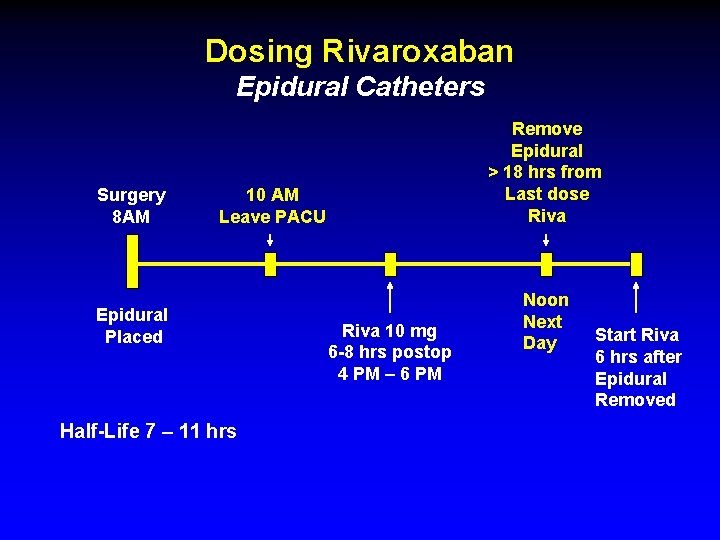
Dosing Rivaroxaban Epidural Catheters Surgery 8 AM Remove Epidural > 18 hrs from Last dose Riva 10 AM Leave PACU Epidural Placed Half-Life 7 – 11 hrs Riva 10 mg 6 -8 hrs postop 4 PM – 6 PM Noon Next Day Start Riva 6 hrs after Epidural Removed

7 Clinical Trials Evaluated 2 Stroke Prophylaxis in Atrial Fibrillation 1 Acute Venous Thromboembolism 1 Acute Coronary Syndrome 3 VTE Prophylaxis Joint Replacement Surgery Uchino K, et al Arch Intern Med 2012; 172: 397 -402
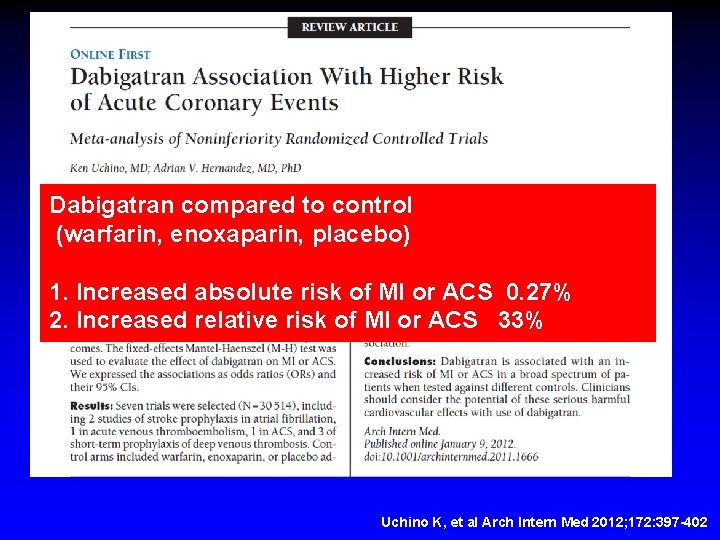
Dabigatran compared to control (warfarin, enoxaparin, placebo) 1. Increased absolute risk of MI or ACS 0. 27% 2. Increased relative risk of MI or ACS 33% Uchino K, et al Arch Intern Med 2012; 172: 397 -402

Eriksson B, et al Thromb Res 2012; 130: 396 -402
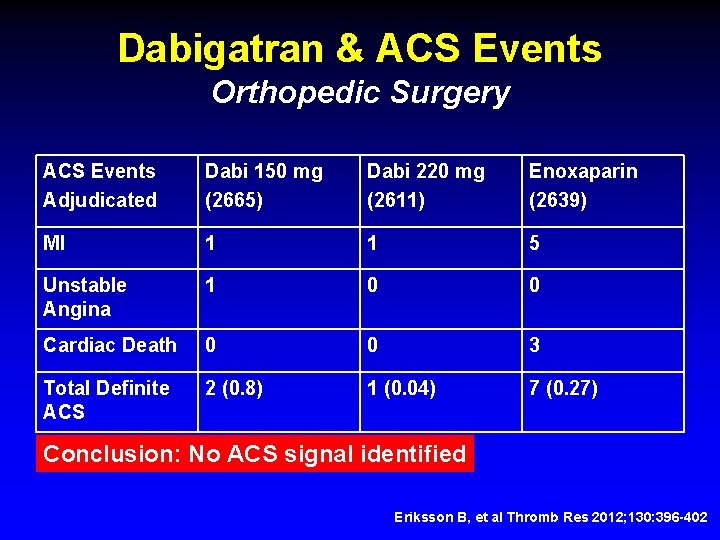
Dabigatran & ACS Events Orthopedic Surgery ACS Events Adjudicated Dabi 150 mg (2665) Dabi 220 mg (2611) Enoxaparin (2639) MI 1 1 5 Unstable Angina 1 0 0 Cardiac Death 0 0 3 Total Definite ACS 2 (0. 8) 1 (0. 04) 7 (0. 27) Conclusion: No ACS signal identified Eriksson B, et al Thromb Res 2012; 130: 396 -402

Major Bleeding
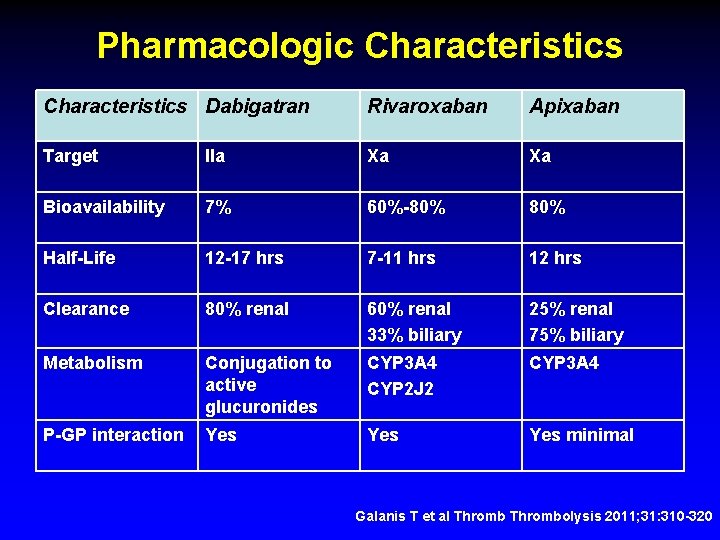
Pharmacologic Characteristics Dabigatran Rivaroxaban Apixaban Target IIa Xa Xa Bioavailability 7% 60%-80% Half-Life 12 -17 hrs 7 -11 hrs 12 hrs Clearance 80% renal 60% renal 33% biliary 25% renal 75% biliary Metabolism Conjugation to active glucuronides CYP 3 A 4 CYP 2 J 2 CYP 3 A 4 P-GP interaction Yes Yes minimal Galanis T et al Thrombolysis 2011; 31: 310 -320
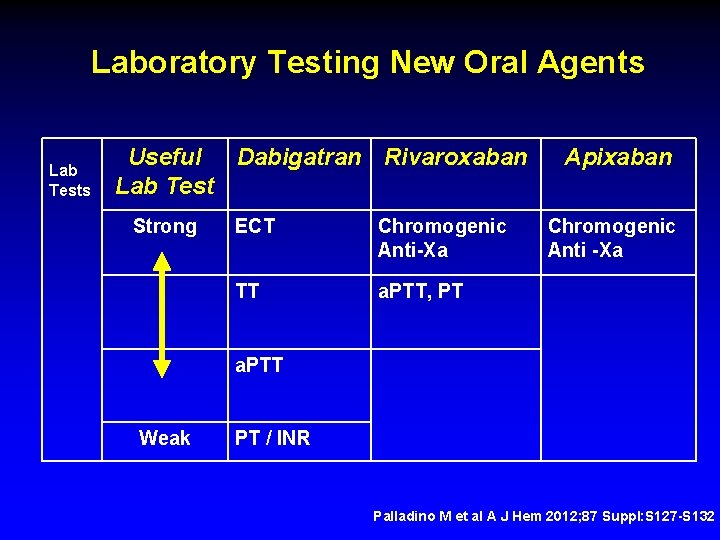
Laboratory Testing New Oral Agents Lab Tests Useful Dabigatran Rivaroxaban Lab Test Strong ECT Chromogenic Anti-Xa TT a. PTT, PT Apixaban Chromogenic Anti -Xa a. PTT Weak PT / INR Palladino M et al A J Hem 2012; 87 Suppl: S 127 -S 132

Novel Anticoagulant Comparison Dabigatran Rivaroxaban Apixaban Dialyzable Yes Probably Not Molecular Weight 628 Daltons 436 Daltons 460 Daltons Protein Binding 35% >90% 87% Catalytic Binding Site Reversible Reversing Agent No Possibly Erikkson BI, et al. Clin Pharmacokinet 2009; 48: 1 -22.

COFACT (Prothrombin Complex Concentrate) 1. Non-activated PCC 2. Factor II, VII, IX, X 3. Protein C, S, ATIII 4. 50 IU PCC/kg dosing Eerenberg E, et al Circulation 2011; 124: 1573 -1579
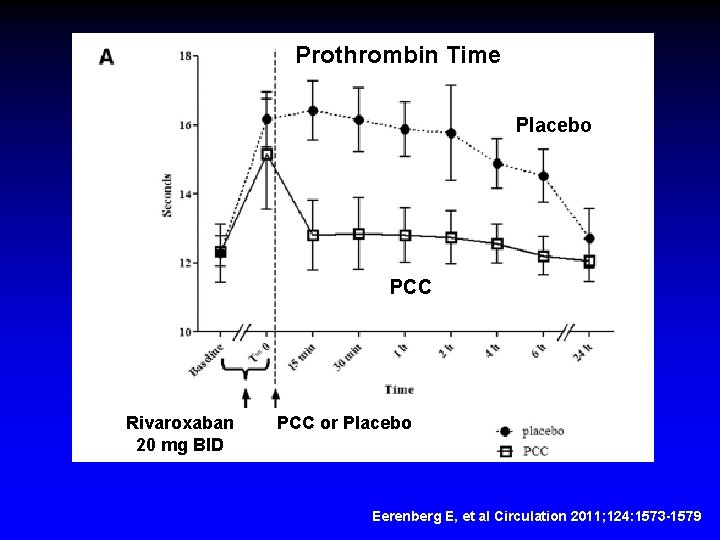
Prothrombin Time Placebo PCC Rivaroxaban 20 mg BID PCC or Placebo Eerenberg E, et al Circulation 2011; 124: 1573 -1579
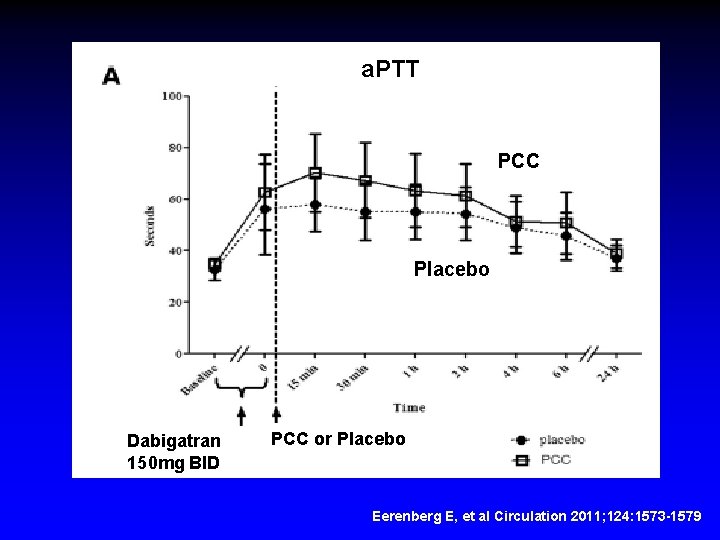
a. PTT PCC Placebo Dabigatran 150 mg BID PCC or Placebo Eerenberg E, et al Circulation 2011; 124: 1573 -1579

Thrombin Time Placebo Dabigatran 150 mg BID PCC or Placebo Eerenberg E, et al Circulation 2011; 124: 1573 -1579
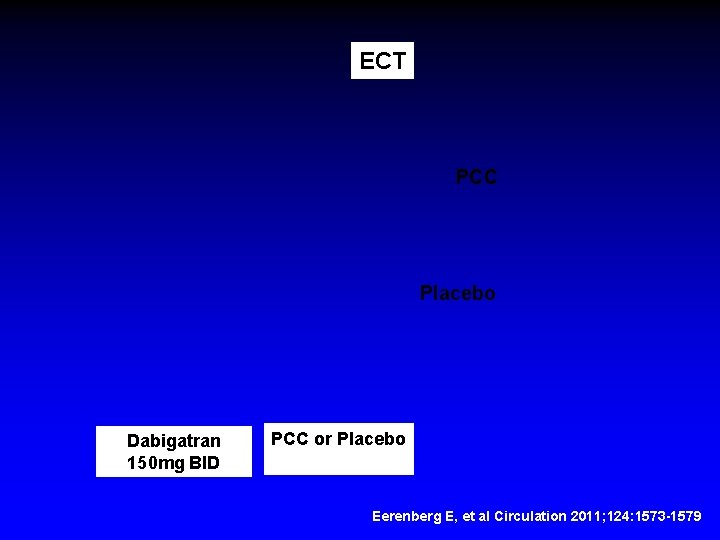
ECT PCC Placebo Dabigatran 150 mg BID PCC or Placebo Eerenberg E, et al Circulation 2011; 124: 1573 -1579
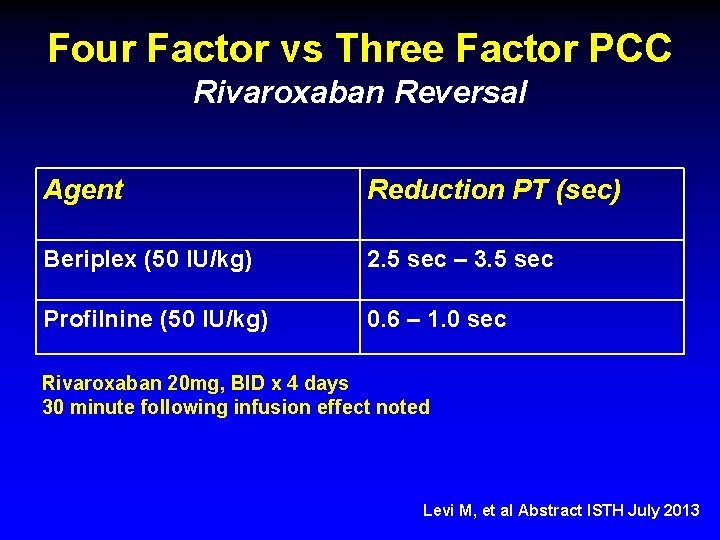
Four Factor vs Three Factor PCC Rivaroxaban Reversal Agent Reduction PT (sec) Beriplex (50 IU/kg) 2. 5 sec – 3. 5 sec Profilnine (50 IU/kg) 0. 6 – 1. 0 sec Rivaroxaban 20 mg, BID x 4 days 30 minute following infusion effect noted Levi M, et al Abstract ISTH July 2013
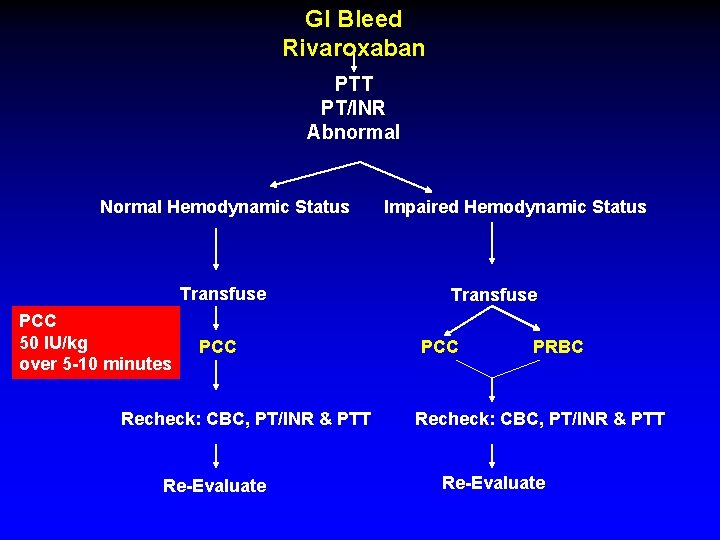
GI Bleed Rivaroxaban PTT PT/INR Abnormal Normal Hemodynamic Status Transfuse PCC 50 IU/kg over 5 -10 minutes PCC Recheck: CBC, PT/INR & PTT Re-Evaluate Impaired Hemodynamic Status Transfuse PCC PRBC Recheck: CBC, PT/INR & PTT Re-Evaluate
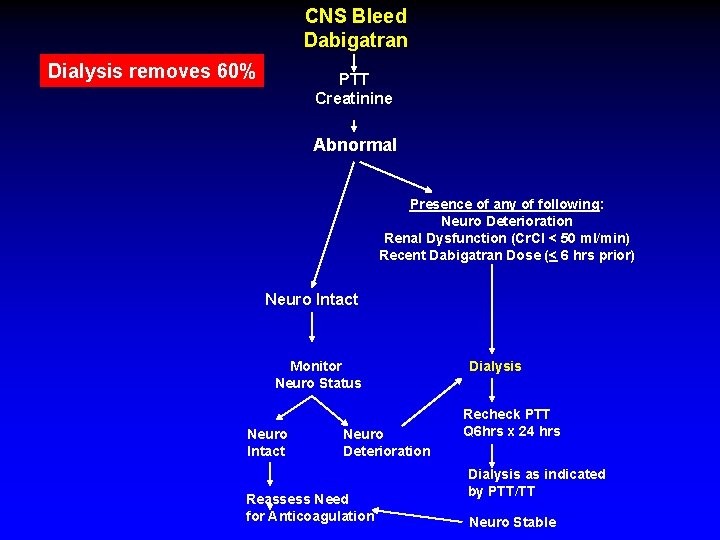
CNS Bleed Dabigatran Dialysis removes 60% PTT Creatinine Abnormal Presence of any of following: Neuro Deterioration Renal Dysfunction (Cr. Cl < 50 ml/min) Recent Dabigatran Dose (< 6 hrs prior) Neuro Intact Monitor Neuro Status Neuro Intact Neuro Deterioration Reassess Need for Anticoagulation Dialysis Recheck PTT Q 6 hrs x 24 hrs Dialysis as indicated by PTT/TT Neuro Stable

Package Insert Recommendations § Dabigatran § FFP, Prothrombin Complex Concentrate § Activated Factor VII § Dialysis § Rivaroxaban & Apixaban § Prothrombin Complex Concentrate § Four Factor Concentrate (KCentra) § FFP
- Slides: 70