Common Bacteria by Site of Infection BetaLactam Structure

Common Bacteria by Site of Infection
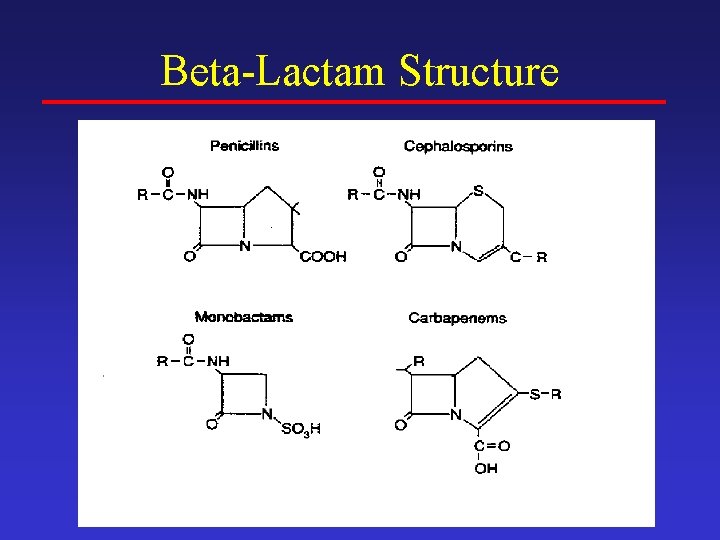
Beta-Lactam Structure

ANTIBIOTICS ACTING ON CELL WALL OF BACTERIA • Beta lactams: • Penicillins, cephalosporins, carbapenems, monobactam • Glycopeptides: • Vancomycin, teicoplanin

PENICILLIN IS GENERALLY VERY SAFE BUT…. • Allergic reactions not uncommon-rashes • Most severe reaction being anaphylaxis • A history of anaphylaxis, urticaria, or rash immediately after penicillin indicates risk of immediate hypersensitivity after a further dose of any penicillin or cephalosporin (therefore these must be avoided) • Allergy is not dependent on the dose given ie, a small dose could cause anaphylaxis • Very high doses of penicillin cause neurotoxicity • Never give penicillin intrathecally

What antibiotics can be used in penicillin allergy? • Macrolides: erythromycin, clarithromycin • (mainly gram positive cover) • Quinolones: ciprofloxacin, levofloxacin (mainly gram positive cover) • Glycopeptides (serious infections) • Fusidic acid, rifampicin, clindamycin (mainly gram positive)

-Lactam l l l Same MOA: Inhibit cell wall synthesis Bactericidal (except against Enterococcus sp. ) Short elimination half-life Primarily renally eliminated (except nafcillin, oxacillin, ceftriaxone, cefoperazone) Cross-allergenicity - except aztreonam

-Lactams Adverse Effects • Hypersensitivity – 3 to 10 % Ø Higher incidence with parenteral administration or procaine formulation Ø Mild to severe allergic reactions – rash to anaphylaxis and death Ø Cross-reactivity exists among all penicillins and even other -lactams Ø Desensitization is possible

-lactams Pharmacology • Absorption: Variable depending on product • Distribution Widely distributed into tissues and fluids Ø Pens only get into CSF in the presence of inflamed meninges; parenteral 3 rd and 4 th generation cephs, meropenem, and aztreonam penetrate the CSF Ø • Elimination most eliminated primarily by the kidney, dosage adj required in the presence of renal insufficiency Ø Nafcillin, oxacillin, ceftriaxone-eliminated by the liver Ø ALL -lactams have short elimination half-lives except for a few cephalosporins (ceftriaxone) Ø

-Lactams Adverse Effects • Neurologic – especially with penicillins and carbapenems (imipenem and meropenem) Ø Especially in patients receiving high doses in the presence of renal insufficiency Ø Irritability, confusion, seizures • Hematologic Ø Leukopenia, neutropenia, thrombocytopenia – prolonged therapy (> 2 weeks)

-Lactams Adverse Effects • Gastrointestinal Ø Increased LFTs, nausea, vomiting, diarrhea, pseudomembranous colitis (C. difficile diarrhea) • Interstitial Nephritis Ø Cellular infiltration in renal tubules (Type IV hypersensitivity reaction – characterized by abrupt increase in serum creatinine; can lead to renal failure Ø Especially with methicillin or nafcillin

Natural Penicillins (penicillin G, penicillin VK) Gram-positive Gram-negative pen-susc S. pneumoniae Group A/B/C/G strep viridans streptococci Enterococcus Neisseria sp. Other Treponema pallidum (syphilis) Anaerobes Above the diaphragm Clostridium sp.

Penicillinase-Resistant Penicillins (nafcillin, oxacillin, methicillin) Developed to overcome the penicillinase enzyme of S. aureus which inactivated natural penicillins Gram-positive Methicillin-susceptible S. aureus Penicillin-susceptible strains of Streptococci

Aminopenicillins (ampicillin, amoxicillin) Developed to increase activity against gramnegative aerobes Gram-positive Gram-negative pen-susc S. aureus Pen-susc streptococci viridans streptococci Enterococcus sp. Listeria monocytogenes Proteus mirabilis Salmonella, some E. coli L- H. influenzae

Carboxypenicillins (carbenicillin, ticarcillin) Developed to further increase activity against resistant gram-negative aerobes Gram-positive marginal Gram-negative Proteus mirabilis Salmonella, Shigella some E. coli L- H. influenzae Enterobacter sp. Pseudomonas aeruginosa

Ureidopenicillins (piperacillin, azlocillin) Developed to further increase activity against resistant gram-negative aerobes Gram-positive Gram-negative viridans strep Group strep some Enterococcus Proteus mirabilis Salmonella, Shigella E. coli L- H. influenzae Enterobacter sp. Pseudomonas aeruginosa Serratia marcescens some Klebsiella sp. Anaerobes Fairly good activity

-Lactamase Inhibitor Combos (Unasyn, Augmentin, Timentin, Zosyn) Developed to gain or enhance activity against lactamase producing organisms (some better than others). Provides some or good activity against: Gram-positive Gram-negative S. aureus (MSSA) H. influenzae E. coli Proteus sp. Klebsiella sp. Neisseria gonorrhoeae Moraxella catarrhalis Anaerobes Bacteroides sp.

Classification and Spectrum of Activity of Cephalosporins • Divided into 4 major groups called “Generations” • Are divided into Generations based on Ø antimicrobial activity Ø resistance to beta-lactamase

First Generation Cephalosporins Best activity against gram-positive aerobes, with limited activity against a few gramnegative aerobes Gram-positive Gram-negative meth-susc S. aureus pen-susc S. pneumoniae Group A/B/C/Gstreptococci viridans streptococci E. coli K. pneumoniae P. mirabilis

Second Generation Cephalosporins Spectrum of Activity Gram-positive Gram-negative meth-susc S. aureus pen-susc S. pneumoniae Group A/B/C/G strep viridans streptococci E. coli K. pneumoniae P. mirabilis H. influenzae M. catarrhalis Neisseria sp.

Second Generation Cephalosporins Spectrum of Activity The cephamycins (cefoxitin and cefotetan) are the only 2 nd generation cephalosporins that have activity against anaerobes Anaerobes Bacteroides fragilis group

Third Generation Cephalosporins Spectrum of Activity • In general, are even less active against grampositive aerobes, but have greater activity against gram-negative aerobes • Ceftriaxone and cefotaxime have the best activity against gram-positive aerobes, including pen-resistant S. pneumoniae

Third Generation Cephalosporins Spectrum of Activity Gram-negative aerobes E. coli, K. pneumoniae, P. mirabilis H. influenzae, M. catarrhalis, N. gonorrhoeae (including beta-lactamase producing); N. meningitidis Citrobacter sp. , Enterobacter sp. , Acinetobacter sp. Morganella morganii, Serratia marcescens, Providencia Pseudomonas aeruginosa (ceftazidime and cefoperazone)

Fourth Generation Cephalosporins • 4 th generation cephalosporins for 2 reasons Ø Extended w w spectrum of activity gram-positives: similar to ceftriaxone gram-negatives: similar to ceftazidime, including Pseudomonas aeruginosa; also covers beta-lactamase producing Enterobacter sp. against -lactamases; poor inducer of extended-spectrum -lactamases Ø Stability • Only cefepime is currently available

Carbapenems (Imipenem, Meropenem and Ertapenem) • Most broad spectrum of activity of all antimicrobials • Have activity against gram-positive and gramnegative aerobes and anaerobes • Bacteria not covered by carbapenems include MRSA, VRE, coagulase-negative staph, C. difficile, Nocardia

Monobactams Spectrum of Activity Aztreonam bind preferentially to PBP 3 of gram-negative aerobes; has little to no activity against gram-positives or anaerobes Gram-negative E. coli, K. pneumoniae, P. mirabilis, S. marcescens H. influenzae, M. catarrhalis Enterobacter, Citrobacter, Providencia, Morganella Salmonella, Shigella Pseudomonas aeruginosa

Fluoroquinolones • Novel group of synthetic antibiotics • The fluorinated quinolones (FQs) represent a major therapeutic advance: Broad spectrum of activity Ø bioavailability, tissue penetration, prolonged Ø

FQs Spectrum of Activity Gram-positive – newer FQs with enhanced potency • Methicillin-susceptible Staphylococcus aureus • Streptococcus pneumoniae (including PRSP) • Group A/B/C/G and viridans streptococci – limited activity • Enterococcus sp. – limited activity

FQs Spectrum of Activity Gram-Negative – all FQs have excellent activity (cipro=levo>gati>moxi) • Enterobacteriaceae – including E. coli, Klebsiella sp, Enterobacter sp, Proteus sp, Salmonella, Shigella, Serratia marcescens, etc. • H. influenzae, M. catarrhalis, Neisseria sp. • Pseudomonas aeruginosa – significant resistance has emerged; ciprofloxacin and levofloxacin with best activity

FQs Spectrum of Activity Atypical Bacteria – all FQs have excellent activity against atypical bacteria including: • • Legionella pneumophila - DOC Chlamydia sp. Mycoplasma sp. Ureaplasma urealyticum Other Bacteria – Mycobacterium tuberculosis, Bacillus anthracis

Fluoroquinolones Pharmacology • Concentration-dependent bacterial killing • Absorption Ø Most FQs have good bioavailability after oral administration • Distribution Extensive tissue distribution – prostate; liver; lung; skin/soft tissue and bone; urinary tract Ø Minimal CSF penetration Ø • Elimination – renal and hepatic; not removed by HD

Fluoroquinolones Adverse Effects • Gastrointestinal – 5 % Ø Nausea, vomiting, diarrhea, dyspepsia • Central Nervous System Ø Headache, agitation, insomnia, dizziness, rarely, hallucinations and seizures (elderly) • Hepatotoxicity Ø LFT elevation (led to withdrawal of trovafloxacin) • Phototoxicity (uncommon with current FQs) Ø More common with older FQs (halogen at position 8) • Cardiac Variable prolongation in QTc interval Ø Led to withdrawal of grepafloxacin, sparfloxacin Ø

Fluoroquinolones Adverse Effects • Articular Damage Ø Arthopathy including articular cartilage damage, arthralgias, and joint swelling Ø Observed in toxicology studies in immature dogs Ø Led to contraindication in pediatric patients and pregnant or breastfeeding women Ø Risk versus benefit • Other adverse reactions: tendon rupture, dysglycemias, hypersensitivity

Macrolides • Erythromycin is a naturally-occurring macrolide derived from Streptomyces erythreus – problems with acid lability, narrow spectrum, poor GI intolerance, short elimination half-life • Structural derivatives include clarithromycin and azithromycin: Ø Ø Ø Broader spectrum of activity Improved PK properties – better bioavailability, better tissue penetration, prolonged half-lives Improved tolerability

Macrolides Mechanism of Action Ø Inhibits protein synthesis by reversibly binding to the 50 S ribosomal subunit § Suppression of RNA-dependent protein synthesis Ø Macrolides typically display bacteriostatic activity, but may be bactericidal when present at high concentrations against very susceptible organisms

Macrolide Spectrum of Activity Gram-Positive Aerobes – erythromycin and clarithromycin display the best activity (Clarithro>Erythro>Azithro) • Methicillin-susceptible Staphylococcus aureus • Streptococcus pneumoniae (only PSSP) – resistance is developing • Group A/B/C/G and viridans streptococci • Bacillus sp. , Corynebacterium sp.

Macrolide Spectrum of Activity Gram-Negative Aerobes – newer macrolides with enhanced activity (Azithro>Clarithro>Erythro) • H. influenzae (not erythro), M. catarrhalis, Neisseria sp. , Campylobacter jejuni, Bordetella pertussis • Do NOT have activity against any Enterobacteriaceae or Pseudomonas

Macrolide Spectrum of Activity Anaerobes – activity against upper airway anaerobes Atypical Bacteria – all macrolides have excellent activity against atypical bacteria including: • • Legionella pneumophila - DOC Chlamydia sp. Mycoplasma sp. Ureaplasma urealyticum Other Bacteria – Mycobacterium avium complex (MAC – only A and C), Treponema pallidum, Campylobacter, Borrelia, Bordetella, Brucella. Pasteurella

Macrolides Pharmacology Absorption Ø Erythromycin – variable absorption (15 -45%); food may decrease the absorption Base: destroyed by gastric acid; enteric coated • Esters and ester salts: more acid stable • Ø Clarithromycin – acid stable and well-absorbed, 55% bioavailable regardless of presence of food Ø Azithromycin –acid stable; 38% bioavailable; food decreases absorption of capsules

Macrolides Pharmacology Distribution Extensive tissue and cellular distribution – clarithromycin and azithromycin with extensive penetration Ø Minimal CSF penetration Ø Elimination Clarithromycin is the only macrolide partially eliminated by the kidney (18% of parent and all metabolites); requires dose adjustment when Cr. Cl < 30 ml/min Ø Hepatically eliminated: ALL Ø Variable elimination half-lives (1. 4 hours for erythro; 3 to 7 hours for clarithro; 68 hours for azithro) Ø

Macrolides Adverse Effects • Gastrointestinal – up to 33 % Ø Nausea, vomiting, diarrhea, dyspepsia Ø Most common with erythro; less with new agents • Cholestatic hepatitis - rare Ø> 1 to 2 weeks of erythromycin estolate • Thrombophlebitis – IV Erythro and Azithro Ø Dilution of dose; slow administration • Other: ototoxicity (high dose erythro in patients with RI); QTc prolongation; allergy

Macrolides Drug Interactions Erythromycin and Clarithromycin ONLY– are inhibitors of cytochrome p 450 system in the liver; may increase concentrations of: Theophylline Carbamazepine Cyclosporine Phenytoin Warfarin Digoxin, Disopyramide Valproic acid Terfenadine, Astemizole Cisapride Ergot alkaloids

Aminoglycosides Mechanism of Action • Multifactorial, but ultimately involves inhibition of protein synthesis • Irreversibly bind to 30 S ribosomes – must bind to and diffuse through outer membrane and cytoplasmic membrane and bind to the ribosome – disrupt the initiation of protein synthesis, decreases overall protein synthesis, and produces misreading of m. RNA • Are bactericidal

Aminoglycosides Spectrum of Activity Gram-Positive Aerobes most S. aureus and coagulase-negative staph (but not DOC) viridans streptococci (in combination with a cell-wall agent) Enterococcus sp. (only in combination with a cell-wall agent) Gram-Negative Aerobes (not streptomycin) E. coli, K. pneumoniae, Proteus sp. Acinetobacter, Citrobacter, Enterobacter sp. Morganella, Providencia, Serratia, Salmonella, Shigella Pseudomonas aeruginosa (amik>tobra>gent) Mycobacteria – tuberculosis - streptomycin – atypical - streptomycin or amikacin

Aminoglycosides Pharmacology • Absorption - poorly absorbed from gi tract • Distribution – primarily in extracellular fluid volume; are widely distributed into body fluids but NOT the CSF – distribute poorly into adipose tissue, use LBW for dosing • Elimination – eliminated unchanged by the kidney via glomerular filtration; 85 -95% of dose – elimination half-life dependent on renal fxn w w normal renal function - 2. 5 to 4 hours impaired renal function - prolonged

Aminoglycosides Adverse Effects Nephrotoxicity – nonoliguric azotemia due to proximal tubule damage; increase in BUN and serum Cr; reversible if caught early – risk factors: prolonged high troughs, long duration of therapy (> 2 weeks), underlying renal dysfunction, elderly, other nephrotoxins Ototoxicity – 8 th cranial nerve damage - vestibular and auditory toxicity; irreversible and saturable – vestibular: dizziness, vertigo, ataxia – auditory: tinnitus, decreased hearing – risk factors: same as for nephrotoxicity

Vancomycin Mechanism of Action • Inhibits bacterial cell wall synthesis at a site different than beta-lactams • Bactericidal (except for Enterococcus)

Vancomycin Spectrum of Activity Gram-positive bacteria – Methicillin-Susceptible AND Methicillin-Resistant S. aureus and coagulase-negative staphylococci – Streptococcus pneumoniae (including PRSP), viridans streptococcus, Group A/B/C/G streptococcus – Enterococcus sp. – Corynebacterium, Bacillus. Listeria, Actinomyces – Clostridium sp. (including C. difficile), Peptococcus, Peptostreptococcus No activity against gram-negative aerobes or anaerobes

Vancomycin Pharmacology • Absorption – absorption from GI tract is negligible after oral administration except in patients with intense colitis – Use IV therapy for treatment of systemic infection • Distribution – widely distributed into body tissues and fluids, including adipose tissue; use TBW for dosing – inconsistent penetration into CSF, even with inflamed meninges • Elimination – primarily eliminated unchanged by the kidney via glomerular filtration – elimination half-life depends on renal function

Vancomycin Clinical Uses • Infections due to methicillin-resistant staph including bacteremia, empyema, endocarditis, peritonitis, pneumonia, skin and soft tissue infections, osteomyelitis • Serious gram-positive infections in -lactam allergic patients • Infections caused by multidrug resistant bacteria • Endocarditis or surgical prophylaxis in select cases • Oral vancomycin for refractory C. difficile colitis

Vancomycin Adverse Effects Red-Man Syndrome – flushing, pruritus, erythematous rash on face and upper torso – related to RATE of intravenous infusion; should be infused over at least 60 minutes – resolves spontaneously after discontinuation – may lengthen infusion (over 2 to 3 hours) or pretreat with antihistamines in some cases

Vancomycin Adverse Effects • Nephrotoxicity and Ototoxicity – rare with monotherapy, more common when administered with other nephro- or ototoxins – risk factors include renal impairment, prolonged therapy, high doses, ? high serum concentrations, other toxic meds • Dermatologic - rash • Hematologic - neutropenia and thrombocytopenia with prolonged therapy • Thrombophlebitis

Oxazolidinones • Linezolid (Zyvox®) is the first available agent which received FDA approval in April 2000; available PO and IV • Developed in response to need for agents with activity against resistant gram-positives (MRSA, VRE)

Linezolid Mechanism of Action • Binds to the 50 S ribosomal subunit near to surface interface of 30 S subunit – causes inhibition of 70 S initiation complex which inhibits protein synthesis • Bacteriostatic (cidal against some bacteria)

Linezolid Spectrum of Activity Gram-Positive Bacteria – Methicillin-Susceptible, Methicillin-Resistant AND Vancomycin-Resistant Staph aureus and coagulasenegative staphylococci – Streptococcus pneumoniae (including PRSP), viridans streptococcus, Group streptococcus – Enterococcus faecium AND faecalis (including VRE) – Bacillus. Listeria, Clostridium sp. (except C. difficile), Peptostreptococcus, P. acnes Gram-Negative Aerobes – relatively inactive Atypical Bacteria – Mycoplasma, Chlamydia, Legionella

Linezolid Pharmacology • Concentration-independent bactericidal activity • Absorption – 100% bioavailable • Distribution – readily distributes into wellperfused tissue; CSF penetration 70% • Elimination – both renally and nonrenally, but primarily metabolized; t½ is 4. 4 to 5. 4 hours; no adjustment for RI; not removed by HD

Linezolid Adverse Effects • Gastrointestinal – nausea, vomiting, diarrhea (6 to 8 %) • Headache – 6. 5% • Thrombocytopenia – 2 to 4% – Most often with treatment durations of > 2 weeks – Therapy should be discontinued – platelet counts will return to normal

Linezolid (Zyvox®) Drug–Drug/Food interactions Linezolid is a reversible, nonselective inhibitor of monoamine oxidase. Tyramine rich foods, adrenergic drugs and serotonergic drugs should be avoided due to the potential drug-food and drug-drug interactions. A significant pressor response has been observed in normal adult subjects receiving linezolid and tyramine doses of more than 100 mg. Therefore, patients receiving linezolid need to avoid consuming large amounts of foods or beverages with high tyramine content.

Clindamycin Mechanism of Action Ø Inhibits protein synthesis by binding exclusively to the 50 S ribosomal subunit § Binds in close proximity to macrolides – competitive inhibition Ø Clindamycin typically displays bacteriostatic activity, but may be bactericidal when present at high concentrations against very susceptible organisms

Clindamycin Spectrum of Activity Gram-Positive Aerobes • Methicillin-susceptible Staphylococcus aureus (MSSA) • Methicillin-resistant Staphylococcus aureus (MRSA) – some isolates • Streptococcus pneumoniae (only PSSP) – resistance is developing • Group and viridans streptococci

Clindamycin Spectrum of Activity Anaerobes – activity against Above the Diaphragm Anaerobes (ADA) Peptostreptococcus some Bacteroides sp Actinomyces Prevotella sp. Propionibacterium Fusobacterium Clostridium sp. (not C. difficile) Other Bacteria – Toxoplasmosis gondii, Malaria

Clindamycin Pharmacology Absorption – available IV and PO Ø Rapidly and completely absorbed (90%); food with minimal effect on absorption Distribution Ø Good serum concentrations with PO or IV Ø Good tissue penetration including bone; minimal CSF penetration Elimination Ø Clindamycin primarily metabolized by the liver; halflife is 2. 5 to 3 hours Ø Clindamycin is NOT removed during hemodialysis

Clindamycin Adverse Effects • Gastrointestinal – 3 to 4 % Ø Nausea, vomiting, diarrhea, dyspepsia • C. difficile colitis – one of worst offenders Ø Mild to severe diarrhea Ø Requires treatment with metronidazole • Hepatotoxicity - rare Ø Elevated transaminases • Allergy - rare

New Guys on the Block • Tigecycline (Tygacil®) • Daptomycin (Cubicin®)

Tigecycline Mechanism of Action Binds to the 30 S ribosomal subunit of susceptible bacteria, inhibiting protein synthesis.

Tigecycline Spectrum of Activity Broad spectrum of activity • Treatment of complicated skin and skin structure infections caused by susceptible organisms, including methicillin-resistant Staphylococcus aureus and vancomycin-sensitive Enterococcus faecalis; treatment of complicated intraabdominal infections

Tigecycline Pharmacokinetics • Metabolism: Hepatic, via glucuronidation, Nacetylation, and epimerization to several metabolites, each <10% of the dose • Half-life elimination: Single dose: 27 hours; following multiple doses: 42 hours • Excretion: Urine (33%; with 22% as unchanged drug); feces (59%; primarily as unchanged drug) – No dose adjustment required in renal dysfunction

Tigecycline Adverse Effects • • • >10%: Gastrointestinal: Nausea (25% to 30%), diarrhea (13%) 2% to 10%: Cardiovascular: Hypertension (5%), peripheral edema (3%), hypotension (2%) Central nervous system: Fever (7%), headache (6%), dizziness (4%), pain (4%), insomnia (2%) Dermatologic: Pruritus (3%), rash (2%) Endocrine: Hypoproteinemia (5%), hyperglycemia (2%), hypokalemia (2%) Hematologic: Thrombocythemia (6%), anemia (4%), leukocytosis (4%) Hepatic: SGPT increased (6%), SGOT increased (4%), alkaline phosphatase increased (4%), amylase increased (3%), bili increased (2%), LDH increased (4%) Neuromuscular & skeletal: Weakness (3%) Renal: BUN increased (2%) Respiratory: Cough increased (4%), dyspnea (3%)

Daptomycin Mechanism of Action Ø Daptomycin binds to components of the cell membrane of susceptible organisms and causes rapid depolarization, inhibiting intracellular synthesis of DNA, RNA, and protein. Ø Daptomycin is bactericidal in a concentrationdependent manner

Daptomycin Spectrum of Activity Gram-Positive Aerobes Treatment of complicated skin and skin structure infections caused by susceptible aerobic Gram-positive organisms; • Staphylococcus aureus bacteremia, including rightsided infective endocarditis caused by MSSA or MRSA

Daptomycin Pharmacokinetics • Absorption – available IV only • Half-life elimination: 8 -9 hours (up to 28 hours in renal impairment) • Excretion: Urine (78%; primarily as unchanged drug); feces (6%) • Dosage adjustment in renal impairment: – Clcr <30 m. L/minute: Administer dose q 48 hr

Daptomycin Adverse Effects • Hematologic: Anemia (2% to 13%) • Gastrointestinal: – Diarrhea (5% to 12%) – vomiting (3% to 12%) – constipation (6% to 11%)

FDA Categorization of Antibiotics in Pregnancy • Category A – Controlled studies in women fail to demonstrate a risk to the fetus in the first trimester (and there is no evidence of a risk in later trimesters), and the possibility of fetal harm appears remote. • Category B – Either animal-reproduction studies have not demonstrated a fetal risk but there are no controlled studies in pregnant women, or animal-reproduction studies have shown an adverse effect (other than a decrease in fertility) that was not confirmed in controlled studies in women in the first trimester (and there is no evidence of a risk in later trimesters). • Category C – Either studies in animals have revealed adverse effects on the fetus (teratogenic or embryocidal or other) and there are no controlled studies in women, or studies in women and animals are not available. Drugs should be given only if the potential benefit justifies the potential risk to the fetus. • Category D – There is positive evidence of human fetal risk, but the benefits from use in pregnant women may be acceptable despite the risk (e. g. , if the drug is needed in a life-threatening situation or for a serious disease for which safer drugs cannot be used or are ineffective). • Category X – Studies in animals or human beings have demonstrated fetal abnormalities, or there is evidence of fetal risk based on human experience or both, and the risk of the use of the drug in pregnant women clearly outweighs any possible benefit. The drug is contraindicated in women who are or may become pregnant.

Antibiotics in Pregnancy FDA Category Antibiotics in Category A B Penicillins, Cephalosporins, Carbapenems (except Imipenem), Daptomycin, Vancomycin (oral), Clindamycin, Erythromycin, Azithromycin, Metronidazole (avoid first trimester), Nitrofurantoin, Acyclovir, Amphoterocin B, Ethambutol C Quinolones, Chloramphenicol, Clarithromycin, Imipenem, Linezolid, Trimethoprim/Sulfa (D if used near term), Vancomycin (IV), Rifampin, INH, PZA, PAS, Fluconazole, Caspofungin D Tetracyclines (Doxy, Tige, Mino), Voriconazole, Aminoglycosides (some put gentamicin as a category C) X Ribavarin
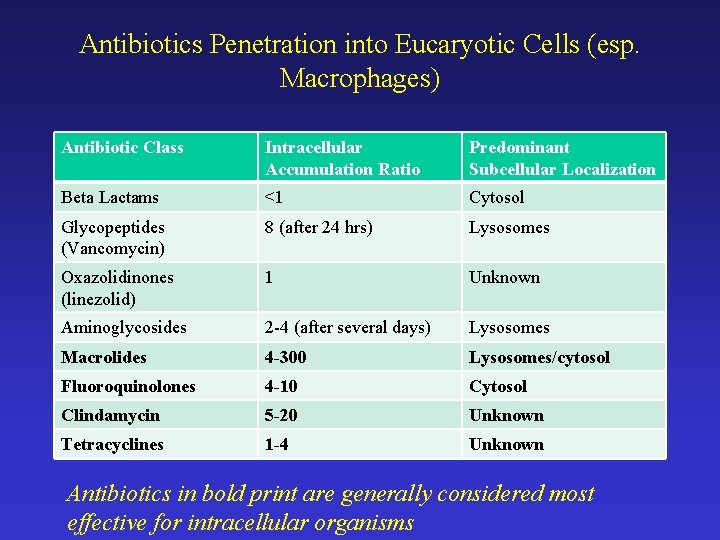
Antibiotics Penetration into Eucaryotic Cells (esp. Macrophages) Antibiotic Class Intracellular Accumulation Ratio Predominant Subcellular Localization Beta Lactams <1 Cytosol Glycopeptides (Vancomycin) 8 (after 24 hrs) Lysosomes Oxazolidinones (linezolid) 1 Unknown Aminoglycosides 2 -4 (after several days) Lysosomes Macrolides 4 -300 Lysosomes/cytosol Fluoroquinolones 4 -10 Cytosol Clindamycin 5 -20 Unknown Tetracyclines 1 -4 Unknown Antibiotics in bold print are generally considered most effective for intracellular organisms

• Sulfonamide or sulphonamide is the basis of several groups of drugs. The original antibacterial sulfonamides (sometimes called sulfa drugs or sulpha drugs) are synthetic antimicrobial agents that contain the sulfonamide group. Some sulfonamides are also devoid of antibacterial activity, e. g. , the anticonvulsant sultiame. The sulfonylureas and thiazide diuretics are newer drug groups based on the antibacterial sulfonamides
![• Allergies to sulfonamide are common, [3] hence medications containing sulfonamides are prescribed • Allergies to sulfonamide are common, [3] hence medications containing sulfonamides are prescribed](http://slidetodoc.com/presentation_image_h/1d1964c6224a39962ca8acb74f5beceb/image-76.jpg)
• Allergies to sulfonamide are common, [3] hence medications containing sulfonamides are prescribed carefully. It is important to make a distinction between sulfa drugs and other sulfur-containing drugs and additives, such as sulfates and sulfites, which are chemically unrelated to the sulfonamide group, and do not cause the same hypersensitivity reactions seen in the sulfonamides. • Because sulfonamides displace bilirubin from albumin, kernicterus (brain damage due to excess bilirubin) is an important


Sulfonamides: antimicrobial activity • • • Gram positive and negative bacteria Nocardia, chlamydia trachomatis Some protoza Some enteric bacteria Rickettisiae stimulated!

Sulfonamides: clinical uses • Oral absorbable agents – Sulfisoxazole, sulfamethoxazole • To treat urinary tract infection – Sulfadiazine: toxoplasmosis – Sulfadoxine: long acting, in a combination for treatment of malaria • Oral nonabsorbable agents – Ulcerative colitis, enteritis, other inflammatory bowel disease • Topical agents – Sulfacetamide: ophthalemic – Mafenide & silver sulfadiazine: topically

Sulfonamides: adverse reactions • Cross allergenic sulfonamide drugs: – Thiazide, furosemide, diazoxide, sulfonylurea hypoglycemic agents, and others – Fever, skin rashes, exfoliative dermatitis, photosensivity, urticaria, nausea, vomiting, diarrhea – Stevens-Johnson syndrom • Urinary tract disturbances – Crystalluria, hemturia, obstruction • Hematopoietic disturbance – – Hemolytic or aplastic anemia Granulocytopenia, thrombocytopenia, leukmoid reaction Hemolysis in G-6 PDH deficient patients Kernicterus in newborn of mothers have taken near the end of pergnancy

Clinical use • Oral trimethoprim – Acute urinary infection • Oral trimethoprim-sulfamethoxazole – P jiroveci pneumonia, shigellosis, systemic salmonella infection, complicated urinary tract infection, – Active against many respiratory pathogens • Intravenous trimethoprim-sulfamethoxazole – Gram negative sepsis, pneumocystis pneumonia – Shigllosis, typhoid fever • Oral pryrimethamine with sulfanamide – With sulfadiazine in Leishmaniasis, toxoplasmosis – With sulfadoxine in malaria

Adverse effects • • Megaloblastic anemia Leukopenia, granulocytopenia Can be prevented by folinic acid The AIDS patients have high frequency of unwanted reactions
- Slides: 82