Colors and Electromagnetic Waves Bands of Electromagnetic Wave

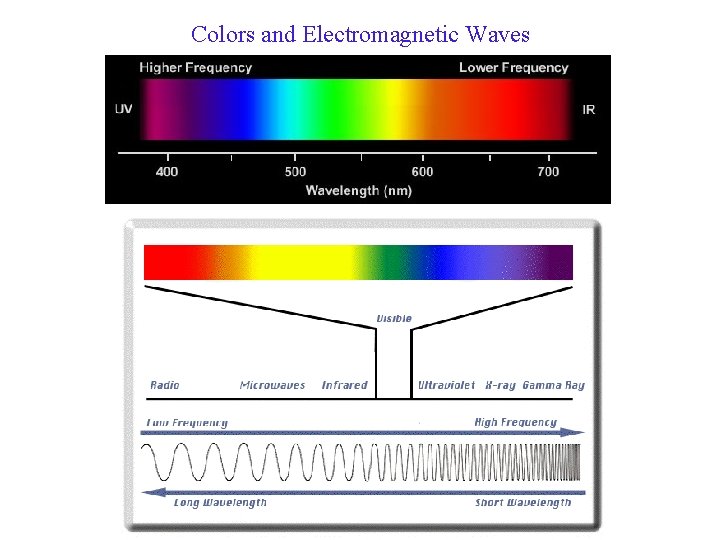
Colors and Electromagnetic Waves
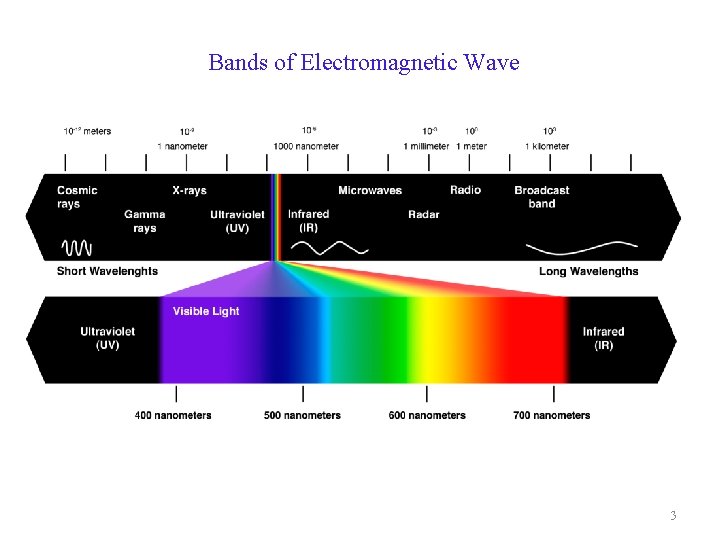
Bands of Electromagnetic Wave 3
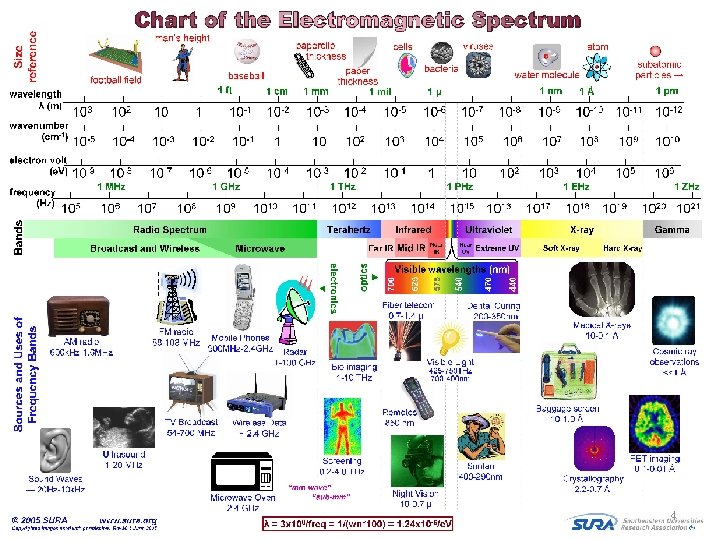
4
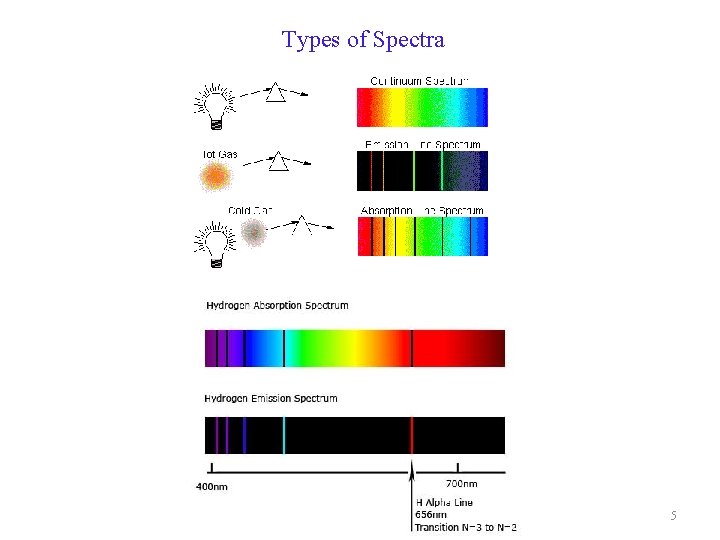
Types of Spectra 5

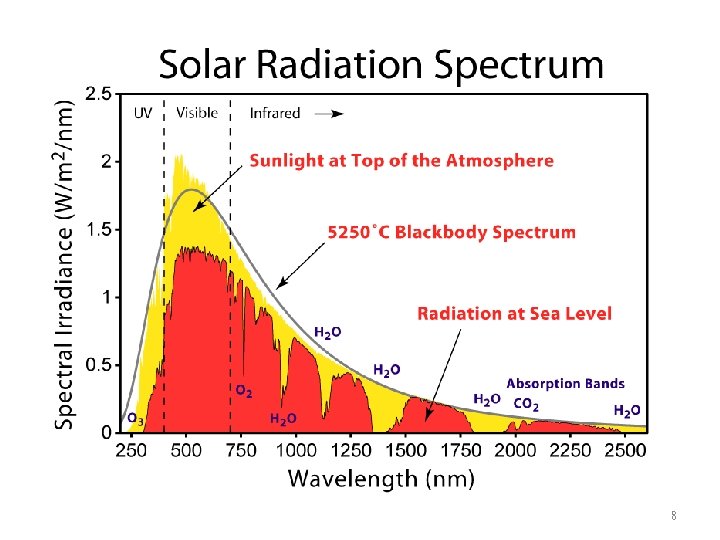
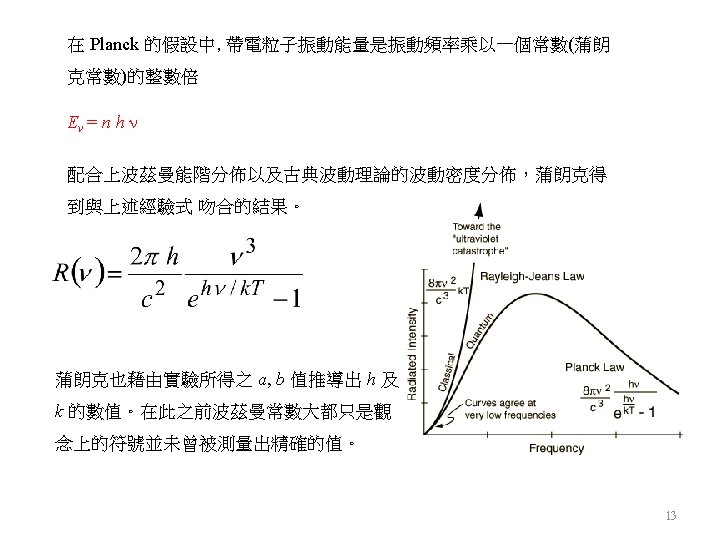
8
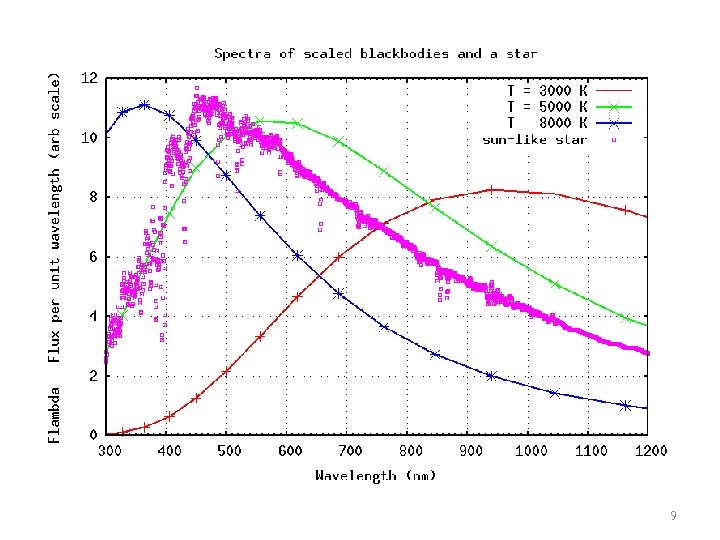
9

Cosmic Microwave Background Discovered in 1964 by Arno Penzias and Robert Wilson in Bell Laboratory. (1978 Nobel Prize) Smooth with very small variation 10
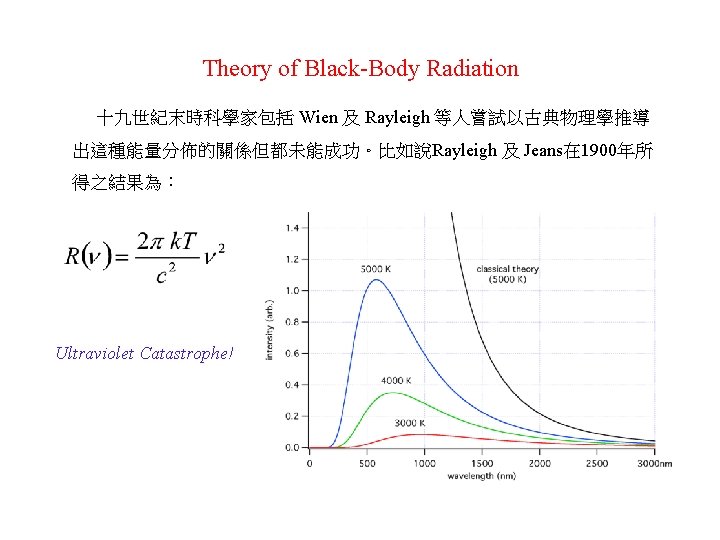



The Photoelectric Effect 15


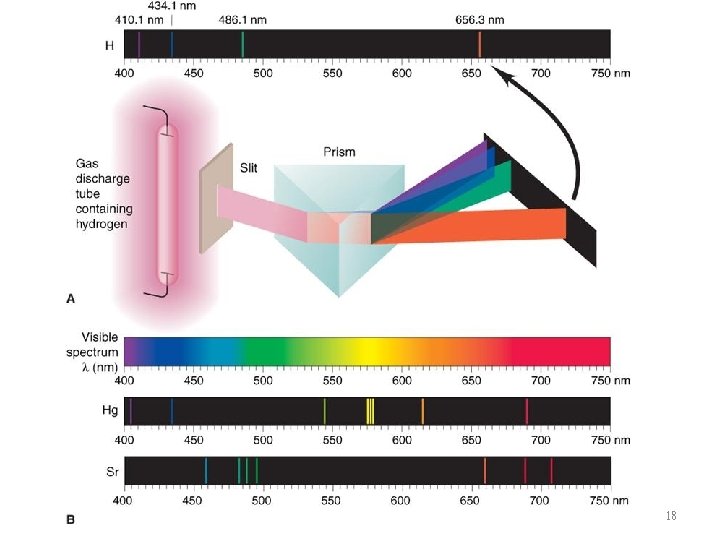
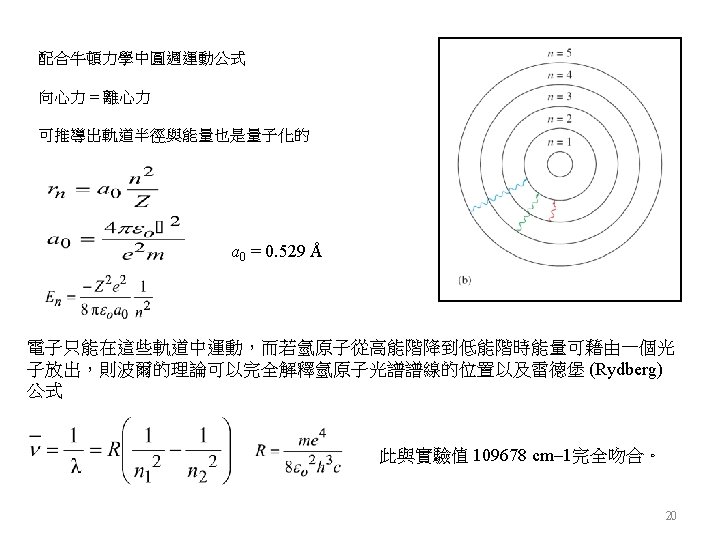
The Line Spectrum of Hydrogen 17
18


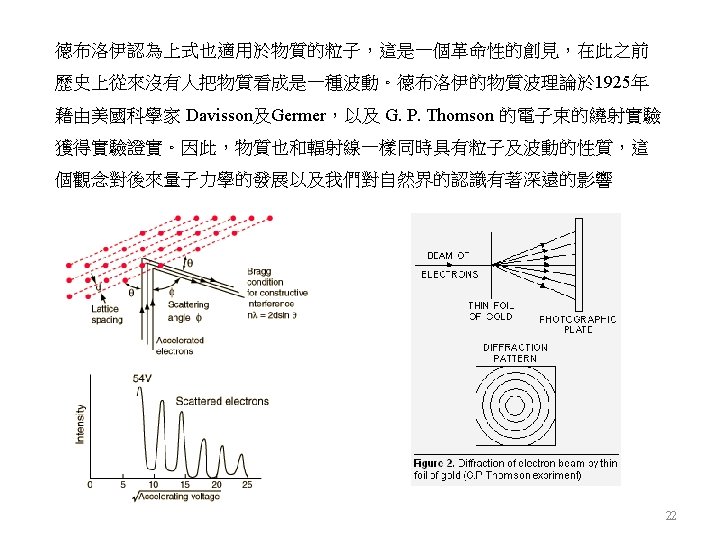

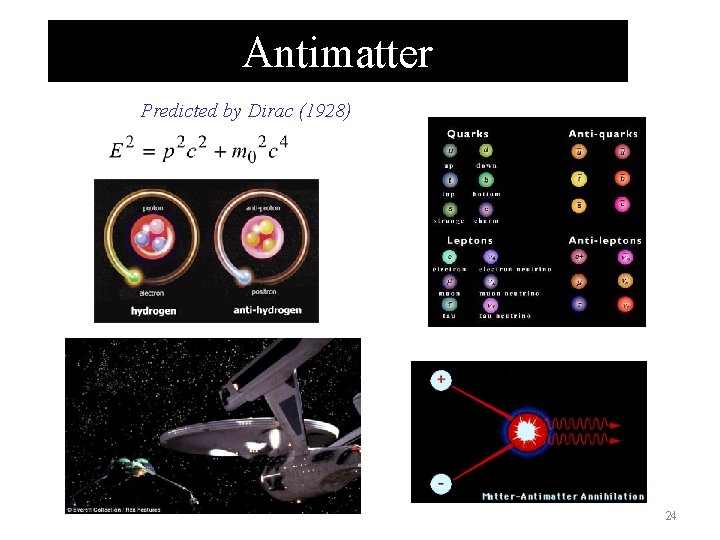
Antimatter Predicted by Dirac (1928) 24

Schrödinger’s Cat 26

The Double Slit Experiment 27
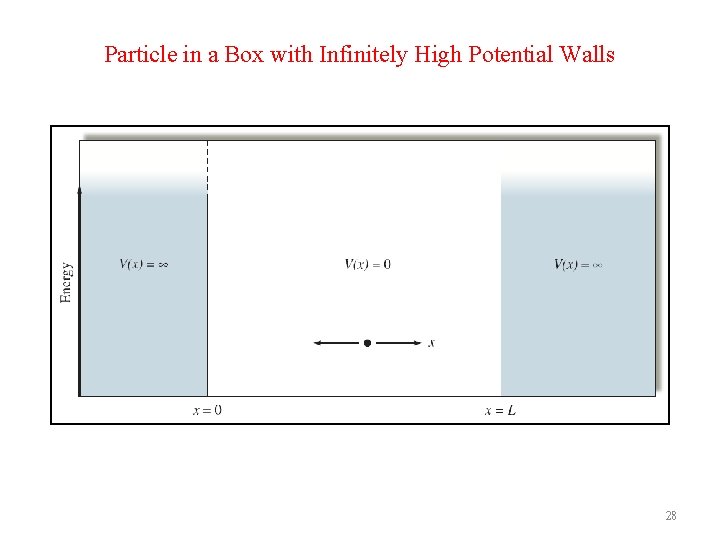
Particle in a Box with Infinitely High Potential Walls 28
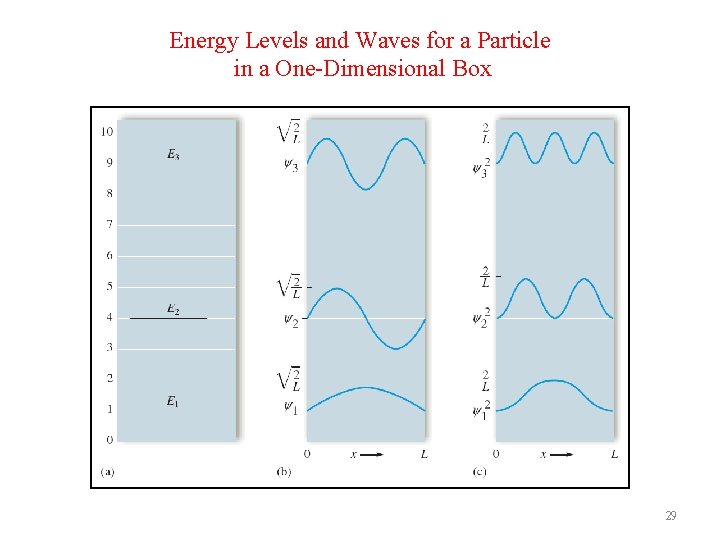
Energy Levels and Waves for a Particle in a One-Dimensional Box 29
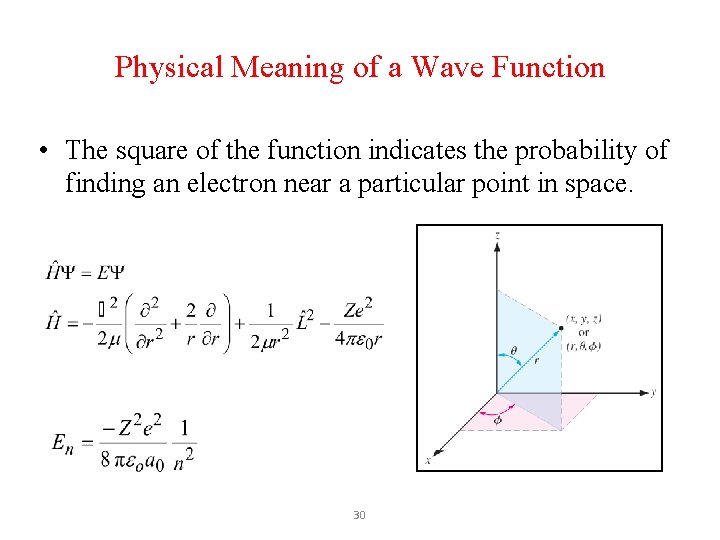
Physical Meaning of a Wave Function • The square of the function indicates the probability of finding an electron near a particular point in space. 30

31
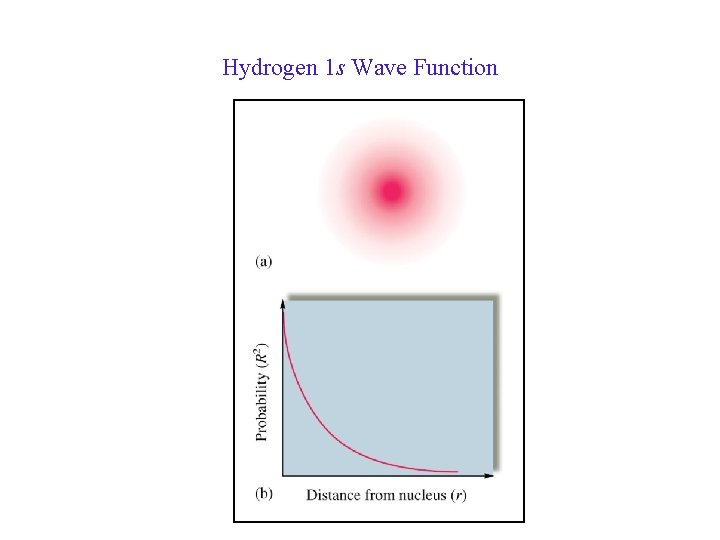
Hydrogen 1 s Wave Function 32
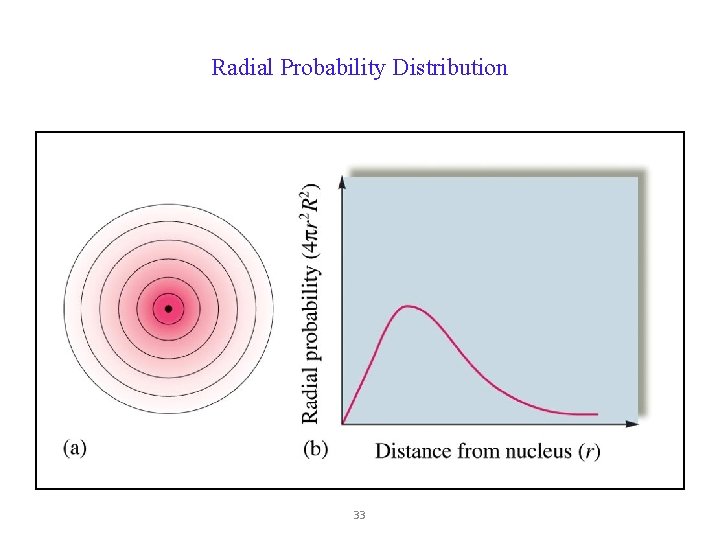
Radial Probability Distribution 33

34


Orbital Energies When electrons are placed in a particular quantum level, they “prefer” the orbitals in the order s, p, d, and then f. When placed in ainparticular Whenelectronsareare placed a particular quantum “prefer” thethe orbitals in the quantumlevel, they “prefer” orbitals in order s, p, d, the order s, and p, d, then andf. then f. 36
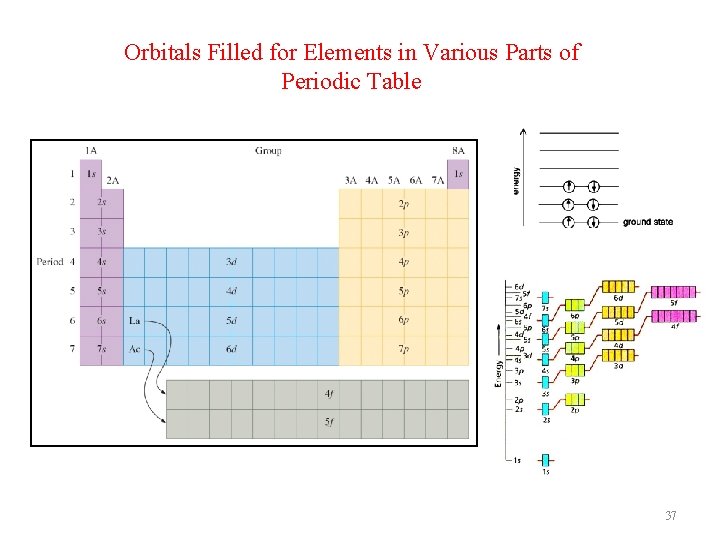
Orbitals Filled for Elements in Various Parts of Periodic Table 37
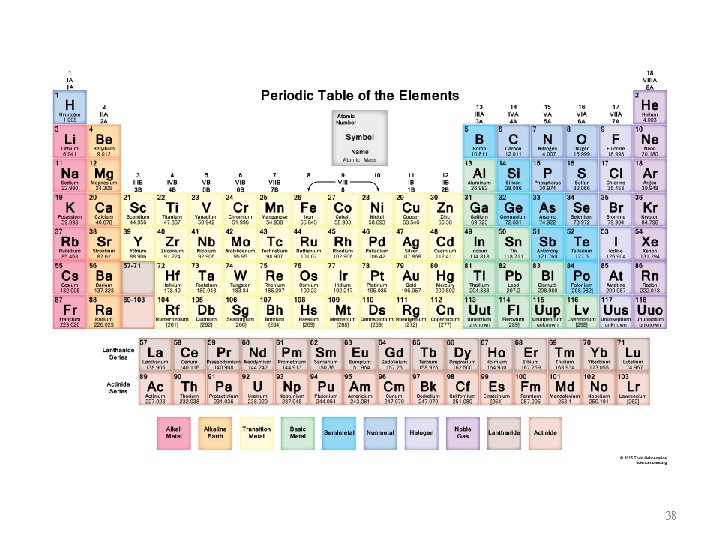
38
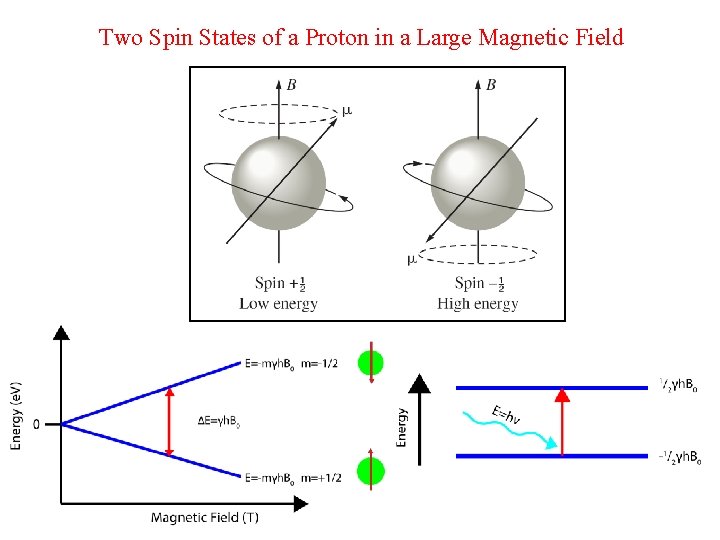
Two Spin States of a Proton in a Large Magnetic Field
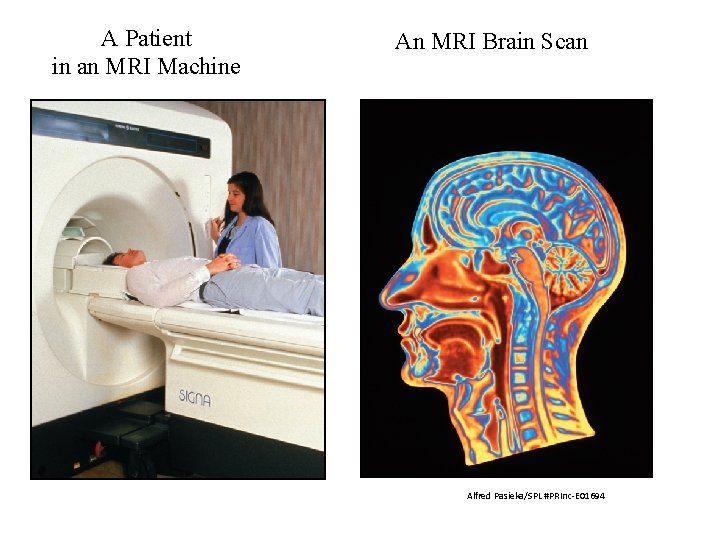
A Patient in an MRI Machine An MRI Brain Scan Alfred Pasieka/SPL #PRInc-E 01694
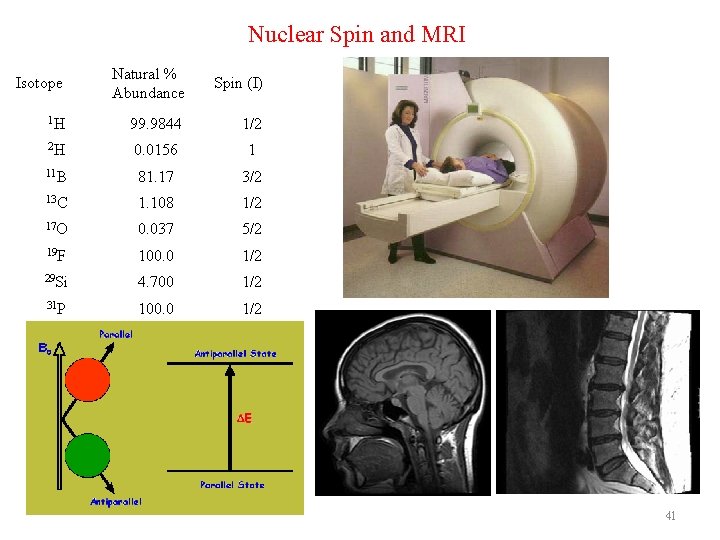
Nuclear Spin and MRI Isotope Natural % Abundance Spin (I) 1 H 99. 9844 1/2 2 H 0. 0156 1 11 B 81. 17 3/2 13 C 1. 108 1/2 17 O 0. 037 5/2 19 F 100. 0 1/2 29 Si 4. 700 1/2 31 P 100. 0 1/2 41
- Slides: 41