Combined Gas Law The Combined Gas Law describes

Combined Gas Law • The Combined Gas Law describes the relationship among the pressure, temperature, and volume of an enclosed gas.

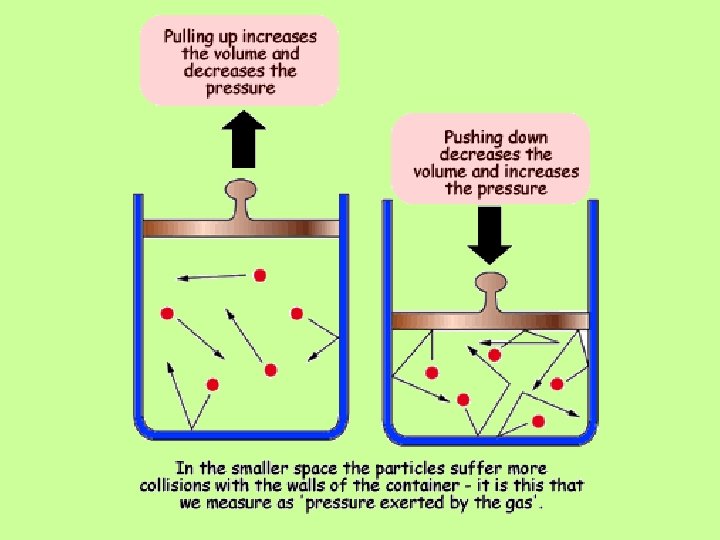
GAS LAWS: Pressure & Volume BOYLE’S LAW: • If the temperature is constant, as the pressure of a gas increases, the volume decreases. (P is inversely proportional to V)

GAS LAWS: Pressure & Temperature Gay-Lussac’s Law • The pressure of a gas is directly proportional to the Kelvin temperature if the volume remains constant.

Gay-Lussac’s Law • As the temperature of an enclosed gas increases, the pressure increases, if the volume is constant.

GAS LAWS: Volume and Temperature Charles’s Law • The volume of a fixed mass of gas is directly proportional to its Kelvin temperature if the pressure is kept constant.

Charles’s Law As the temperature of the water increases (in constant pressure) the volume of the balloon increases.

GAS LAWS: Boyle’s Law Sample Problem: A balloon contains 30. 0 L of helium gas at 103 k. Pa. What is the volume of the helium when the pressure is 25. 0 k. Pa? 103 k. Pa x 30. 0 L = 25. 0 k. Pa x V 2 = (103 k. Pa x 30. 0 L) 25. 0 k. Pa V 2 = 124 L
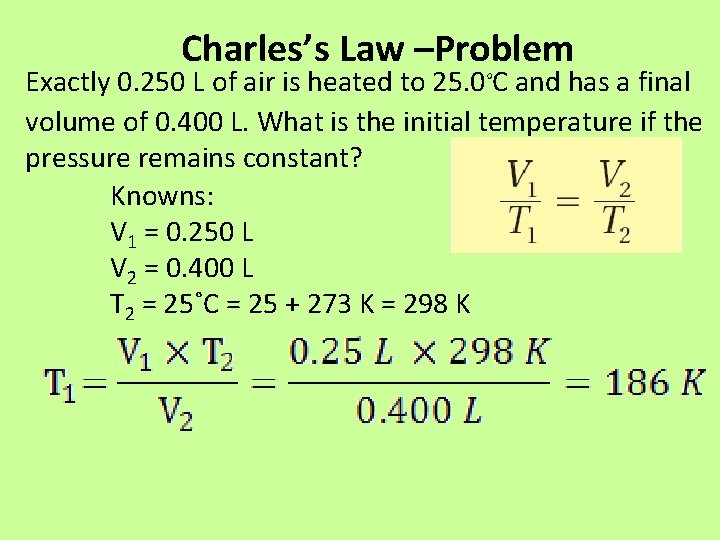
Charles’s Law –Problem Exactly 0. 250 L of air is heated to 25. 0˚C and has a final volume of 0. 400 L. What is the initial temperature if the pressure remains constant? Knowns: V 1 = 0. 250 L V 2 = 0. 400 L T 2 = 25˚C = 25 + 273 K = 298 K

Gay-Lussac’s Law - Problem The pressure in a car tire is 198 k. Pa at 27˚C. After a long drive, the pressure is 225 k. Pa. What is the temperature of the air in the tire (Assume V is constant). Knowns: P 1 = 198 k. Pa T 1 = 27˚C = 27+ 273 K = 300 K P 2 = 225 k. Pa

Combined Gas Law Problem The volume of a gas-filled balloon is 30. 0 L at 313 K and 153 k. Pa pressure. What would the volume be at standard temperature and pressure (STP)?
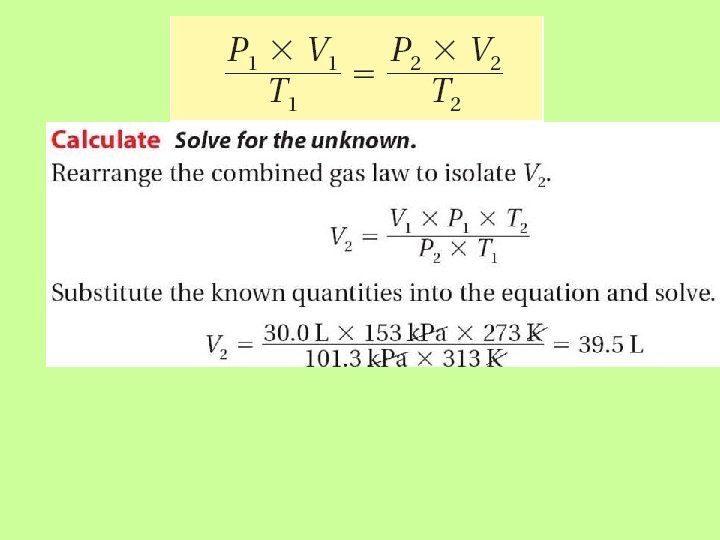

Combined Gas Law - Problem A 5. 00 L air sample has a pressure of 107 k. Pa at a temperature of -50. 0˚C. If the temperature is raised to 102˚C and the volume expands to 7. 00 L, what will the new pressure be? Knowns: V 1 = 5. 00 L P 1 = 107 k. Pa T 1 = -50˚C = -50+ 273 K = 223 K T 2 = 102˚C = 102+ 273 K = 375 K V 2 = 7. 00 L

Combined Gas Law - Problem

Charles’s Law –Problem A gas sample at 40. 0˚C occupies a volume of 2. 32 L. If the temperature is raised to 75. 0˚C, what will the volume be, assuming the pressure remains constant? Knowns: V 1 = 2. 32 L T 1 = 40˚C + 273 K = 313 K T 2 = 75˚C + 273 K = 348 K

Gay-Lussac’s Law - Problem A gas in a sealed container has a pressure of 125 k. Pa at a temperature of 303 K. If the pressure in the container is increased to 1. 98 atm, what is the new temperature? Knowns: P 1 = 125 k. Pa T 1 = 303 K P 2 = 1. 98 atm x (101. 3 k. Pa/1 atm) = 200. 6 k. Pa 486 K

Combined Gas Law - Problem A gas at 110 k. Pa and 303 K fills a flexible container with an initial volume of 2. 00 L. If the temperature is raised to 353 K and the pressure increased to 440 k. Pa, what is the new volume? Knowns: V 1 = 2. 00 L P 1 = 110 k. Pa T 1 = 303 K T 2 = 353 K P 2 = 440 k. Pa
- Slides: 17