Common Inspection Findings and Inspections postJuly 2012 March









- Slides: 12

Common Inspection Findings and Inspections post-July 2012 March 2013

GPv. P inspection findings July 2012 to March 2013 • 39 inspections – 16 critical findings (from 11 inspections) – 121 major findings (from 35 inspections) – 92 other findings (from 33 inspections) • Type of company – 16 innovative Pharma – 21 generics – 2 others

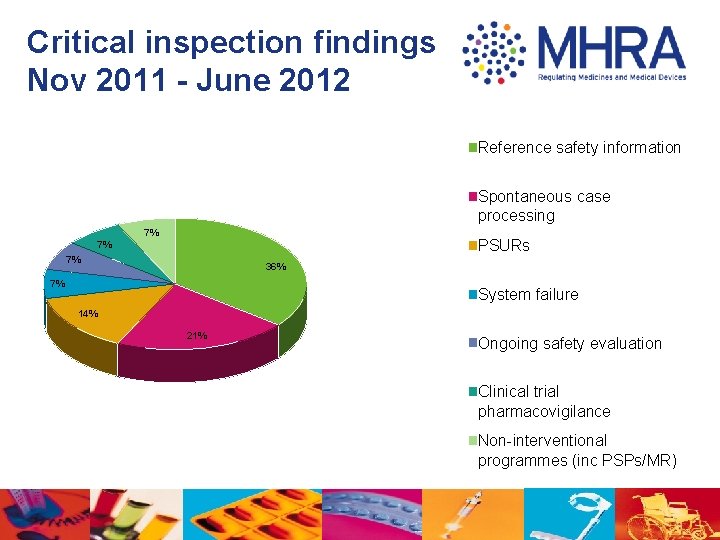
Critical inspection findings Nov 2011 - June 2012 Reference safety information Spontaneous case processing 7% PSURs 7% 7% 36% 7% System failure 14% 21% Ongoing safety evaluation Clinical trial pharmacovigilance Non-interventional programmes (inc PSPs/MR)

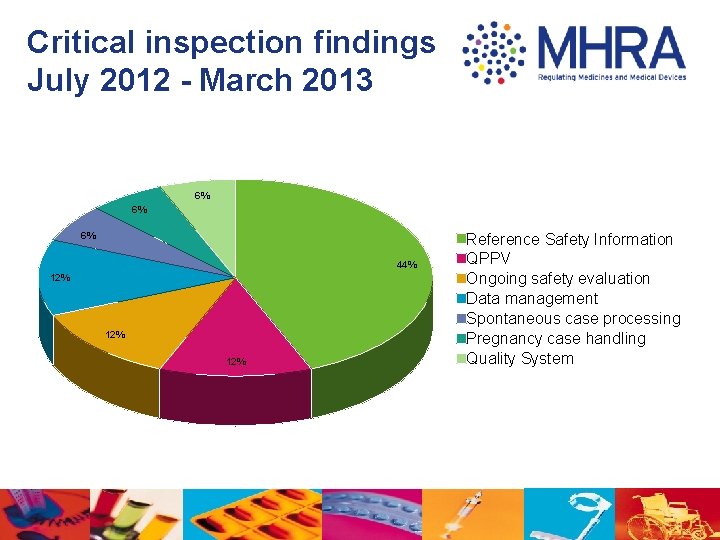
Critical inspection findings July 2012 - March 2013 6% 6% 6% 44% 12% 12% Reference Safety Information QPPV Ongoing safety evaluation Data management Spontaneous case processing Pregnancy case handling Quality System

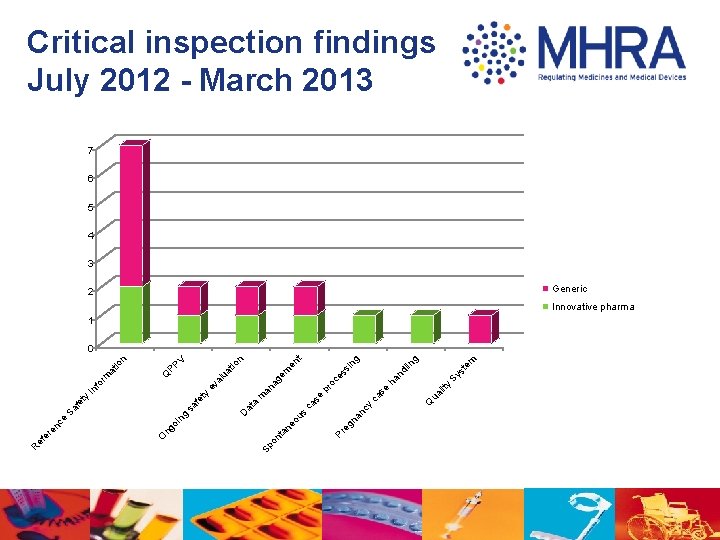
g in st em Sy nd l ha y ua lit Q se ca g in ss ce pr o t en n V ua tio em ag m an se ca nc y na eg Pr eo us ta n on Sp at a D al ev PP Q n at io m or In f ty sa fe ty fe Sa g oi n ng O e nc re ef e R Critical inspection findings July 2012 - March 2013 7 6 5 4 3 2 Generic 1 Innovative pharma 0

Reference Safety Information (RSI) • Failure to update Sm. PCs with safety information identified through company signal detection processes • Failure to submit variations requested by a Competent Authority • Failure to implement updated PILs in product packs • Failure to update Sm. PCs and PILs on company websites • Creation of core safety information documents, but failure to assess impact on Sm. PCs

RSI: What’s new on inspection • No major changes with new legislation (the key obligations remain the same) • Some additional questions regarding tracking the timeliness of safety variation submission (a description of this process is required for the PSMF) • When additional monitoring and the black symbol ▼ is fully implemented, inspectors will examine compliance with the new requirements • Continued non-compliance has been identified in this area

Ongoing Safety Evaluation • Failure to conduct signal detection at an appropriate periodicity (or at all) • Failure to include all sources of safety information in signal detection activities (e. g. all non-serious, invalid or solicited) • Failure to conduct cumulative review of all sources of safety information • Failure to include other sources (non-ICSR data) in signal detection activities, such as relevant publications • Signal detection methodologies poorly described in SOPs / WIs

Ongoing Safety Evaluation: What’s new on inspection? • For MAs where no PSUR is produced or PSURs are produced infrequently, the robustness of ongoing safety evaluation will be even more important • Have procedural documents and templates been updated, where required, to reflect the new requirements and definitions contained in the new legislation and in GVPs? • Is there a process in place for tracking all validation, prioritisation, assessment, timelines, decisions, actions, plans, reporting as well as all other key steps for confirmed and unconfirmed signals

Potential Source of Major Findings? (1) • Pharmacovigilance System Summary submitted with new applications or MA renewals, but no PSMF produced at that time • Lack of procedures describing PSMF production and maintenance • Failure to submit variations to update the DDPS for existing marketing authorisations, when a system summary has been submitted in a new MAA but not yet introduced for all products

Potential Source of Major Findings? (2) • PSMF not containing the required detail described in GVP Module II, e. g. inadequate description of sources of safety data or the approach used to plan audits • PSMF not containing sufficient information about contractors and/or partners N. B. An adequate description of the system can aid the QPPV to gain oversight and may lead inspectors to request fewer documents on inspection

Crown copyright 2013 The materials featured within this MHRA presentation are subject to Crown copyright protection. Any other copy or use of Crown copyright materials featured in this presentation, in any form or medium, is subject to prior approval of the MHRA which has Delegated Authority from Her Majesty's Stationery Office (HMSO) to administer Crown copyright for MHRA originated material. Applications should be in writing, clearly stating the proposed use/reuse of the information, and should be sent to the MHRA at the following address: Conference and Education Function, MHRA, 151 Buckingham Palace Road, London SW 1 W 9 SZ. Fax 020 3080 7522 or e-mail speakers@mhra. gsi. gov. uk. You may not sell or resell any information reproduced to any third party without prior agreement. The permission to reproduce Crown copyright protected material does not extend to any material in this pack which is subject to a separate licence or is the copyright of a third party. Authorisation to reproduce such material must be obtained from the copyright holders concerned.