Combined Gas Laws PreAP Chemistry Mr Guerrero Combined

Combined Gas Laws Pre-AP Chemistry, Mr. Guerrero
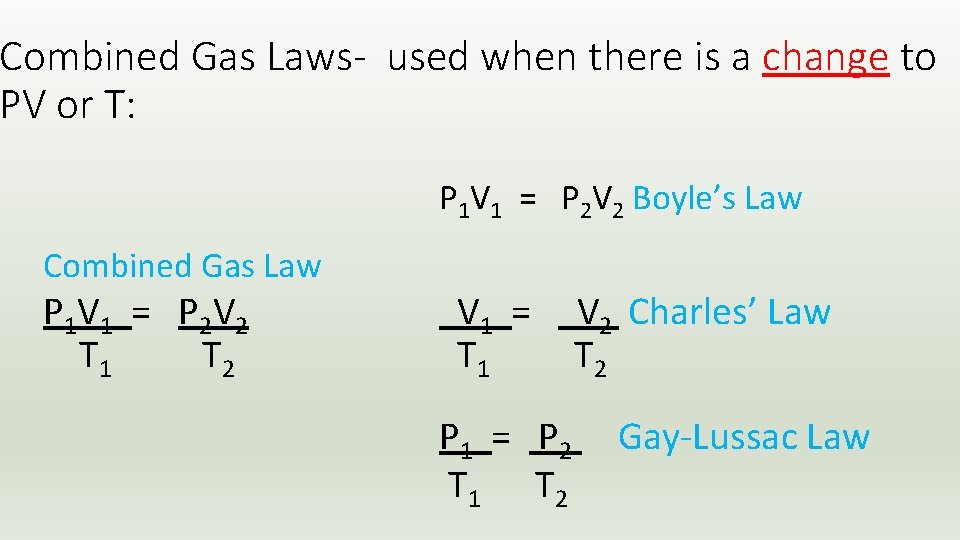
Combined Gas Laws- used when there is a change to PV or T: P 1 V 1 = P 2 V 2 Boyle’s Law Combined Gas Law P 1 V 1 = P 2 V 2 T 1 T 2 V 1 = T 1 P 1 = P 2 T 1 T 2 V 2 Charles’ Law T 2 Gay-Lussac Law

Identify the Gas Law and solve each:

• 355 m. L of Ne gas are at 355 K and exert 2. 35 atm of pressure. What pressure will it exert at 288 K and 368 m. L?

A 1. 25 dm 3 balloon exerts 795 torr. It then expands to 1. 85 dm 3. What is the final pressure? (Assume constant temperature)

• A steel tank of air exerts 395 KPa at 25 o. C. At what temperature does it exert 462 KPa?
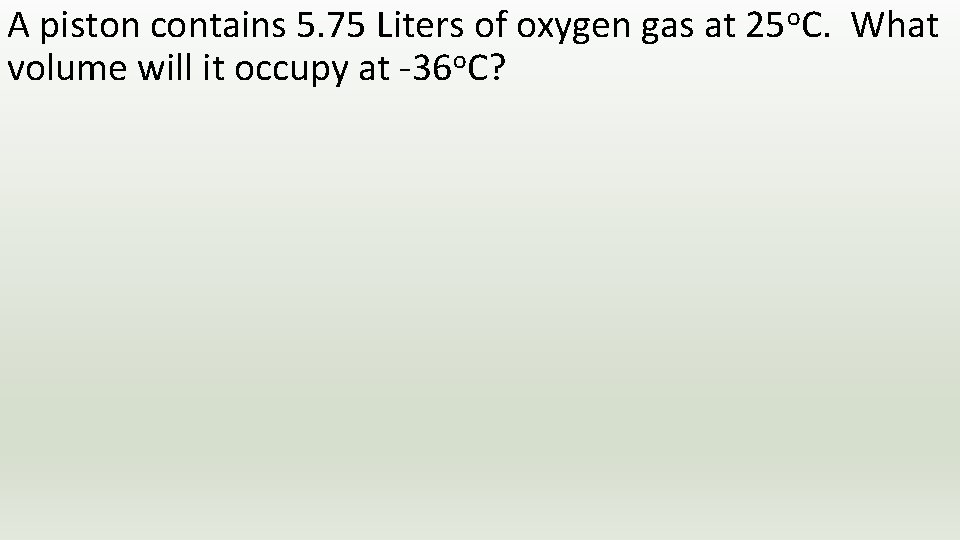
A piston contains 5. 75 Liters of oxygen gas at 25 o. C. What volume will it occupy at -36 o. C?

85. 5 m. L of CO 2 gas are at STP. What pressure will it exert at 100. o. C and compressed to 50. 0 m. L?

Day 2

A sample of He at 38 o. C, is heated to 95 o. C and now occupies 75. 0 m. L. What was the original volume? Identify the gas law:

Dalton’s Law of Partial Pressures-if different gases are mixed together, the total pressure of the mixture equals the sum of the partial pressures. Dalton’s Law: Ptotal = P 1 + P 2 + P 3…. Partial Pressure- the pressure exerted by an individual gas in a mixture, as if it were the only gas in the container.

• A gas mixture the following partial pressures: 1. 00 atm Ar, 3. 00 atm Kr, 2. 00 atm CO 2. • What is the total pressure? • What gas is present in the least and greatest amount? • What is the mole fraction of each?

Wet Gas- A gas collected by bubbling through water. A wet gas is not pure, it contains water vapor, it is a gas mixture. The following picture shows how to collect a wet gas in the lab:

• A sample of O 2 is collected over water. The total pressure of the wet O 2 is 785 torr. What is the pressure of the dry O 2? (VPH 2 O = 24 torr) • What is the mole fraction of the O 2 & H 2 O?

(Review Q. ) How many grams NH 3 are in a wet NH 3 gas(VPH 2 O= 16 torr) 425 m. L at 158 K at 795 torr?

(Review Q. ) Compare the relative rates of diffusion of carbon dioxide and nitrogen gas

- Slides: 17