Combinatorial Chemistry and Drug Discovery Lab Jasmine Erfe

Combinatorial Chemistry and Drug Discovery Lab Jasmine Erfe Miramar College/Lab Technician Ericka Senegar-Mitchell Science in the City/ Director; Junipero Serra High School/Science Educator Sandra Slivka Southern CA Biotechnology Center @ Miramar College/Director

Background This lab protocol was adapted from the original work of Scott Wolkenberg and Andrew Su of The Scripps Research Institute in La Jolla, California. The experiment was originally published in the June 2001 issue of the Journal of Chemical Education and implemented in the San Diego area from May 2002 thru 2005. CITATION: Wolkenberg, Scott E. ; Su, Andrew I. J Chem. Educ. 2001 78 784

Overview • In this lab students will identify a drug that kills bacteria by producing libraries of compounds based on the A-B model. • They will test the mixtures for antibiotic activity and then isolate the individual compound(s) which possess antibiotic properties. • Students will screen the mixtures by utilizing techniques used to conduct Kirby-Bauer and Ouchterlony tests.

California Science Content Standards Subject Area Content Standard Chemistry 2. a. Chemical Bonds – formation of ionic and covalent (peptide) bonds 3. g. Stoichiometry – redox reactions, dehydration synthesis (condensation) 10. b. Organic Chemistry – bonding characteristics of carbon 10. e. Functional Groups – formation of a hydrazone from an aldehyde and a hydrazine, identification and analysis of amine groups Biology Genetics (Molecular Biology) Genetics (Biotechnology) Evolution Physiology 1. a. Membrane Regulation – membrane structure and function 1. c. Prokaryotic and Eukaryotic Cells – structure and function 1. h. Macromolecules – structure and function 4. c. Mutations – antibiotic expression 4. e. Proteins – structure 4. f. Proteins – function/chemical properties 5. c. Biotechnology – production of novel biomedical and agricultural products 7. a. Natural Selection – phenotype vs. genotype 7. d. Genetic Variation – influence of environmental factors on the natural selection of adaptive traits 8. a. Natural Selection – selective fitness; differential survival of groups 10. b. Immune Response – antibody/antigen response 10. d. Bacterial Infections – use of antibiotics in treating bacterial infections; use of antibacterial agents to control the growth of bacteria

National Science Education Standards Category Content Standard Unifying Concepts and Processes Change, constancy and measurement – process of generating chemical libraries and screening for effective compounds, combinatorial data Evidence, models, and explanation – Kirby-Bauer & Ouchterlony Tests Form and function – cells, macromolecules, prokaryotes (E. coli ) Science as Inquiry Abilities necessary to do scientific inquiry – production of mixtures (libraries) of compounds using the A-B model; deconvolution (separation) of the mixtures to identify the compound(s) with antibiotic properties Life Science and Technology Science in Personal and Social Perspectives History and Nature of Science The Cell – types of cells, cell membrane structure and function Biological evolution – development of antibiotic resistance Behavior of organisms – growing bacterial cultures (lag vs. log phase), interpretation of bacterial plates (deconvolution) Understandings about science and technology – process and design in combinatorial chemistry; bringing a new pharmaceutical to market Personal and community health – drug discovery, pharmacology, diagnosis of human disease and course of treatment Natural and human-induced hazards – aseptic (sterile) technique, safety protocols including Material Safety Data Sheets (MSDS) Science as a human endeavor – biomedical research, clinical trials, bioethics of the biotechnology industry

Chemicals

Chemicals CHEMICAL VENDOR CATALOG # PRICE* A 1: 2 -nitrobenzaldehyde Sigma-Aldrich 772780 -50 g $116. 50 A 2: 5 -nitro-2 -furaldehyde Sigma-Aldrich 170968 -25 g $102. 50 A 3: 3 -nitrobenzaldehyde Sigma-Aldrich N 10845 -100 g B 1: 4 -bromophenylhydrazine hydrochloride Sigma-Aldrich 143219 -10 g $101. 50 B 2: 4 -cyanophenylhydrazine hydrochloride Sigma-Aldrich 453471 -5 g $42. 20 B 3: aminoguanidine bicarbonate Sigma-Aldrich 109266 -100 g $15. 00 $17. 80 Total: $395. 50

Suggested Materials

Presenter Notes Preparing Overnight Culture of E. coli 1. Transfer 10 ml of sterile LB broth in a culture tube. 2. To obtain a sample of E. coli, take the inoculating loop and dip into the frozen E. coli stock. Be certain that some of the stock has adhered to the loop. 3. Dip the inoculating loop into the culture tube containing the 10 ml of LB broth. Stir the broth using the loop to ensure that the E. coli is thoroughly mixed in the broth. 4. Cap the tube and incubate the E. coli culture overnight at 37°C with agitation.

Presenter Notes Growing E. Coli into Log Phase 1. 2. 3. Transfer 25 ml of sterile LB broth in a 50 ml Erlenmeyer flask. Take 1 ml of the prepared overnight E. coli culture and transfer the sample into the Erlenmeyer flask containing 25 ml of LB broth. Cap the Erlenmeyer flask and incubate at 37°C with agitation.

Presenter Notes Spectrophotometer 1. Turn on the spectrophotometer and set to O. D. 550. 2. Take a 3 ml sample of the E. coli culture and transfer into a test tube. Wipe and handle the test tube with Kimwipes and place into the spectrophotometer. 3. Note the reading. The culture will reach log phase once the O. D. 550 reaches 0. 3 -0. 4. 4. If the reading has not reached log phase, repeat steps 2 and 3 using a clean test tube and waiting 30 minutes between each new reading. 5. Once the culture has reached log phase, note the total volume of culture that remains in the flask. According to the volume, add glycerol in the amount of 15% of the total volume to the flask and immediately transfer 1 ml aliquots of the log phase culture into cryogenic tubes and freeze at -80ºC.

Instructor Notes Prepare stock solutions You have been provided 6 uniquely labeled conical tubes, each containing a specific chemical. Add 12 m. L of deionized water to each tube and shake vigorously for approximately ten seconds.

Instructor Notes If mixtures are not completely dissolving using a hot water bath may be effective.

Combinatorial Chemistry and Drug Discovery Lab Student Protocol

Lab Safety Tips for handling E. coli: 1. Wipe down the lab bench or station with a 10% bleach solution or 70% isopropanol solution at the beginning and end of each laboratory session. 2. When creating mixtures and transferring chemical solutions or liquid bacterial cultures, keep nose and mouth away from the opening of the tube to avoid inhaling any aerosols that may be created. 3. All spills should be reported to your instructor and cleaned up immediately according to the Material Safety Data Sheets (MSDS) for each chemical used in the experiment. Be sure to wear proper footwear (closed toe) to prevent injury. 4. Dispose of any materials that have come in contact with bacterial cultures (i. e. tubes, pipettes) in special waste containers as provided by your instructor. 5. Wash hands with soap and water before leaving the lab.

Overview • This lab protocol was adapted from the original work of Scott Wolkenberg and Andrew Su of The Scripps Research Institute in La Jolla, CA. The experiment was originally published in the June 2001 issue of the Journal of Chemical Education. • Combinatorial Chemistry is a technique used to synthesize a library of compounds and screen for a desired property. Instead of screening one compound at a time, the compounds are screened more efficiently in mixtures. CITATION: Wolkenberg, Scott E. ; Su, Andrew I. J Chem. Educ. 2001 78 784
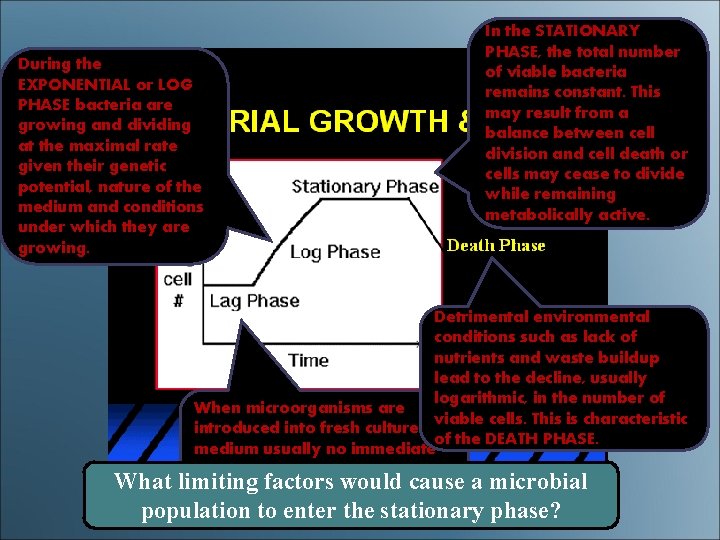
During the EXPONENTIAL or LOG PHASE bacteria are growing and dividing at the maximal rate given their genetic potential, nature of the medium and conditions under which they are growing. In the STATIONARY PHASE, the total number of viable bacteria remains constant. This may result from a balance between cell division and cell death or cells may cease to divide while remaining metabolically active. Detrimental environmental conditions such as lack of nutrients and waste buildup lead to the decline, usually logarithmic, in the number of When microorganisms are viable cells. This is characteristic introduced into fresh culture of the DEATH PHASE. medium usually no immediate increase in cell number occurs What limiting factors would cause a microbial therefore this is referred to as the LAG PHASE. population to enter the stationary phase?

Kirby-Bauer Test • Disk-diffusion method used for routine testing in a clinical laboratory in which an isolated microbe is tested for susceptibility to numerous antibiotics. • The isolated organism is uniformly placed on an agar plate with paper disk of fixed concentrations of antibiotics. • Growth of the organism and diffusion of the antibiotic occur simultaneously resulting in a circular zone of inhibition if the antibiotic has antibacterial properties. Measuring Antibiotic Resistance

Ouchterlony Test • A double diffusion technique developed by Organ Ouchterlony more than 40 years ago. • A technique in which reaction partners, antigen and antibody, are allowed to diffuse to each other in an agar gel in a precipitation reaction. • Classical procedure used to detect the presence of antibodies and determine their specificity by visualization of "lines of identity" or precipitin lines.

Student/Group Lab Set-up q 3 Luria Broth (LB) agar plates q 6, 15 m. L conical tubes containing stock solutions: • A 1: 2 -nitrobenzaldehyde • A 2: 5 -nitro-2 -furaldehyde • A 3: 3 -nitrobenzaldehyde • B 1: 4 -bromophenylhydrazine hydrochloride • B 2: 4 -cyanophenylhydrazine hydrochloride • B 3: aminoguanidine bicarbonate q 1 cryotube (orange cap) containing 1. 0 m. L E. coli q 1 cell spreader q 15 disposable transfer pipettes or P-1000 micropipette q 9 eppendorf tubes q 1 plastic straw, wrapped q 1 sterile wrapped transfer pipette q. Conical tube rack q. Sharpie Marker

Label transfer pipettes 1. Label 6 of the transfer pipettes A 1, A 2, A 3, B 1, B 2, B 3. These will be used to prepare your compounds. 2. Label 9 transfer pipettes M 1, M 2, M 3, M 4, M 5, M 6, A#B 1, A#B 2, A#B 3. These will be used to transfer your compounds onto the plates. Each student or group will be assigned mixtures to test for confirmation of antibiotic activity (i. e. # = 1, 2, or 3 for A 1 B 1, A 1 B 2, A 1 B 3; A 2 B 2… ).

Label epitubes

Label Petri dishes M 1 M 2 M 3 M 4 M 5 A#B 1 A#B 2 M 6 A#B 3 Each student or group will be assigned mixtures to test for confirmation of antibiotic activity. (i. e. # = 1, 2, or 3 for A 1 B 1, A 1 B 2, A 1 B 3; A 2 B 2. . . ). M 1 M 2 M 3 M 4 M 5 A#B 1 A#B 2 M 6 A#B 3 Create wells in plates

Carefully invert the tube containing 1. 0 ml of thawed E. coli several times before opening. Spread the E. coli culture evenly on the surface of each agar plate.
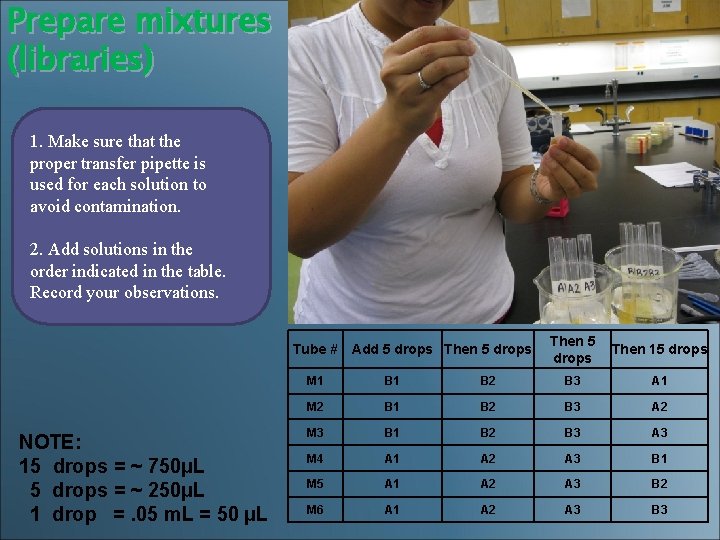
Prepare mixtures (libraries) 1. Make sure that the proper transfer pipette is used for each solution to avoid contamination. 2. Add solutions in the order indicated in the table. Record your observations. Tube # Add 5 drops Then 5 drops NOTE: 15 drops = ~ 750µL 5 drops = ~ 250µL 1 drop =. 05 m. L = 50 µL Then 5 Then 15 drops M 1 B 2 B 3 A 1 M 2 B 1 B 2 B 3 A 2 M 3 B 1 B 2 B 3 A 3 M 4 A 1 A 2 A 3 B 1 M 5 A 1 A 2 A 3 B 2 M 6 A 1 A 2 A 3 B 3

Add compounds to wells and wait approximately 15 -20 minutes for liquid to absorb.

Incubate at 37ºC or store at room temperature overnight. (48 hours is optimal for room temperature option) 1. If no incubator is available: Plates can be stored overnight at room temp. with the agar side down. 2. If incubator is available: Allow compounds to absorb into agar then incubate agar side up.
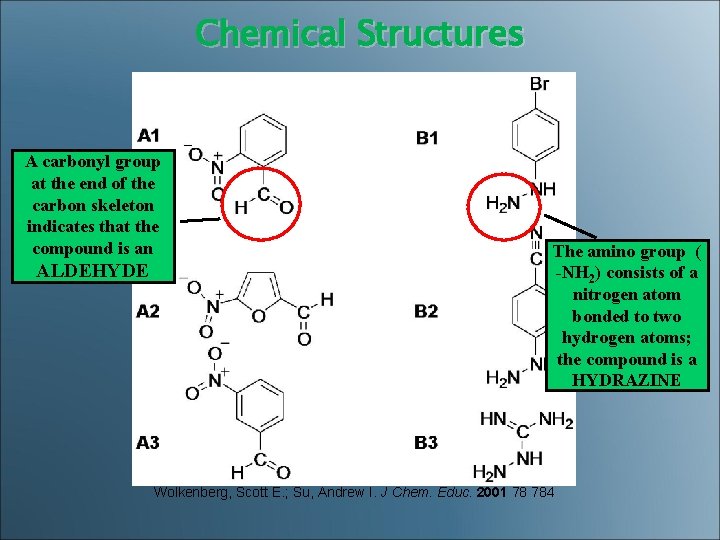
Chemical Structures A carbonyl group at the end of the carbon skeleton indicates that the compound is an ALDEHYDE The amino group ( -NH 2) consists of a nitrogen atom bonded to two hydrogen atoms; the compound is a HYDRAZINE Wolkenberg, Scott E. ; Su, Andrew I. J Chem. Educ. 2001 78 784
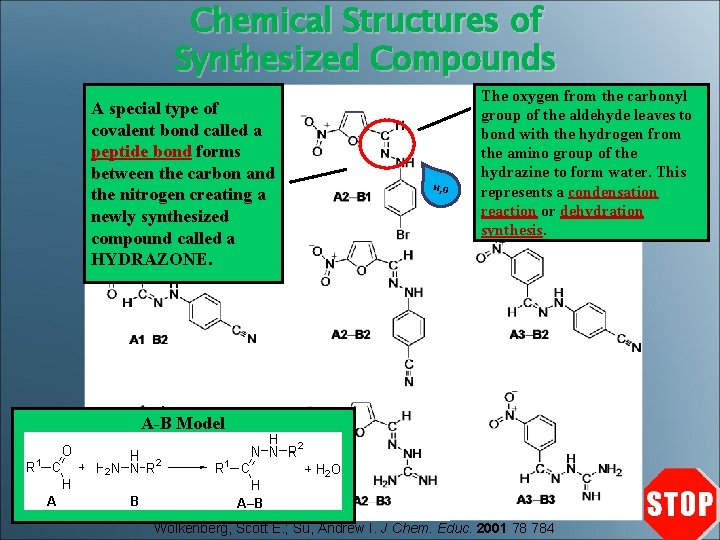
Chemical Structures of Synthesized Compounds A special type of covalent bond called a peptide bond forms between the carbon and the nitrogen creating a newly synthesized compound called a HYDRAZONE. H 2 O The oxygen from the carbonyl group of the aldehyde leaves to bond with the hydrogen from the amino group of the hydrazine to form water. This represents a condensation reaction or dehydration synthesis. A-B Model Wolkenberg, Scott E. ; Su, Andrew I. J Chem. Educ. 2001 78 784

Combinatorial Chemistry and Drug Discovery Lab Data Analysis/ Results
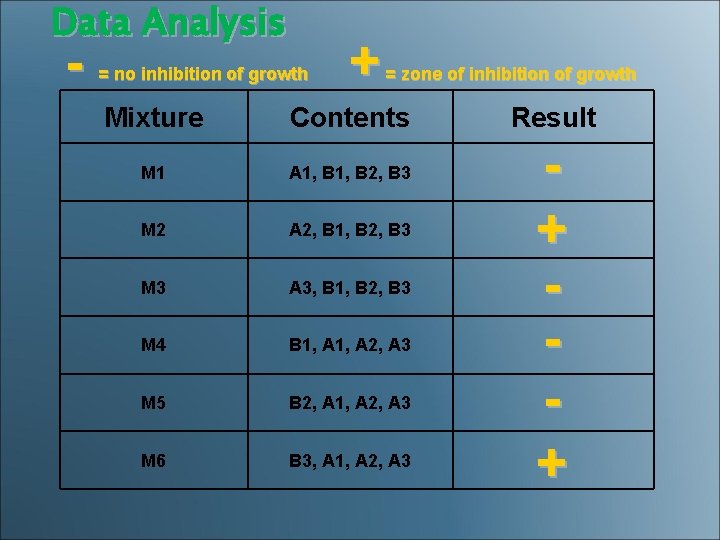
Data Analysis - + = no inhibition of growth = zone of inhibition of growth Mixture Contents M 1 A 1, B 2, B 3 M 2 A 2, B 1, B 2, B 3 M 3 A 3, B 1, B 2, B 3 M 4 B 1, A 2, A 3 M 5 B 2, A 1, A 2, A 3 M 6 B 3, A 1, A 2, A 3 Result + +
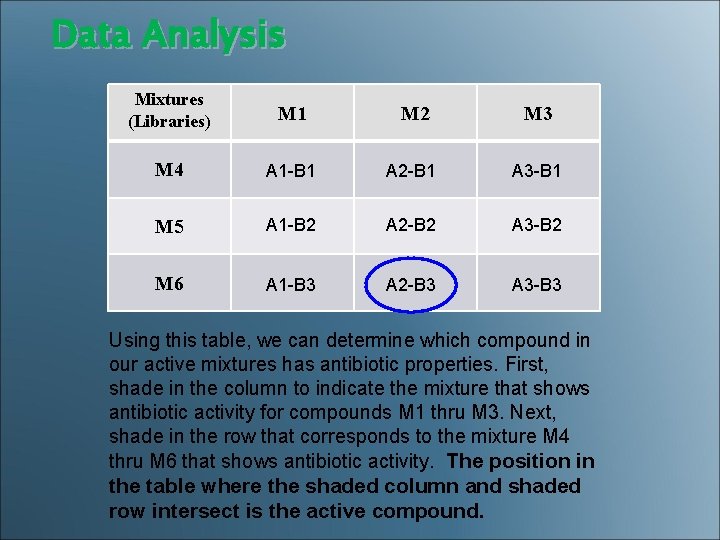
Data Analysis Mixtures (Libraries) M 1 M 2 M 3 M 4 A 1 -B 1 A 2 -B 1 A 3 -B 1 M 5 A 1 -B 2 A 2 -B 2 A 3 -B 2 M 6 A 1 -B 3 A 2 -B 3 A 3 -B 3 Using this table, we can determine which compound in our active mixtures has antibiotic properties. First, shade in the column to indicate the mixture that shows antibiotic activity for compounds M 1 thru M 3. Next, shade in the row that corresponds to the mixture M 4 thru M 6 that shows antibiotic activity. The position in the table where the shaded column and shaded row intersect is the active compound.

Extension: Kirby–Bauer Test Determining Class Average Zone Size 1. After 24 hours in the incubator or 48 hours at room temperature check for the presence of antibiotic activity. This is done by looking for a clear area, called a zone of inhibition, surrounding a well. Remember to never open the Petri dish for a better view. Use the agar side to observe and measure any zone of inhibition. 2. Using a ruler, measure the diameter (in millimeters) of any zone of inhibition and record your individual group data on the data table provided. 3. Now gather class data to determine the average zones of inhibition, if present, for each of the different compounds M 1 thru M 6 and record the averages. 4. After recording the average class data for the diameters of the zones of inhibition, decide whether your groups sample of E. coli bacteria is susceptible (sensitive) displaying a CLEAR zone of inhibition, unaffected (resistant) showing no observable zone of inhibition or intermediate (somewhere in between) for each of the compounds M 1 thru M 6. Record your conclusion in the final column of the data table.

Extension: Kirby–Bauer Test Determining Class Average Zone Size Using a ruler, measure the diameter (in millimeters) of any zone of inhibition and record your individual group data on the data table provided.
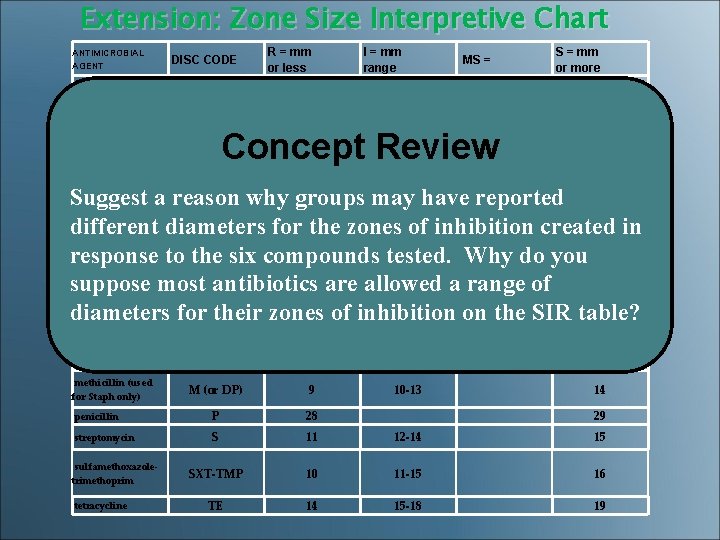
Extension: Zone Size Interpretive Chart ANTIMICROBIAL AGENT DISC CODE R = mm or less I = mm range S = mm or more MS = amoxicillin (Staph) AMC 19 20 amoxicillin (other bacteria) AMC 13 14 -17 18 ampicillin (Staph) AM 28 29 ampicillin (other bacteria) AM 11 12 -13 14 E 13 14 -22 23 GM 12 13 -14 15 M (or DP) 9 10 -13 14 penicillin P 28 29 streptomycin S 11 12 -14 15 SXT-TMP 10 11 -15 16 TE 14 15 -18 19 Concept Review Suggest a reason why groups may have reported carbenicillin CB 13 14 -16 17 (Pseudomonas) different diameters for the zones of inhibition created in carbenicillin (other CB 17 18 -22 23 bacteria) response to the six compounds tested. Why do you cefoxatime CTX 14 15 -22 23 suppose most antibiotics are allowed a range of cephalothin CF 14 15 -17 18 diameters for their zones of inhibition on the SIR table? chloramphenicol C 12 13 -17 18 erythromycin gentamycin methicillin (used for Staph only) sulfamethoxazoletrimethoprim tetracycline

Expected Results at 37ºC

Results at Room Temperature
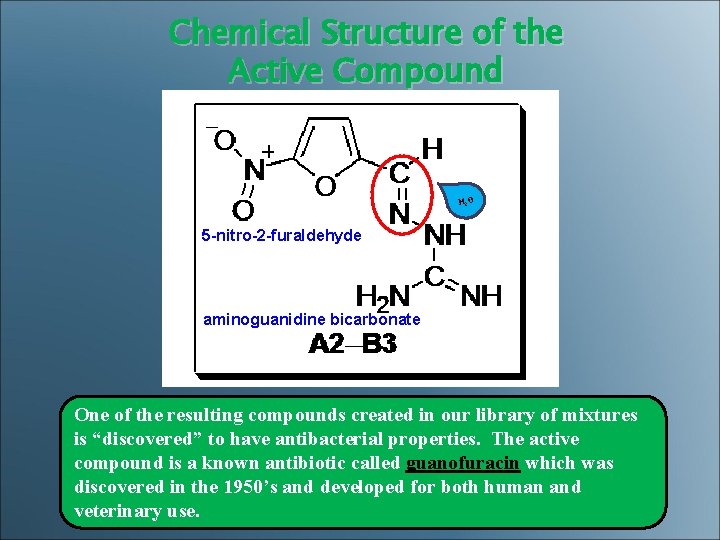
Chemical Structure of the Active Compound H 2 O 5 -nitro-2 -furaldehyde aminoguanidine bicarbonate One of the resulting compounds created in our library of mixtures is “discovered” to have antibacterial properties. The active compound is a known antibiotic called guanofuracin which was discovered in the 1950’s and developed for both human and veterinary use.

Combinatorial Chemistry and Drug Discovery Lab Post-lab Discussion

Autoclaving is the most effective and most efficient means of sterilization. All autoclaves operate on a time/ temperature relationship. The usual standard temperature/ pressure employed is 121ºC/15 psi for 15 minutes. Antibacterial Agents

Three Major Classes of Antibacterial Agents Controlling the growth of microorganims usually involves the use of physical or chemical agents which either kill or prevent the growth of microorganisms. 1) Bactericidal – agents that kill bacteria are called cidal agents; also referred to as bactericides. 2) Bacteriostatic – agents which inhibit the growth of cells (without killing them) are referred to as static agents. 3) Bacteriolytic – agents that have the ability to lyse or break apart, dissolve, and destroy bacteria by the use of an enzyme or other agent. Bacteria have the ability to develop resistance following repeated or subclinical (insufficient) doses, so more advanced antibiotics and synthetic antimicrobials are continually required to overcome them.
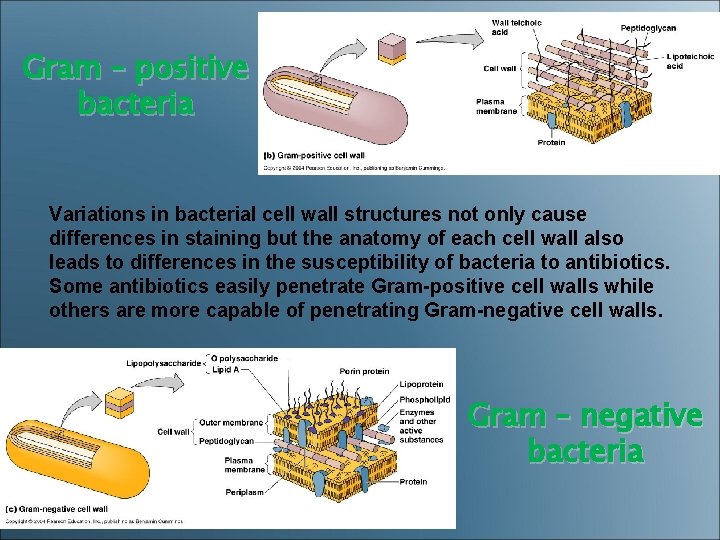
Gram – positive bacteria Variations in bacterial cell wall structures not only cause differences in staining but the anatomy of each cell wall also leads to differences in the susceptibility of bacteria to antibiotics. Some antibiotics easily penetrate Gram-positive cell walls while others are more capable of penetrating Gram-negative cell walls. Gram – negative bacteria
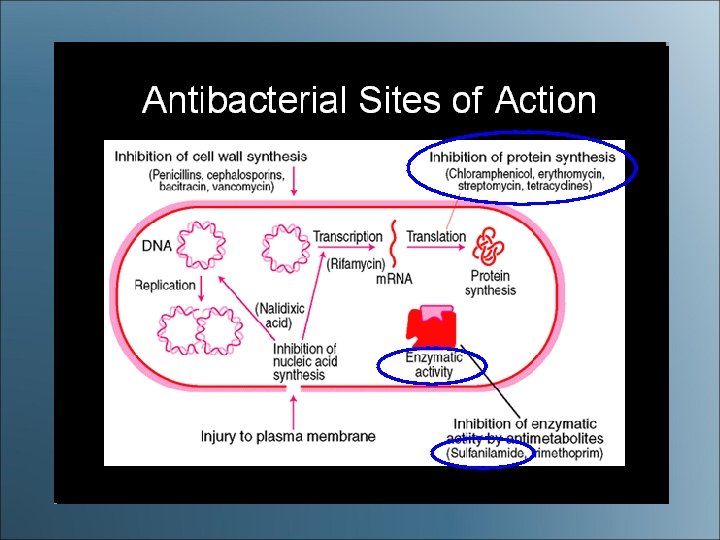
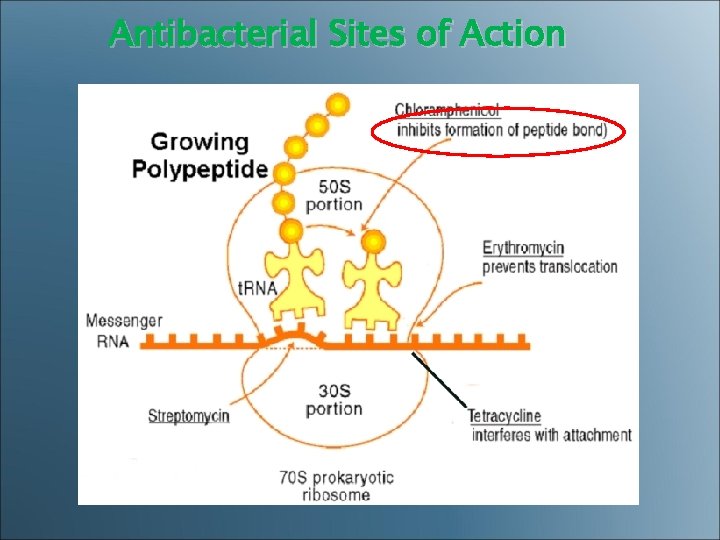
Antibacterial Sites of Action

Concept Review • Analyze the graph by describing the phase of growth being represented by lines A thru E: LAG, LOG, STATIONARY, or DEATH. • If the graph shown is the result of antibiotic resistance testing, what is the effect of the antibiotics on the growth of the bacteria at A thru E: NORMAL, BACTERIOSTATIC, RESISTANT or BACTERICIDAL?

Great Job Jasmine Erfe!
- Slides: 46