Common Ion Effect Buffers Common Ion Effect l

Common Ion Effect Buffers

Common Ion Effect l l l Sometimes the equilibrium solutions have 2 ions in common For example if I mixed HF & Na. F The main reaction is HF H+ + FBut some additional F- ions are being added from the Na. F These are worked the same way, you just start with a different initial amount of the ion

Common Ion Effect l Which way will the reaction shift if Na. F is added? l To the reactant side l What effect will this have on p. H? l [H+] will go down…so p. H will go up

Example l What is the p. H of a 0. 10 M solution of HC 2 H 3 O 2 (Ka = 1. 8 x 10 -5)
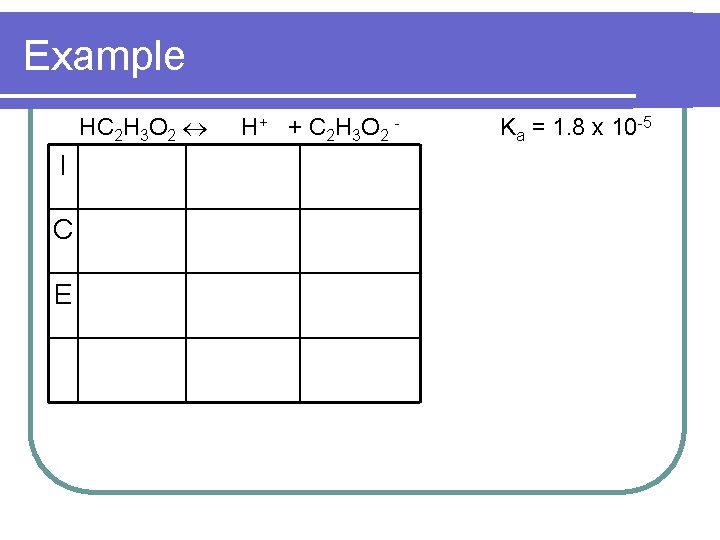
Example HC 2 H 3 O 2 I C E H + + C 2 H 3 O 2 - Ka = 1. 8 x 10 -5
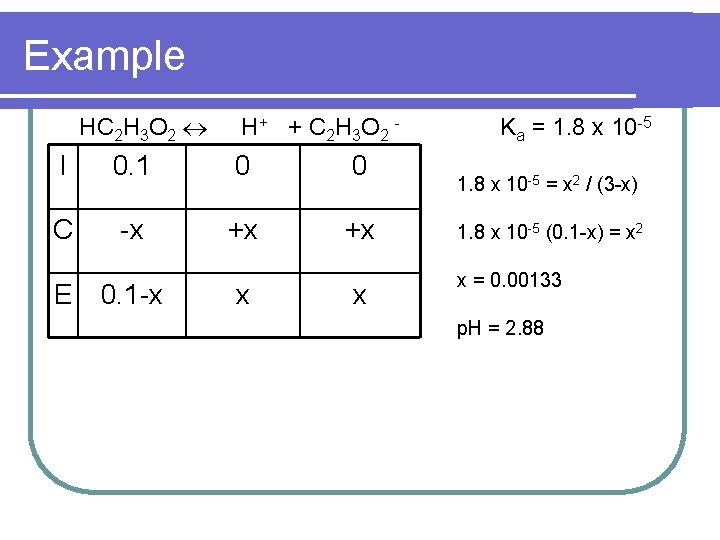
Example HC 2 H 3 O 2 H + + C 2 H 3 O 2 - I 0. 1 0 0 C -x +x +x E 0. 1 -x x x Ka = 1. 8 x 10 -5 = x 2 / (3 -x) 1. 8 x 10 -5 (0. 1 -x) = x 2 x = 0. 00133 p. H = 2. 88

Example l. A mixture contains 0. 10 M HC 2 H 3 O 2 (Ka = 1. 8 x 10 -5) & 0. 10 M Na. C 2 H 3 O 2. Calculate the p. H.
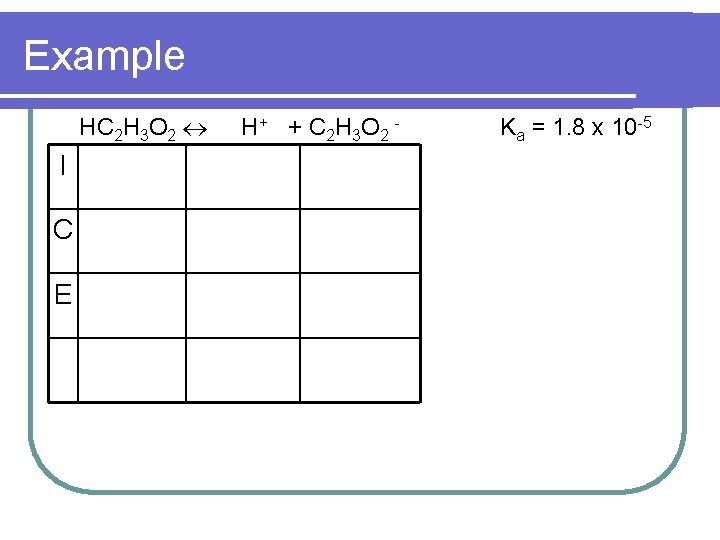
Example HC 2 H 3 O 2 I C E H + + C 2 H 3 O 2 - Ka = 1. 8 x 10 -5
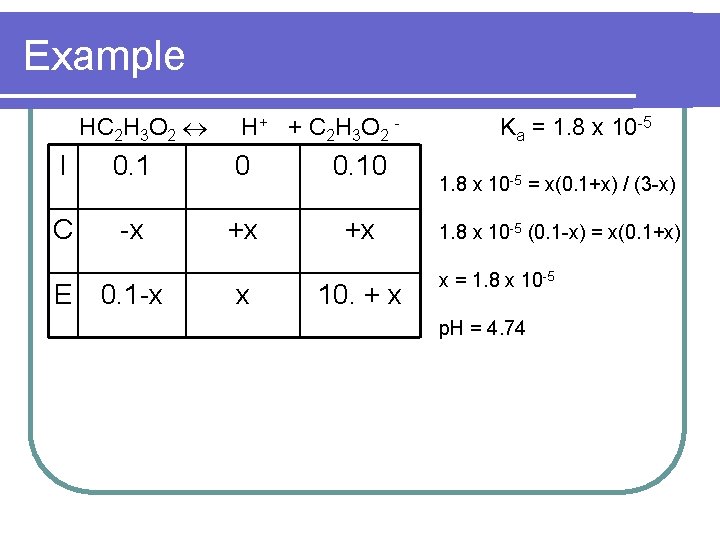
Example HC 2 H 3 O 2 H + + C 2 H 3 O 2 - I 0. 1 0 0. 10 C -x +x +x E 0. 1 -x x 10. + x Ka = 1. 8 x 10 -5 = x(0. 1+x) / (3 -x) 1. 8 x 10 -5 (0. 1 -x) = x(0. 1+x) x = 1. 8 x 10 -5 p. H = 4. 74

What are buffers? Buffers resist changes in p. H l They must have 2 parts… l 1. 2. l Weak acid & a conjugate base OR Weak base & a conjugate acid The concentrations of the 2 MUST be with in a factor of 10!!!
![Buffers [Na. C 2 H 3 O 2 ] [HC 2 H 3 O Buffers [Na. C 2 H 3 O 2 ] [HC 2 H 3 O](http://slidetodoc.com/presentation_image/114742f6d345599c5b692385bb6c2fc0/image-11.jpg)
Buffers [Na. C 2 H 3 O 2 ] [HC 2 H 3 O 2 ] Buffer? 0. 10 M Yes 0. 10 M 1. 0 M Yes 0. 01 M 1. 0 M NO (Not within a factor of 10)

Example l l l l l What is the p. H of a solution containing 50. m. L of 0. 50 M Na. C 2 H 3 O 2 & 25 m. L of 0. 25 M HC 2 H 3 O 2. (Ka = 1. 8 x 10 -5). Na. C 2 H 3 O 2 M = mol/L 0. 5 = mol / 50. 25 mmol Na. C 2 H 3 O 2/ 75 m. L = 0. 33 M HC 2 H 3 O 2 M = mol/L 0. 25 = mol / 25 6. 25 mmol HC 2 H 3 O 2/ 75 m. L = 0. 083 M
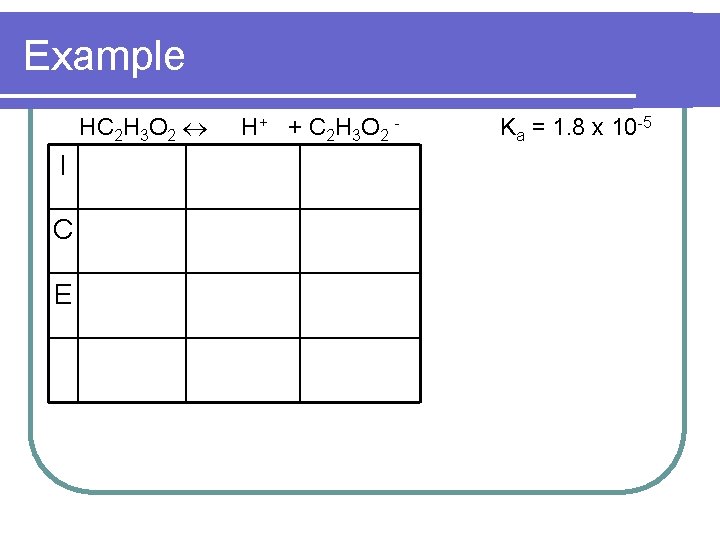
Example HC 2 H 3 O 2 I C E H + + C 2 H 3 O 2 - Ka = 1. 8 x 10 -5
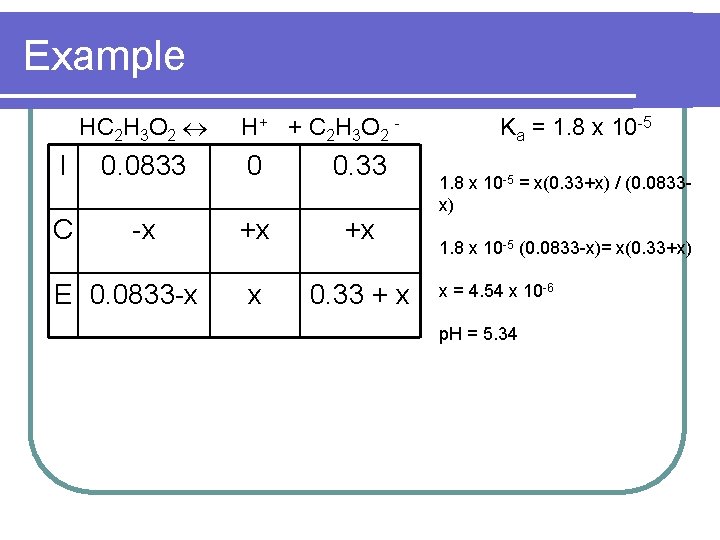
Example HC 2 H 3 O 2 H + + C 2 H 3 O 2 - I 0. 0833 0 0. 33 C -x +x +x x 0. 33 + x E 0. 0833 -x Ka = 1. 8 x 10 -5 = x(0. 33+x) / (0. 0833 x) 1. 8 x 10 -5 (0. 0833 -x)= x(0. 33+x) x = 4. 54 x 10 -6 p. H = 5. 34

Henderson Hasselbach Equation l Really easy equation to use ONLY with buffer solutions!!! l p. H = p. Ka + log [B]/[A]

The last example using HH l l l l p. H = p. Ka + log [B]/[A] p. H = 4. 74 + log [25 mmol / 75 m. L] [6. 25 mmol / 75 m. L] The m. L cross out, so on HH you can use mmol p. H = 4. 74 + log (25/6. 25) p. H = 5. 34 Same answer as we got with the ICE table Pick the way you like better & use it!

Example (ICE TABLE) l l l l l What is the p. H of a solution containing 25 m. L of 0. 150 MHCl. O & 32 m. L of 0. 45 M KCl. O. Ka = 3. 5 x 10 -8 HCl. O M = mol/L 0. 15 = mol / 25 3. 75 mmol HCl. O/ 57 m. L = 0. 0658 M KCl. O M = mol/L 0. 45 = mol / 32 14. 4 mmol KCl. O/ 57 m. L = 0. 253 M
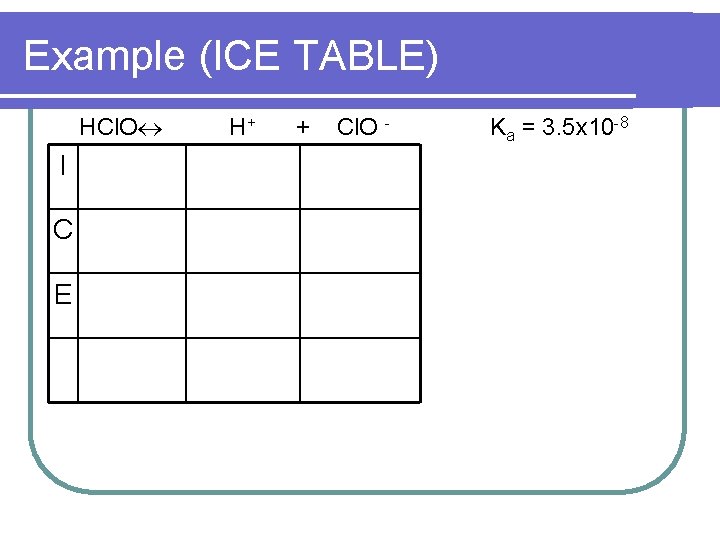
Example (ICE TABLE) HCl. O I C E H+ + Cl. O - Ka = 3. 5 x 10 -8
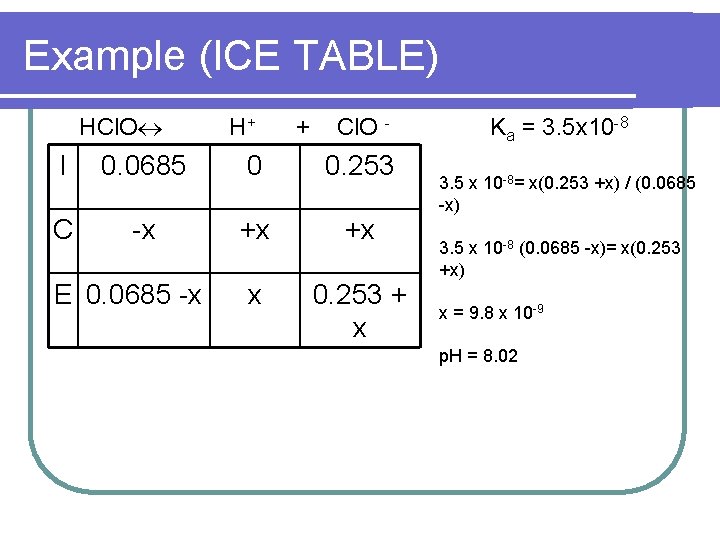
Example (ICE TABLE) HCl. O H+ + Cl. O - I 0. 0685 0 0. 253 C -x +x +x x 0. 253 + x E 0. 0685 -x Ka = 3. 5 x 10 -8 3. 5 x 10 -8= x(0. 253 +x) / (0. 0685 -x) 3. 5 x 10 -8 (0. 0685 -x)= x(0. 253 +x) x = 9. 8 x 10 -9 p. H = 8. 02
![Example (HH) l p. H = p. Ka + log [B]/[A] l p. H Example (HH) l p. H = p. Ka + log [B]/[A] l p. H](http://slidetodoc.com/presentation_image/114742f6d345599c5b692385bb6c2fc0/image-20.jpg)
Example (HH) l p. H = p. Ka + log [B]/[A] l p. H = 7. 45 + log (14. 4/3. 75) l p. H = 8. 03

Example (ICE TABLE) l l l l l What is the p. H of a solution containing 25 m. L of 0. 50 M CH 3 NO 3 is mixed with 75 m. L of 0. 30 M CH 3 NH 2 (Kb CH 3 NH 2 = 4. 38 x 10 -4) CH 3 NO 3 M = mol/L 0. 50 = mol / 25 12. 5 mmol / 100 m. L = 0. 125 M CH 3 NO 3 CH 3 NH 2 M = mol/L 0. 30 = mol / 75 22. 5 mmol/ 100 m. L = 0. 225 M CH 3 NH 2
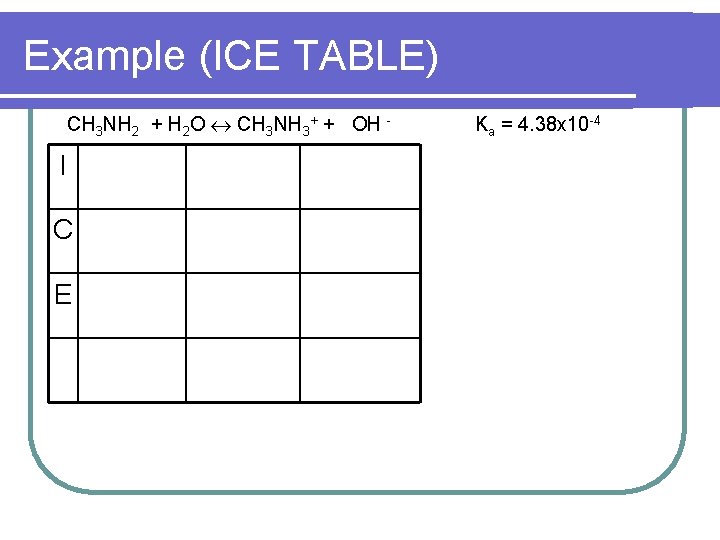
Example (ICE TABLE) CH 3 NH 2 + H 2 O CH 3 NH 3+ + OH - I C E Ka = 4. 38 x 10 -4
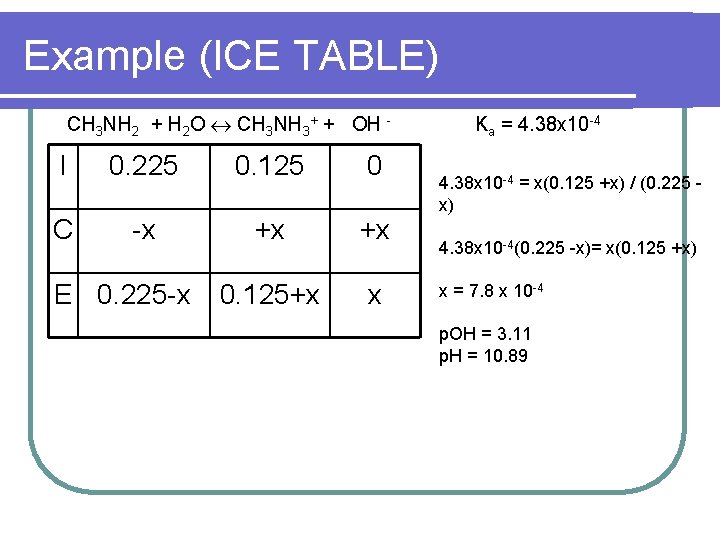
Example (ICE TABLE) CH 3 NH 2 + H 2 O CH 3 NH 3+ + OH - I 0. 225 0. 125 0 C -x +x +x 0. 125+x x E 0. 225 -x Ka = 4. 38 x 10 -4 = x(0. 125 +x) / (0. 225 x) 4. 38 x 10 -4(0. 225 -x)= x(0. 125 +x) x = 7. 8 x 10 -4 p. OH = 3. 11 p. H = 10. 89
![Example (HH) l p. H = p. Ka + log [B]/[A] l p. H Example (HH) l p. H = p. Ka + log [B]/[A] l p. H](http://slidetodoc.com/presentation_image/114742f6d345599c5b692385bb6c2fc0/image-24.jpg)
Example (HH) l p. H = p. Ka + log [B]/[A] l p. H = 10. 65 + log (22. 5/12/5) l p. H = 10. 91

Example l Calculate the mass of Na. F that must be added to 1000. 0 ml of 0. 50 M HF to form a solution with a p. H of 4. 00. Ka = 7. 2 x 10 -4
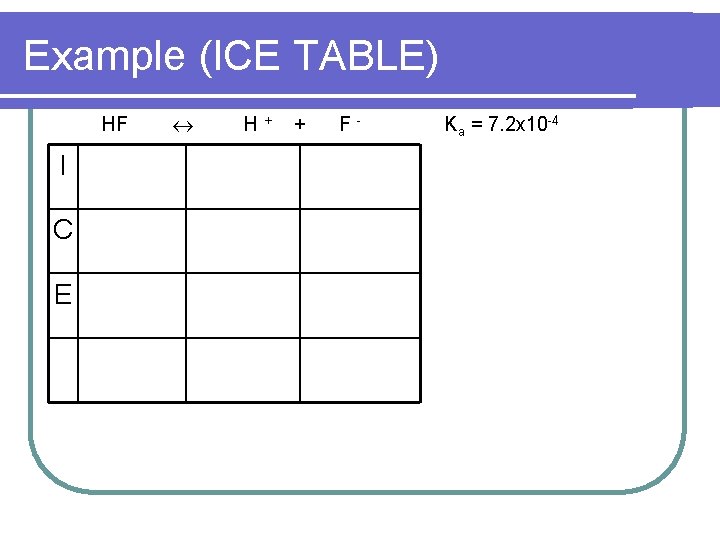
Example (ICE TABLE) HF I C E H+ + F- Ka = 7. 2 x 10 -4
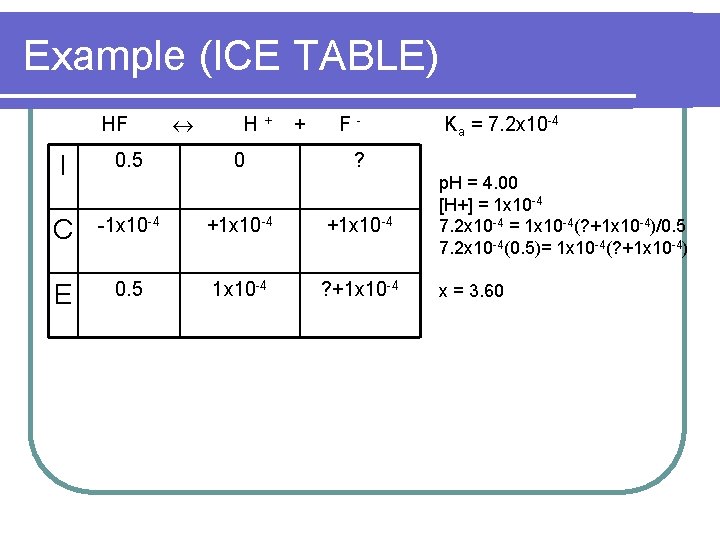
Example (ICE TABLE) HF H+ 0 + F- I 0. 5 C -1 x 10 -4 +1 x 10 -4 E 0. 5 1 x 10 -4 ? +1 x 10 -4 Ka = 7. 2 x 10 -4 ? p. H = 4. 00 [H+] = 1 x 10 -4 7. 2 x 10 -4 = 1 x 10 -4(? +1 x 10 -4)/0. 5 7. 2 x 10 -4(0. 5)= 1 x 10 -4(? +1 x 10 -4) x = 3. 60
![Example (ICE TABLE) l 3. 60 M [F-] = 3. 60 M Na. F Example (ICE TABLE) l 3. 60 M [F-] = 3. 60 M Na. F](http://slidetodoc.com/presentation_image/114742f6d345599c5b692385bb6c2fc0/image-28.jpg)
Example (ICE TABLE) l 3. 60 M [F-] = 3. 60 M Na. F l M = mol/L l 3. 60 = mol / 0. 100 L l 3. 60 mol Na. F x 41. 99 g Na. F l 1 mol Na. F l 151 g Na. F
![Example (HH) l p. H = p. Ka + log [B]/[A] l 4. 00 Example (HH) l p. H = p. Ka + log [B]/[A] l 4. 00](http://slidetodoc.com/presentation_image/114742f6d345599c5b692385bb6c2fc0/image-29.jpg)
Example (HH) l p. H = p. Ka + log [B]/[A] l 4. 00 = 3. 14 + log (x/0. 5) l 0. 86 = log (x/0. 5) l Antilog(0. 86) = (x/0. 5) l 7. 24 = x/0. 5 l X = 3. 62 mol 152 g Na. F
- Slides: 29