COMBINATION OF BLOODBASED BIOMARKERS AND NEUROPSYCHOLOGICAL ASSESSMENT ENABLES

COMBINATION OF BLOOD-BASED BIOMARKERS AND NEUROPSYCHOLOGICAL ASSESSMENT ENABLES RELIABLE CLASSIFICATION OF TESTED SUBJECTS BY CONTROLS, MILD COGNITIVE IMPAIRMENT AND ALZHEIMER'S DISEASE Martin Kleinschmidt © Fraunhofer
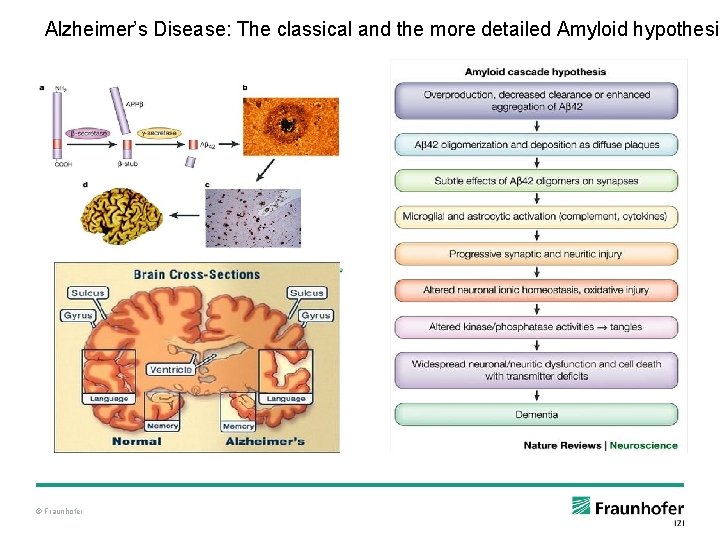
Alzheimer’s Disease: The classical and the more detailed Amyloid hypothesis © Fraunhofer
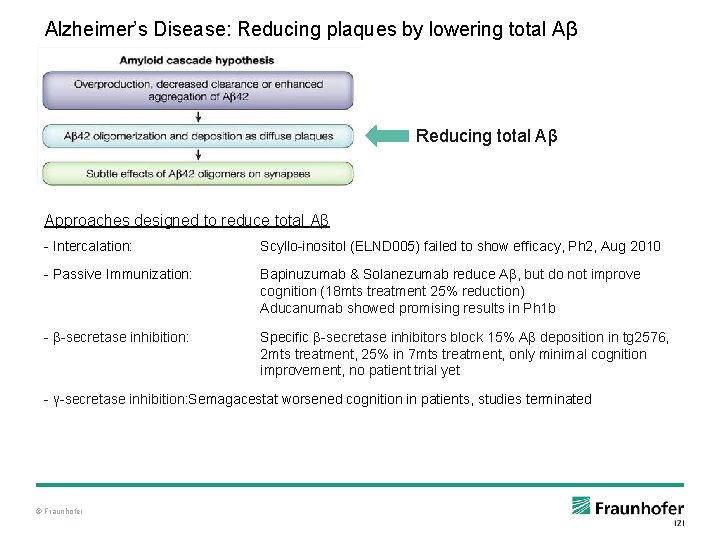
Alzheimer’s Disease: Reducing plaques by lowering total Aβ Reducing total Aβ Approaches designed to reduce total Aβ - Intercalation: Scyllo-inositol (ELND 005) failed to show efficacy, Ph 2, Aug 2010 - Passive Immunization: Bapinuzumab & Solanezumab reduce Aβ, but do not improve cognition (18 mts treatment 25% reduction) Aducanumab showed promising results in Ph 1 b - β-secretase inhibition: Specific β-secretase inhibitors block 15% Aβ deposition in tg 2576, 2 mts treatment, 25% in 7 mts treatment, only minimal cognition improvement, no patient trial yet - γ-secretase inhibition: Semagacestat worsened cognition in patients, studies terminated © Fraunhofer
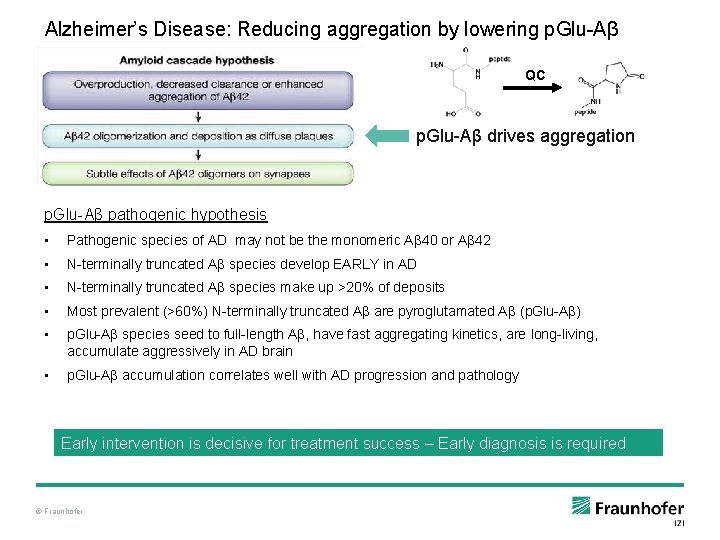
Alzheimer’s Disease: Reducing aggregation by lowering p. Glu-Aβ QC p. Glu-Aβ drives aggregation p. Glu-Aβ pathogenic hypothesis • Pathogenic species of AD may not be the monomeric Aβ 40 or Aβ 42 • N-terminally truncated Aβ species develop EARLY in AD • N-terminally truncated Aβ species make up >20% of deposits • Most prevalent (>60%) N-terminally truncated Aβ are pyroglutamated Aβ (p. Glu-Aβ) • p. Glu-Aβ species seed to full-length Aβ, have fast aggregating kinetics, are long-living, accumulate aggressively in AD brain • p. Glu-Aβ accumulation correlates well with AD progression and pathology Early intervention is decisive for treatment success – Early diagnosis is required © Fraunhofer

Alzheimer’s Disease: Strategy for suitable biomarkers MRI and PET pros: established methods, state of the art cons: resource intensive, not suitable for phase I, good in phase II / III CSF biomarkers (e. g. Aβ 42/Aβ 40, Tau, P-Tau) pros: reliable biomarker, state of the art, commercially available diagnostic tools cons: invasive, most volunteers decline LP, not suitable for area-wide screening Plasma (e. g. Aβ 42/Aβ 40, Tau, P-Tau) pros: blood withdrawal is generally accepted, suitable for initial screening cons: currently no reliable biomarker, 10 -times lower Aβ conc. than in CSF, but 100 -times higher overall protein content Development of plasma based biomarker assay systems © Fraunhofer
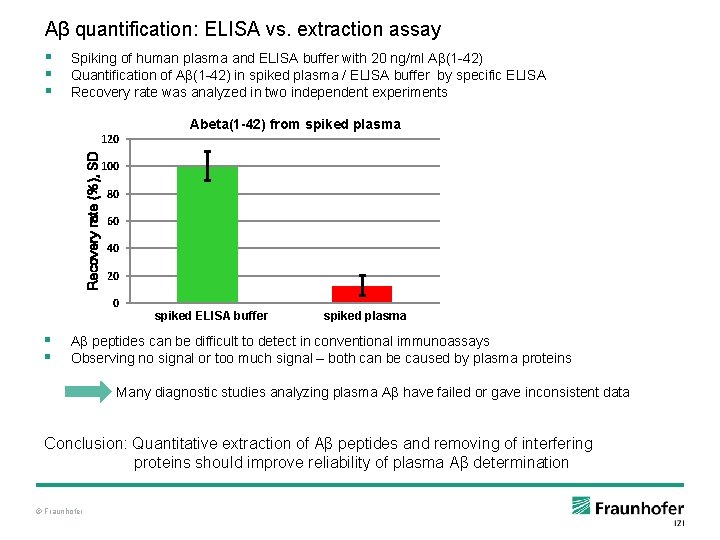
Aβ quantification: ELISA vs. extraction assay § § § Spiking of human plasma and ELISA buffer with 20 ng/ml Aβ(1 -42) Quantification of Aβ(1 -42) in spiked plasma / ELISA buffer by specific ELISA Recovery rate was analyzed in two independent experiments Recovery rate (%), SD 120 100 80 60 40 20 0 § § Abeta(1 -42) from spiked plasma spiked ELISA internal STDbuffer spiked plasma direct ELISA Aβ peptides can be difficult to detect in conventional immunoassays Observing no signal or too much signal – both can be caused by plasma proteins Many diagnostic studies analyzing plasma Aβ have failed or gave inconsistent data Conclusion: Quantitative extraction of Aβ peptides and removing of interfering proteins should improve reliability of plasma Aβ determination © Fraunhofer
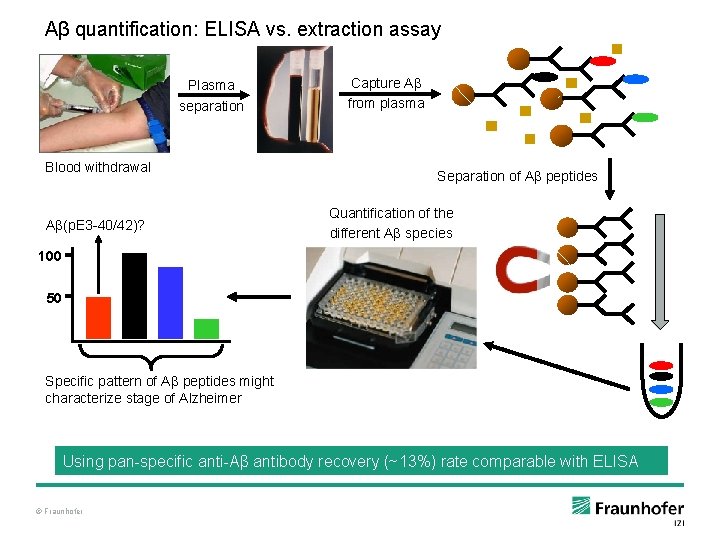
Aβ quantification: ELISA vs. extraction assay Plasma separation Blood withdrawal Aβ(p. E 3 -40/42)? Capture Aβ from plasma Separation of A peptides Quantification of the different Aβ species 100 50 Specific pattern of Aβ peptides might characterize stage of Alzheimer Using pan-specific anti-Aβ antibody recovery (~13%) rate comparable with ELISA © Fraunhofer
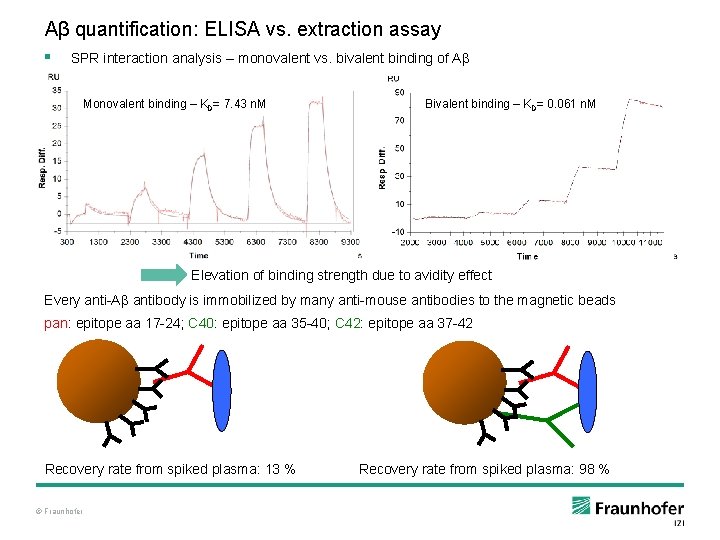
Aβ quantification: ELISA vs. extraction assay § SPR interaction analysis – monovalent vs. bivalent binding of Aβ Monovalent binding – KD= 7. 43 n. M Bivalent binding – KD= 0. 061 n. M Elevation of binding strength due to avidity effect Every anti-Aβ antibody is immobilized by many anti-mouse antibodies to the magnetic beads pan: epitope aa 17 -24; C 40: epitope aa 35 -40; C 42: epitope aa 37 -42 Recovery rate from spiked plasma: 13 % © Fraunhofer Recovery rate from spiked plasma: 98 %

Aβ quantification: ELISA vs. extraction assay § Quantification of p. E 3 -Aβ in plasma and CSF p. E 3 -Aβ is not quantifiable neither in human plasma nor in human CSF, although these species are the most abundant in brain deposits © Fraunhofer
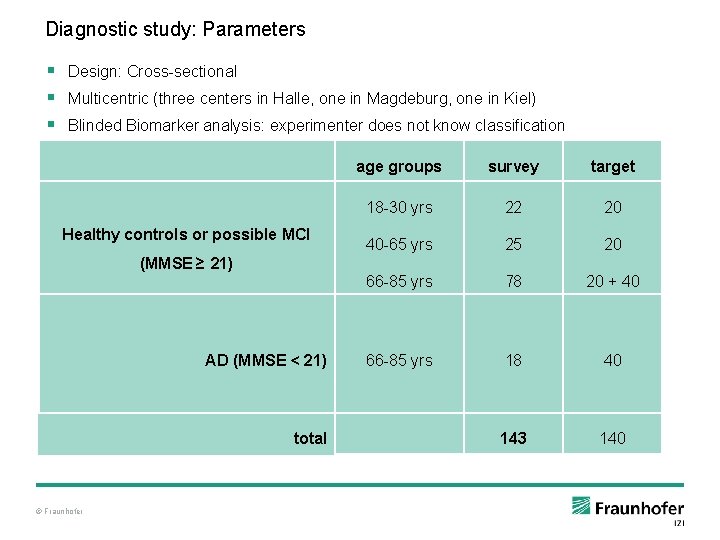
Diagnostic study: Parameters § Design: Cross-sectional § Multicentric (three centers in Halle, one in Magdeburg, one in Kiel) § Blinded Biomarker analysis: experimenter does not know classification Healthy controls or possible MCI age groups survey target 18 -30 yrs 22 20 40 -65 yrs 25 20 66 -85 yrs 78 20 + 40 66 -85 yrs 18 40 143 140 (MMSE ≥ 21) AD (MMSE < 21) total © Fraunhofer

Diagnostic study – Neuropsychology and Biomarker § neuropsychological analysis (independent variable) • learning & memory: CERAD (word lists), WMS-R (narration), ROCF (recall) • attention & executive func. : ROCF (copy), RWT (semantic/phonetic, alternating), TMT (psycho-motor activity/cog. Flexibilit • cognitive decline: premorbid IQ (MWT-B, social formula), “non-hold” function (HAWIE-R Similarity) • functioning: performance activities of daily living (FAST) § Analysis of major depression episode (HADS-D & BDI) – subject exclusion § Genetic risk factor – Apo E allele analysis § plasma and serum biomarker (dependent variables) • plasma Aβ(1 -40) • plasma Aβ(1 -42) • total h. CCL 2 / p. E 1 -h. CCL 2 in serum • auto antibodies against Aβ(1/p. E 3 -x): Ig. G 2, Ig. G 3, Ig. M, Ig. A, Ig. G, Ig’s • pro-inflammatory cytokines: IL-10, IL-1β, IFN-γ, IL-6 and TNF-α © Fraunhofer
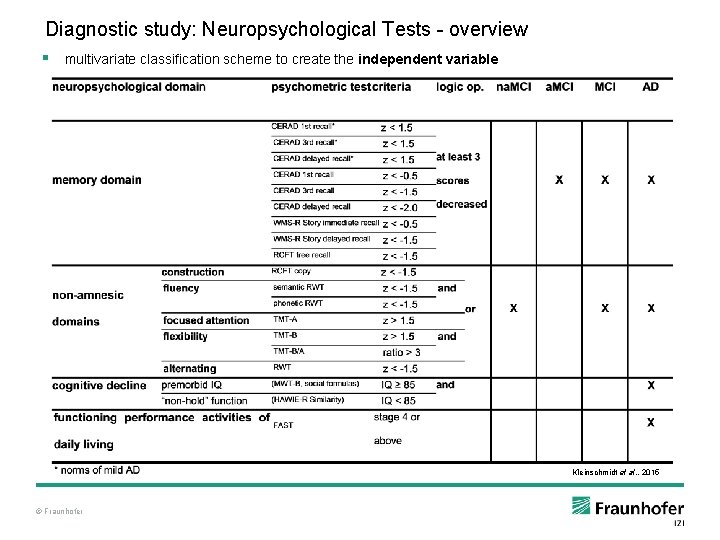
Diagnostic study: Neuropsychological Tests - overview § multivariate classification scheme to create the independent variable Kleinschmidt et al. , 2015 © Fraunhofer
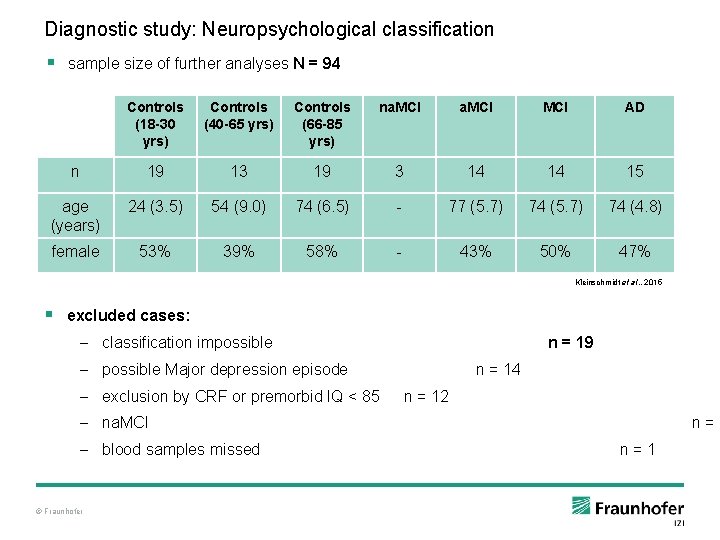
Diagnostic study: Neuropsychological classification § sample size of further analyses N = 94 Controls (18 -30 yrs) Controls (40 -65 yrs) Controls (66 -85 yrs) na. MCI MCI AD n 19 13 19 3 14 14 15 age (years) 24 (3. 5) 54 (9. 0) 74 (6. 5) - 77 (5. 7) 74 (4. 8) female 53% 39% 58% - 43% 50% 47% Kleinschmidt et al. , 2015 § excluded cases: – classification impossible n = 19 – possible Major depression episode – exclusion by CRF or premorbid IQ < 85 n = 14 n = 12 – na. MCI – blood samples missed © Fraunhofer n=3 n=1
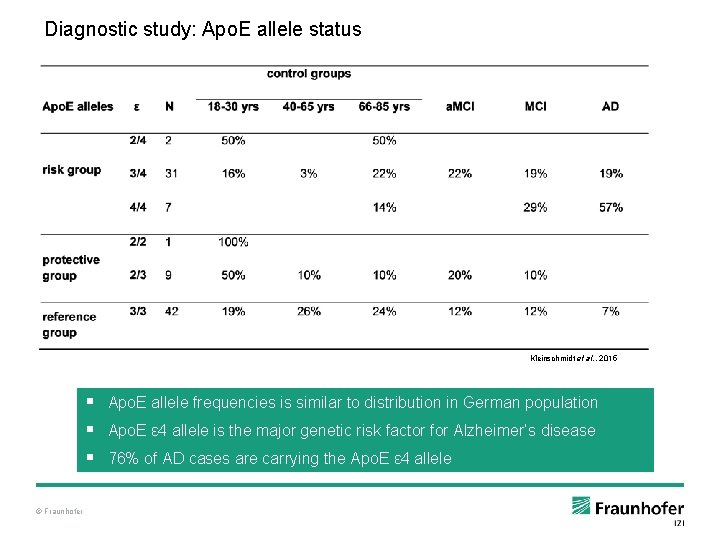
Diagnostic study: Apo. E allele status Kleinschmidt et al. , 2015 § Apo. E allele frequencies is similar to distribution in German population § Apo. E ε 4 allele is the major genetic risk factor for Alzheimer’s disease § 76% of AD cases are carrying the Apo. E ε 4 allele © Fraunhofer
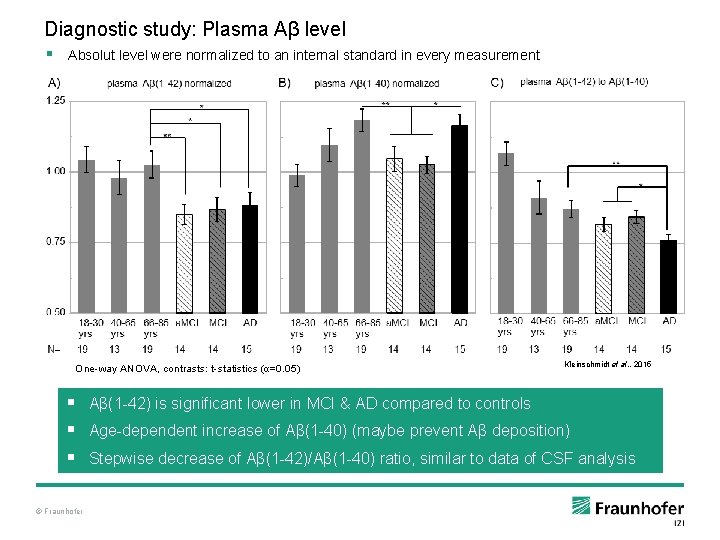
Diagnostic study: Plasma Aβ level § Absolut level were normalized to an internal standard in every measurement One-way ANOVA, contrasts: t-statistics (α=0. 05) Kleinschmidt et al. , 2015 § Aβ(1 -42) is significant lower in MCI & AD compared to controls § Age-dependent increase of Aβ(1 -40) (maybe prevent Aβ deposition) § Stepwise decrease of Aβ(1 -42)/Aβ(1 -40) ratio, similar to data of CSF analysis © Fraunhofer
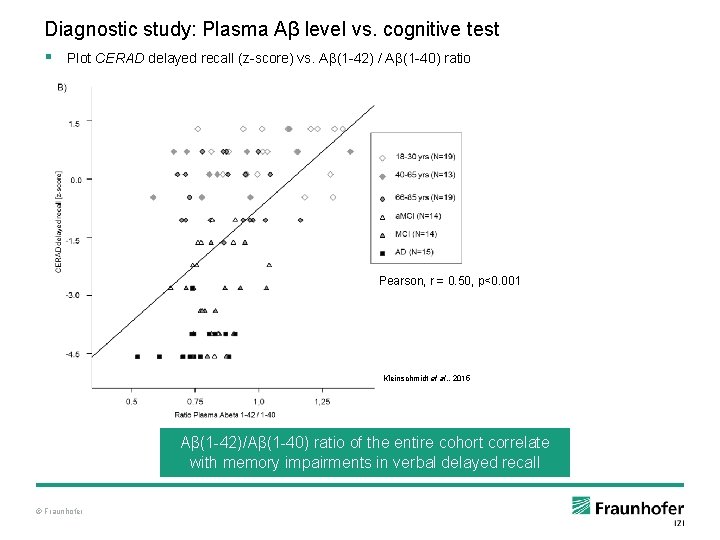
Diagnostic study: Plasma Aβ level vs. cognitive test § Plot CERAD delayed recall (z-score) vs. Aβ(1 -42) / Aβ(1 -40) ratio Pearson, r = 0. 50, p<0. 001 Kleinschmidt et al. , 2015 Aβ(1 -42)/Aβ(1 -40) ratio of the entire cohort correlate with memory impairments in verbal delayed recall © Fraunhofer
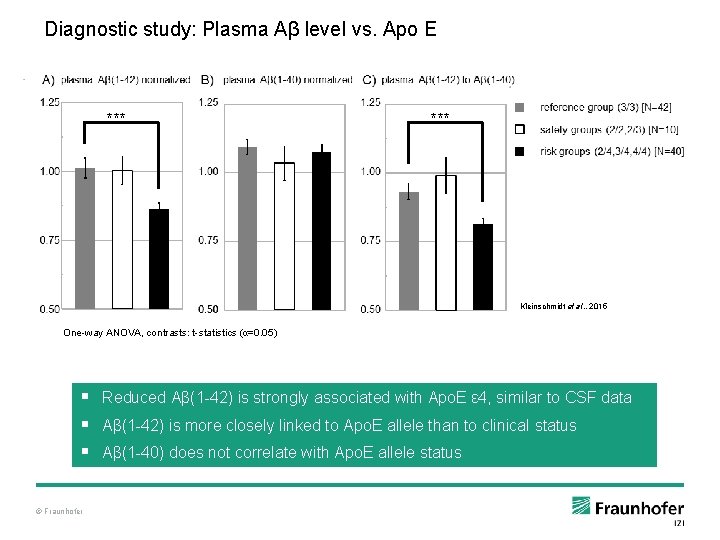
Diagnostic study: Plasma Aβ level vs. Apo E *** Kleinschmidt et al. , 2015 One-way ANOVA, contrasts: t-statistics (α=0. 05) § Reduced Aβ(1 -42) is strongly associated with Apo. E ε 4, similar to CSF data § Aβ(1 -42) is more closely linked to Apo. E allele than to clinical status § Aβ(1 -40) does not correlate with Apo. E allele status © Fraunhofer
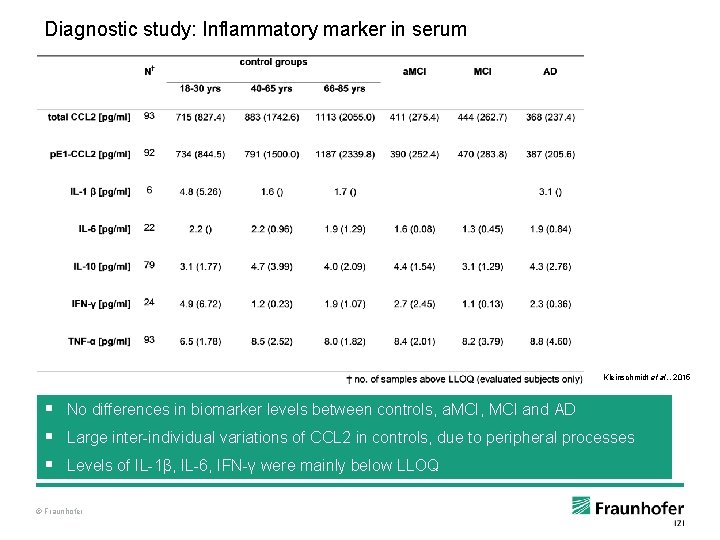
Diagnostic study: Inflammatory marker in serum Kleinschmidt et al. , 2015 § No differences in biomarker levels between controls, a. MCI, MCI and AD § Large inter-individual variations of CCL 2 in controls, due to peripheral processes § Levels of IL-1β, IL-6, IFN-γ were mainly below LLOQ © Fraunhofer
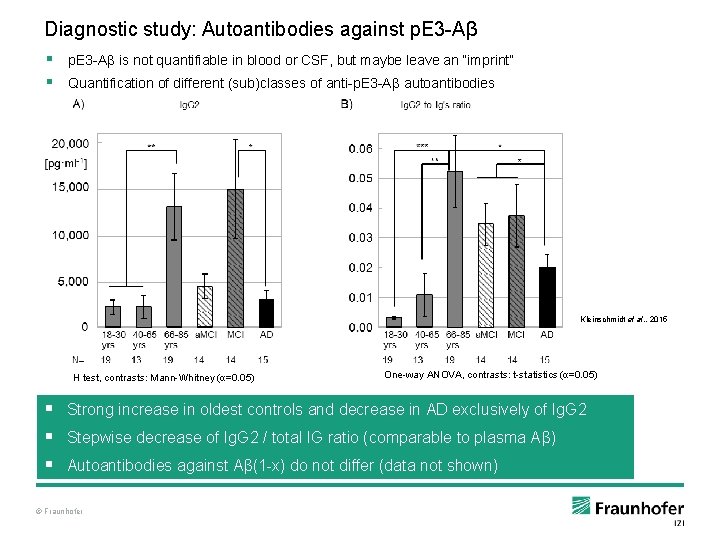
Diagnostic study: Autoantibodies against p. E 3 -Aβ § p. E 3 -Aβ is not quantifiable in blood or CSF, but maybe leave an “imprint” § Quantification of different (sub)classes of anti-p. E 3 -Aβ autoantibodies Kleinschmidt et al. , 2015 H test, contrasts: Mann-Whitney (α=0. 05) One-way ANOVA, contrasts: t-statistics (α=0. 05) § Strong increase in oldest controls and decrease in AD exclusively of Ig. G 2 § Stepwise decrease of Ig. G 2 / total IG ratio (comparable to plasma Aβ) § Autoantibodies against Aβ(1 -x) do not differ (data not shown) © Fraunhofer
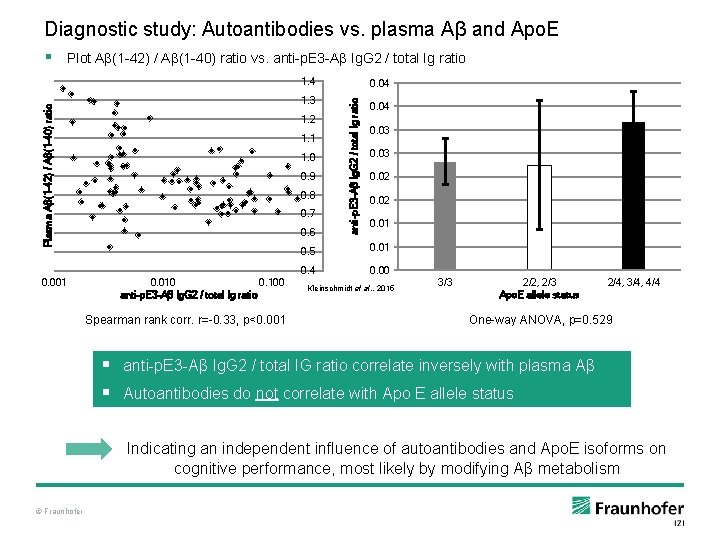
Diagnostic study: Autoantibodies vs. plasma Aβ and Apo. E § Plot Aβ(1 -42) / Aβ(1 -40) ratio vs. anti-p. E 3 -Aβ Ig. G 2 / total Ig ratio 1. 4 0. 001 1. 2 1. 1 1. 0 0. 9 0. 8 0. 7 0. 6 0. 010 0. 100 anti-p. E 3 -Aβ Ig. G 2 / total Ig ratio Spearman rank corr. r=-0. 33, p<0. 001 anti-p. E 3 -Aβ Ig. G 2 / total Ig ratio Plasma Aβ(1 -42) / Aβ(1 -40) ratio 1. 3 0. 04 0. 03 0. 02 0. 01 0. 5 0. 01 0. 4 0. 00 Kleinschmidt et al. , 2015 3/3 2/2, 2/3 Apo. E allele status 2/4, 3/4, 4/4 One-way ANOVA, p=0. 529 § anti-p. E 3 -Aβ Ig. G 2 / total IG ratio correlate inversely with plasma Aβ § Autoantibodies do not correlate with Apo E allele status Indicating an independent influence of autoantibodies and Apo. E isoforms on cognitive performance, most likely by modifying Aβ metabolism © Fraunhofer
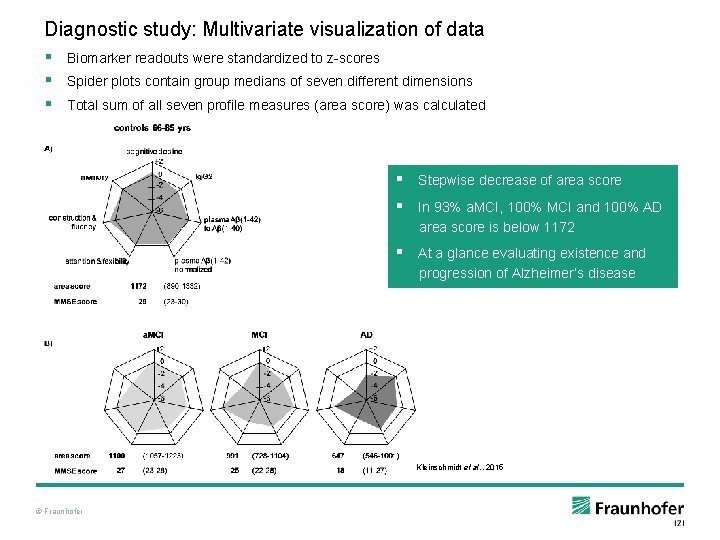
Diagnostic study: Multivariate visualization of data § Biomarker readouts were standardized to z-scores § Spider plots contain group medians of seven different dimensions § Total sum of all seven profile measures (area score) was calculated § Stepwise decrease of area score § In 93% a. MCI, 100% MCI and 100% AD area score is below 1172 § At a glance evaluating existence and progression of Alzheimer’s disease Kleinschmidt et al. , 2015 © Fraunhofer

Acknowledgements Prof. Hans-Ulrich Demuth Prof. Bernd Leplow Kathrin Gnoth Robby Schönfeld Daniel Bittner Nadine Taudte Nadine Jänckel Mercedes Scharfe Medical facilities in Halle: Gallmed Gmb. H (Monitoring) Clinics for geriatrics of Diakonie Neurology Eger & Jakob Claudia Göttlich Anja Weber © Fraunhofer Prof. Josef Aldenhoff
- Slides: 22