COMBO A Combination of SirolimousEluting Biodegradable Polymer and

COMBO: A Combination of Sirolimous-Eluting Biodegradable Polymer and Anti-CD 34 Antibody Coated Stent Mitchell W. Krucoff MD, FACC Professor of Medicine / Cardiology Duke University Medical Center Director, Cardiovascular Devices Unit Duke Clinical Research Institute
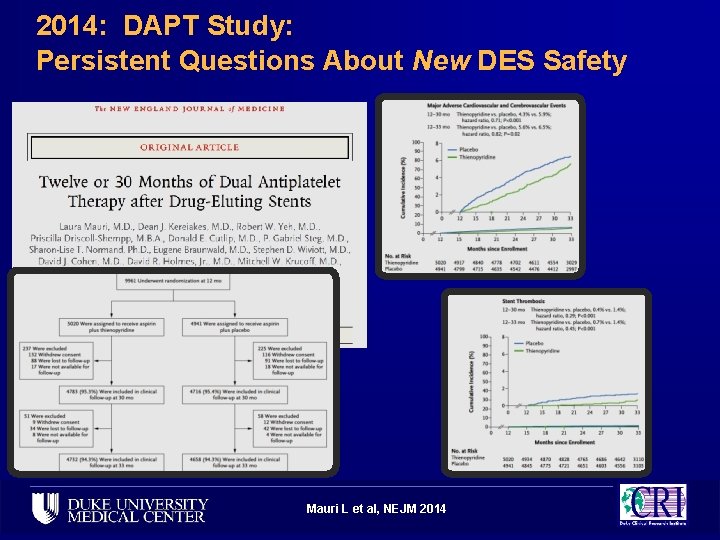
2014: DAPT Study: Persistent Questions About New DES Safety Mauri L et al, NEJM 2014

DES: Unmet medical needs Ø First 30 day safety Ø Very early DAPT interruption Ø VLST & duration of DAPT Ø Thrombotic lesions (ACS, STEMI)
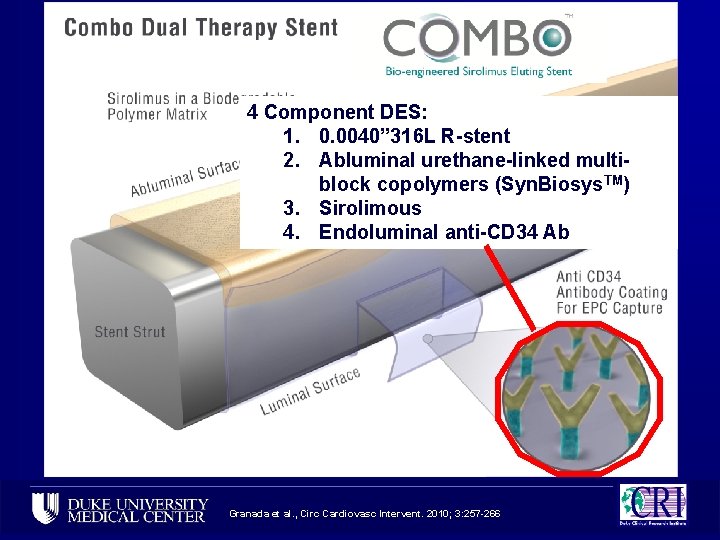
4 Component DES: 1. 0. 0040” 316 L R-stent 2. Abluminal urethane-linked multiblock copolymers (Syn. Biosys. TM) 3. Sirolimous 4. Endoluminal anti-CD 34 Ab Granada et al. , Circ Cardiovasc Intervent. 2010; 3: 257 -266
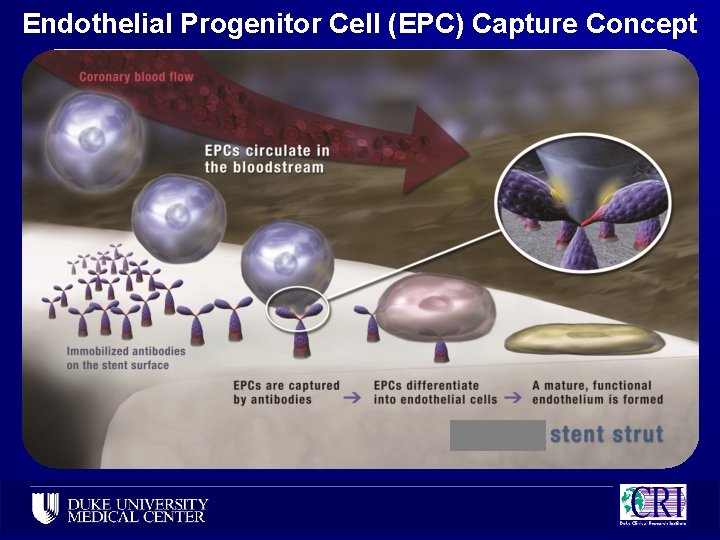
Endothelial Progenitor Cell (EPC) Capture Concept
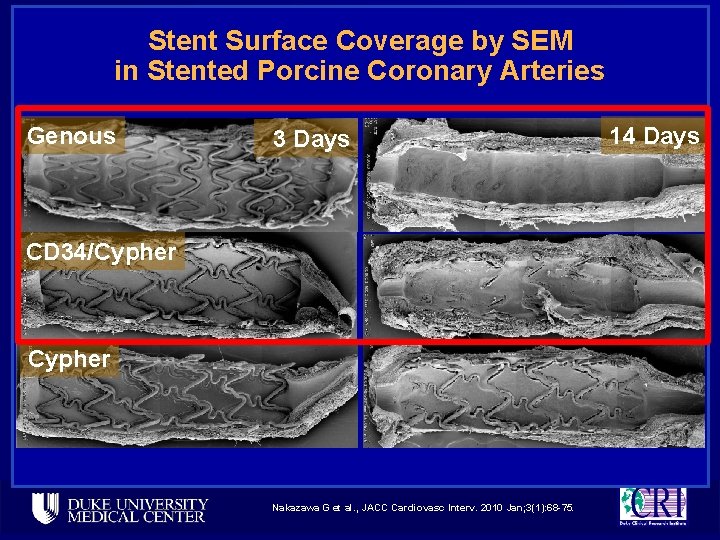
Stent Surface Coverage by SEM in Stented Porcine Coronary Arteries Genous 3 Days CD 34/Cypher Nakazawa G et al. , JACC Cardiovasc Interv. 2010 Jan; 3(1): 68 -75. 14 Days
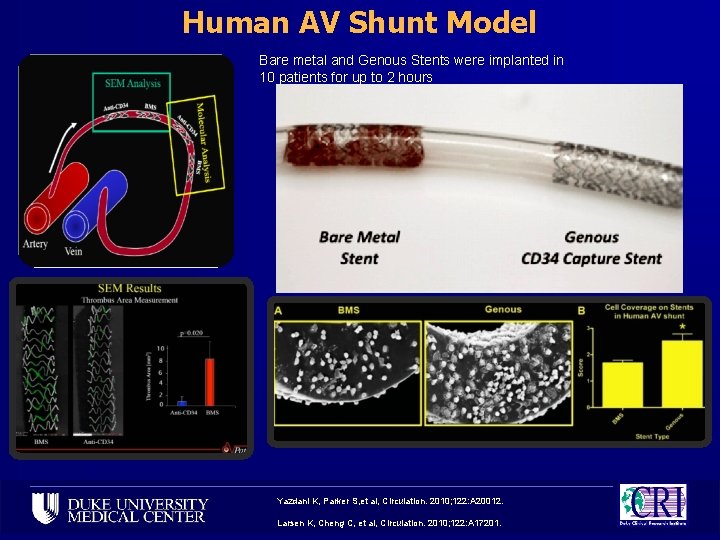
Human AV Shunt Model Bare metal and Genous Stents were implanted in 10 patients for up to 2 hours Yazdani K, Parker S, et al, Circulation. 2010; 122: A 20012. Larsen K, Cheng C, et al, Circulation. 2010; 122: A 17201.
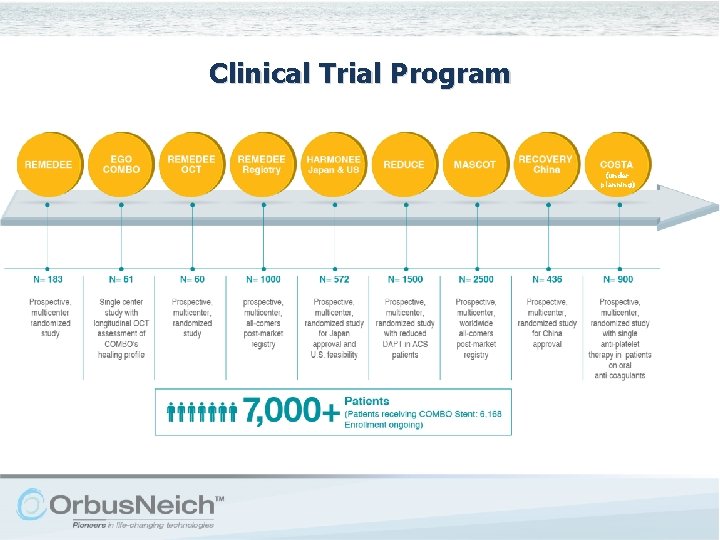
Clinical Trial Program (under planning)
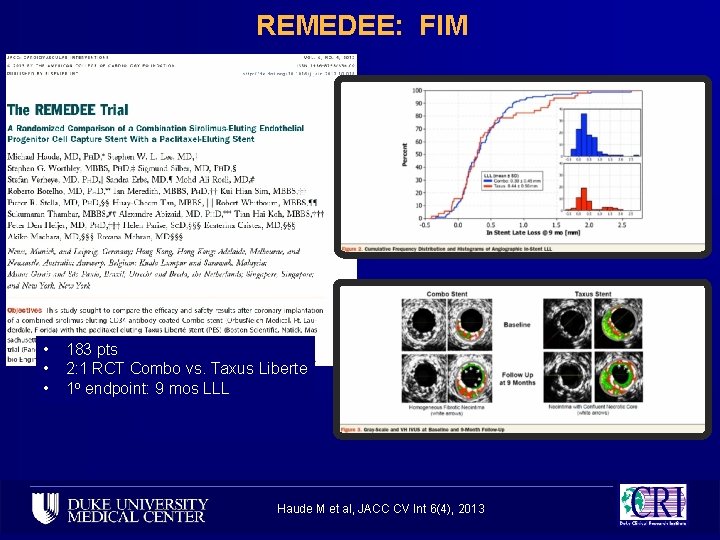
REMEDEE: FIM • • • 183 pts 2: 1 RCT Combo vs. Taxus Liberte 1 o endpoint: 9 mos LLL Haude M et al, JACC CV Int 6(4), 2013

TCT 63: One Year Results of the REMEDEE Registry Clinical Outcomes After Deployment of the Abluminal Sirolimus-Coated Bioengineered Stent (COMBO) in a Multicenter, Prospective Postmarket Registry Robbert J de Winter, MD Ph. D FESC On behalf of the REMEDEE Registry Investigators One Year Results REMEDEE Registry TCT 2015
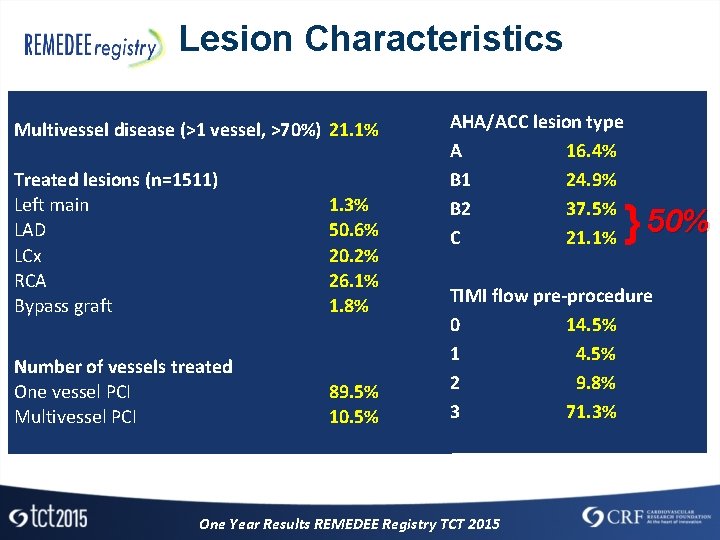
Lesion Characteristics Multivessel disease (>1 vessel, >70%) 21. 1% Treated lesions (n=1511) Left main LAD LCx RCA Bypass graft 1. 3% 50. 6% 20. 2% 26. 1% 1. 8% Number of vessels treated One vessel PCI Multivessel PCI 89. 5% 10. 5% AHA/ACC lesion type A 16. 4% B 1 24. 9% B 2 37. 5% C 21. 1% } 50% TIMI flow pre-procedure 0 14. 5% 1 4. 5% 2 9. 8% 3 71. 3% One Year Results REMEDEE Registry TCT 2015
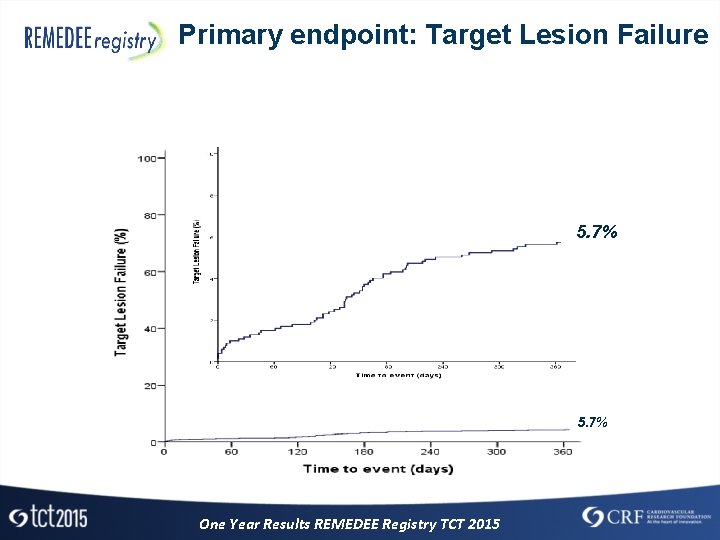
Primary endpoint: Target Lesion Failure 5. 7% One Year Results REMEDEE Registry TCT 2015
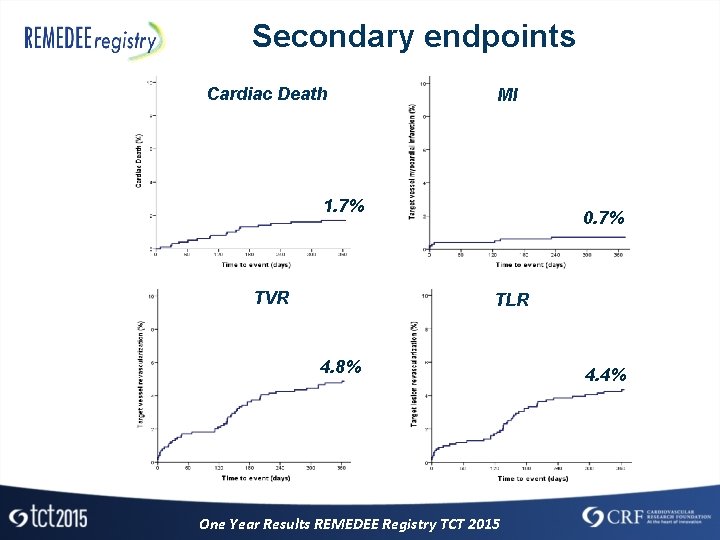
Secondary endpoints Cardiac Death MI 1. 7% TVR 0. 7% TLR 4. 8% One Year Results REMEDEE Registry TCT 2015 4. 4%
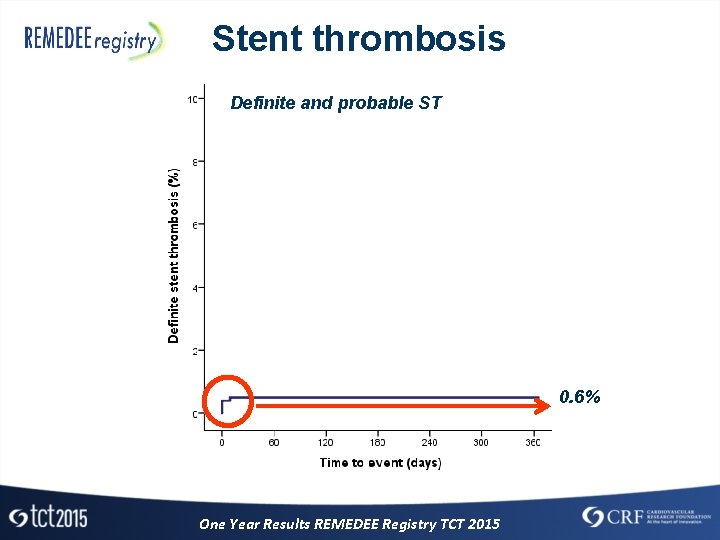
Stent thrombosis Definite and probable ST 0. 6% One Year Results REMEDEE Registry TCT 2015

Pivotal DES registration data: Japan & USA 572 pts 1: 1 RCT vs. EES 1 year clinical & angio follow up • • • Includes MV CAD and non-STEMI ACS FFR guided repeat revascularization OCT subgroups: 6 month & 1 year strut coverage Sites: Japan & USA Trial Structure: Sponsor: Coordinating Center: Site Monitoring: Angio Core Lab: OCT Core Lab: Orbus. Neich DCRI CMIC (Japan) CRF 15
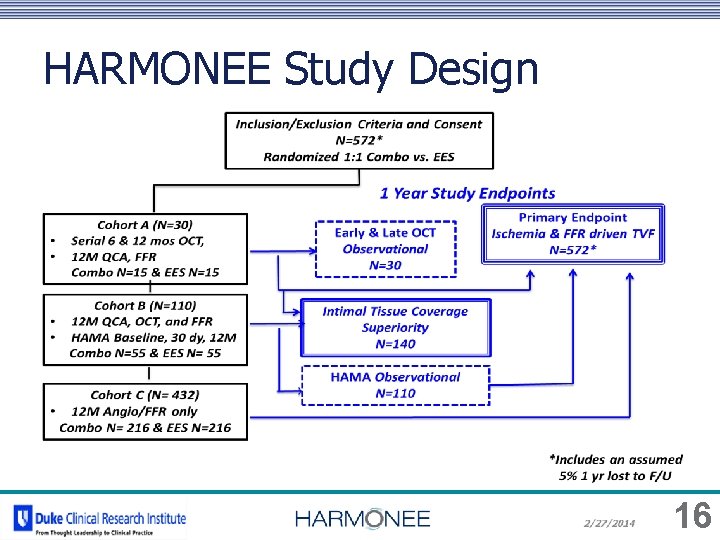
HARMONEE Study Design 16

HBD WG 1 Proof of Concept Global Trial Program Uchida T et al, Circulation Journal 2013
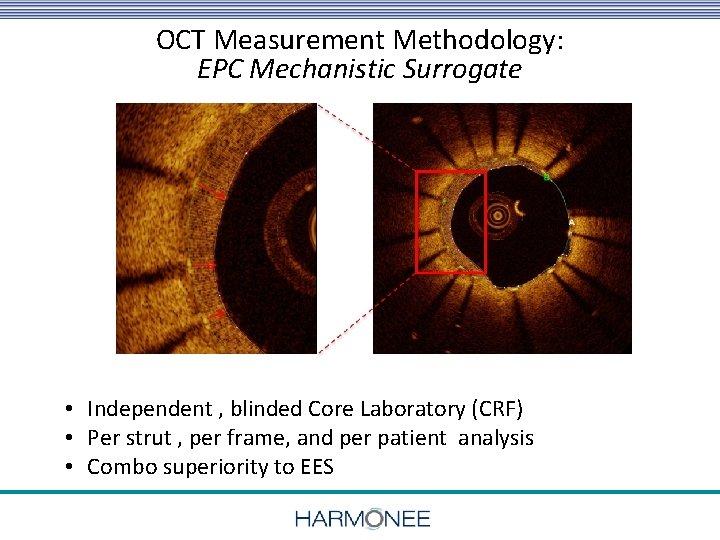
OCT Measurement Methodology: EPC Mechanistic Surrogate • Independent , blinded Core Laboratory (CRF) • Per strut , per frame, and per patient analysis • Combo superiority to EES 18
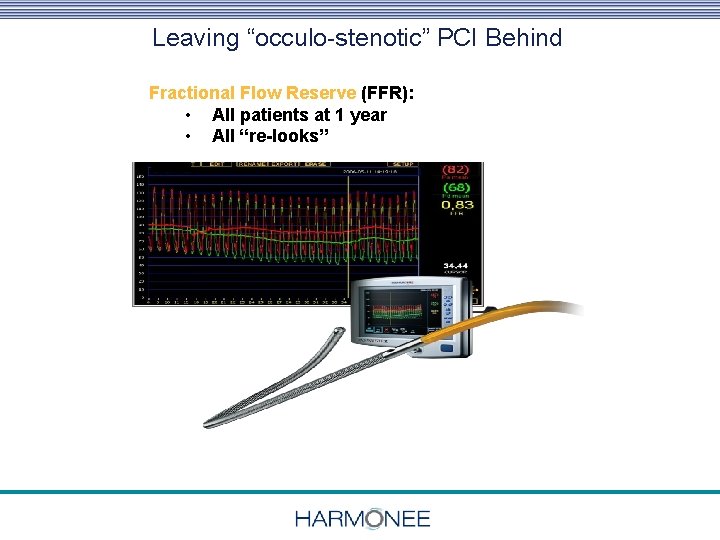
Leaving “occulo-stenotic” PCI Behind Physiologic & High Resolution Anatomic F/U Fractional Flow Reserve (FFR): • All patients at 1 year • All “re-looks” 19

Japan-USA HARMONEE Study • • • First First 4 -component DES biologic EPC capture technology Japan-USA pivotal coronary DES study physiologic & anatomic DES evaluation “non-occulo-stenotic reflex” invasive F/U study Enrolled >500 pts! 20

COMBO Stent: Conclusions Ø DES technology still evolving Ø Acute implant injury affects all stents Ø Very late ST affects all stents (BMS & DES) Ø Thrombotic (ACS) lesions = worse outcomes Ø “Pro-healing” DES design might answer unmet needs for CAD patients

COMBO: A Combination of Sirolimous-Eluting Biodegradable Polymer and Anti-CD 34 Antibody Coated Stent Mitchell W. Krucoff MD, FACC Professor of Medicine / Cardiology Duke University Medical Center Director, Cardiovascular Devices Unit Duke Clinical Research Institute
- Slides: 22