Colorimeter Labs What is the Relationship Between the

Colorimeter Labs What is the Relationship Between the Amount of Transmitted Light Through a Solution and Its Concentration? Determination of the Amount of Copper in Brass

Thursday’s Lab • The important thing to take away from Thursday’s lab-that you need to use on Friday-is how to setup a dilution series for a standard solution, and use the colorimeter to make a calibration curve. • Prelab questions need to be answered on plain paper. • Postlab questions will go in your notebooks. If you have questions here, we can discuss it next week.
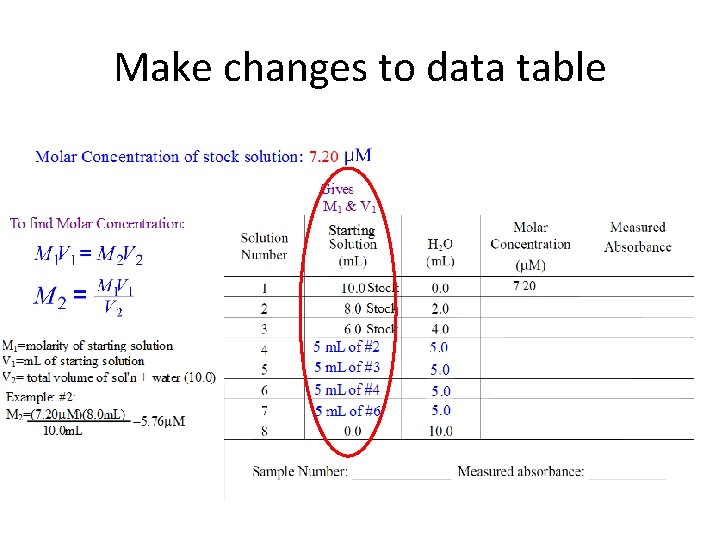
Make changes to data table

• You will need to print your interpolated graph. (See next slide. ) Copy your absorbance values into your notebook. If we have trouble printing, you can enter your data into Logger. Pro Light or Excel later.

Results • From the analysis tab, do a curve fit-linear. • From the analysis tab, interpolate your absorption value for the brass solution. • While on that screen, try to autofit your graph into the available window. • Print the screen from the file menu.

Using a pipet and pipet filler: Watch video below https: //www. youtube. com/watch ? v=d 7 Dc 3 i. WKo 70

Setting up the Lab. Quest http: //www. vernier. com/training/videos/ play/? video=197
Friday’s Lab Determination of Percent Copper in Brass

Prelab Questions • Part I: Do on a separate sheet of paper. Turn in with lab. • Part II: #1: Here is the equation (You’re welcome!) Cu(s) + 4 HNO 3 (aq) Cu(NO 3)2(aq) + 2 NO 2(g) + 2 H 2 O(l) • Answer #2 & #3 in your lab notebooks before your procedure for Part II. • For #4, use the graphed data from the Prelab questions in Part I to find the [Cu].

Procedure-Part I • When writing your procedures, make the following changes. See next slide As written in handout:

Procedure-Part I • Page 2, direction #2 tells you to make the standard Cu(NO 3)2 solution. I will have this already made up for you. The solution will be 0. 400 M.
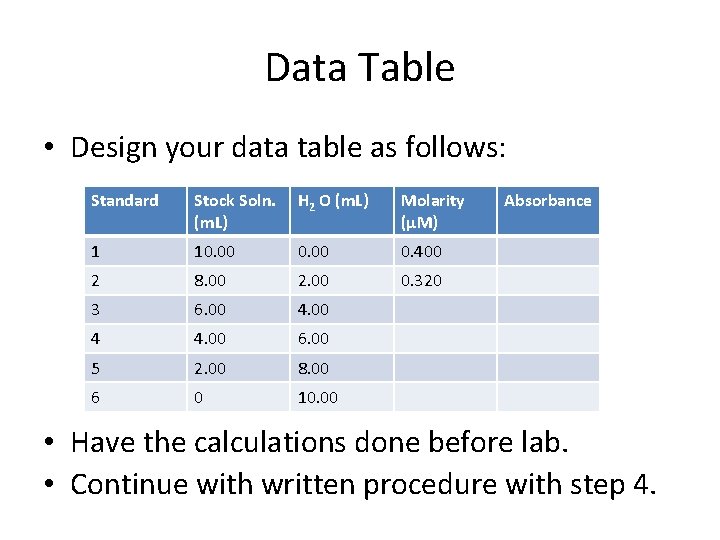
Data Table • Design your data table as follows: Standard Stock Soln. (m. L) H 2 O (m. L) Molarity (µM) 1 10. 00 0. 400 2 8. 00 2. 00 0. 320 3 6. 00 4 4. 00 6. 00 5 2. 00 8. 00 6 0 10. 00 Absorbance • Have the calculations done before lab. • Continue with written procedure with step 4.

Procedure-Part II • Replace the above instruction with what follows on the next slide. • You can shorten this in your write-up.

Procedure-Part II • Obtain a piece of brass and a weighing dish. • Cut the brass into 5 -6 pieces. (This will shorten the time necessary for the reaction. ) • Get another weighing dish and zero the balance with the empty dish on the platform. • Add the brass to the dish and find the mass of the brass to nearest 0. 001 g. • Add the brass to a 25. 0 m. L volumetric flask.

Reaction with Nitric Acid •

Reaction with Nitric Acid • IN THE FUME HOOD add the correct amount of acid to the volumetric flask. WEAR APRON, GOGGLES AND GLOVES. Mr Dowdy or myself must be with you for this step. • Continue with procedure for Part II, Step #4 and 5.

On Friday: • Divide and conquer! One or two partners prepare the standard solutions as listed on the following slides for Part I and the other prepare the brass sample for Part II.

Results • From the analysis tab, do a curve fit-linear. • From the analysis tab, interpolate your absorption value for the brass solution. • While on that screen, try to autofit your graph into the available window. • Print the screen from the file menu.

Postlab Questions • You will answer these in your notebook before you write a conclusion.

Disposal & Clean-up • Cu(NO 3)2 solutions can be rinsed down the drain. • Use test tube brush and soap to clean test tubes and volumetric flasks. • Return beakers, cylinders to glass front cabinets. • Return test tube racks to center cabinet at your station. • Return other materials to back table.
- Slides: 20