Colorado Department of Public Health and the Environment

Colorado Department of Public Health and the Environment Frequently Asked Questions Daniel Goetz – CDPHE Tom Fawks – Consultant Pharmacist

Online Resources? CDPHE Medical and Pharmaceutical Waste Guidance Documents can be found here: https: //www. colorado. gov/pacific/cdp he/medical-and-pharmaceutical-waste -guidance

Have Questions? Hazardous Waste Contact Dan Goetz: 303 -692 -3341 Sharps, Biohazardous, Medical Waste (non RCRA waste) Contact Jace Driver: 303 -691 -4059 Household Medical Waste Contact

Regulatory Updates 1. DEA regulatory update (Sept 2014) 2. EPA regulatory update (2015) 3. Health Facilities regulatory update (2016)

Highlights from 2015: 279, 000 units of medicine donated, valued at over $120, 000 (NADAC). Retail price would be valued at 3 x that number. We were able to transfer 58, 000 units worth $13, 500 to a network of free and charitable providers. Highlights of 2016 through late March: 102, 000 units of medicine worth $55, 000. Transfers 19, 000 units of medicine to a network of 6 clinics worth over $11, 000.

Interested in learning more medicine donation? Is there a local charitable clinic you think would be interested in receiving donated medicine? Contact: Kiah Williams, Director kiah@sirum. org • 650. 488. 7434 office • sirum. org

Hazardous Medication Disposal Questions encountered by a consultant pharmacist Tom Simpleman Consultant Pharmacist RPh, CGP, FASCP, CPHQ www. fawks. com 720 -281 -3512

Disposal of solutions • How do I dispose of Intravenous solutions or Irrigation Solutions? i. e. Normal Saline, D 5 W, Lactated Ringers

Commercially Available Disposal Systems • Can we really just throw it away in normal trash?

Commercially Available Disposal Systems • • • Cactus sink Drug Buster Rx Destroyer Medsaway C 2 R

Commercially Available Disposal Systems Dispose of it in regular trash?
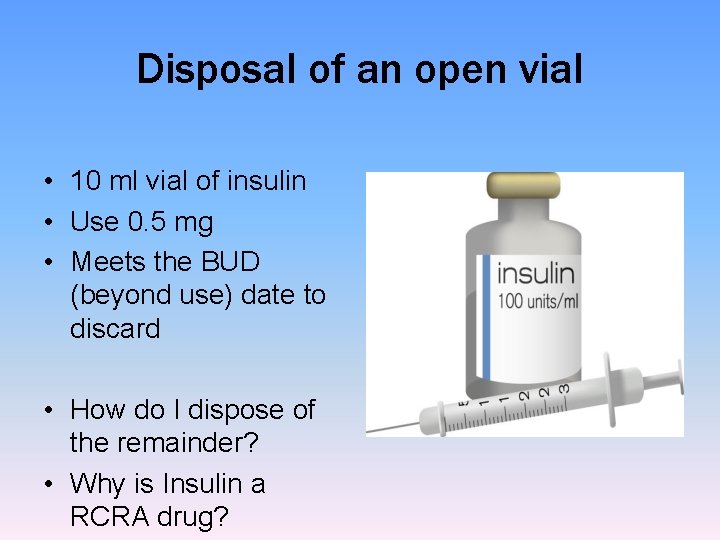
Disposal of an open vial • 10 ml vial of insulin • Use 0. 5 mg • Meets the BUD (beyond use) date to discard • How do I dispose of the remainder? • Why is Insulin a RCRA drug?
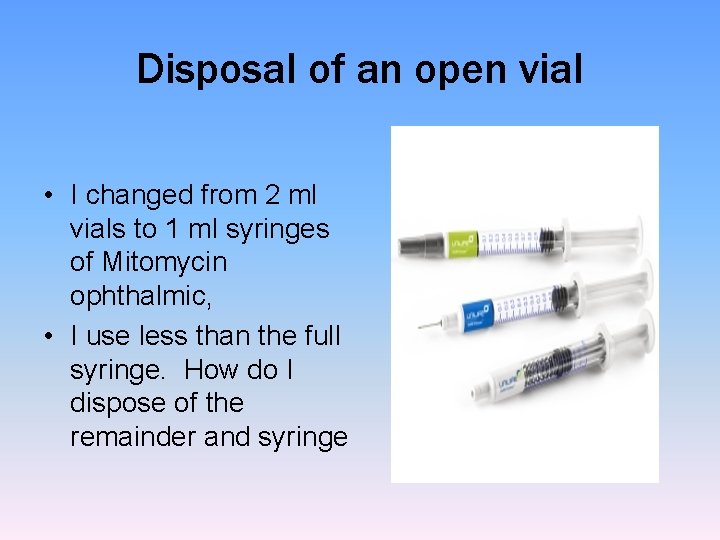
Disposal of an open vial • I changed from 2 ml vials to 1 ml syringes of Mitomycin ophthalmic, • I use less than the full syringe. How do I dispose of the remainder and syringe

ASC location • ASC located adjacent to a hospital building. • Can we use their medication disposal system? • Are we responsible for maintaining records of how the medical wasted is disposed? • If so, what records are we responsible for keeping and how long?

Medication Disposal Companies • How can I find medication disposal companies? • Are they expensive?

Companies that do not respond • Contacted Steri-Cycle – Get run around to find contact – They promise someone will call back – Don’t receive call – Contacted, promised a representative would stop by the next week – Never stopped or returned call • What are my responsibilities in this situation?

How much? • Where did the 2. 2 pounds come from? • How frequently do I need to send it to the disposal company? • Do I need to inspect the containers they are stored in regularly? • Can I return the RCRA waste to a pharmacy and not have to track it?

Tracking • What documentation is needed? • Is simply stating I sent it to Stericycle enough? • Why do I have to keep this documentation? • Is it OK if only one person does everything?

Training • What education is required? • How many should be trained? • Should responsibilities be assigned? – If so which ones? • What about non. RCRA meds such as antibiotics?

Question: How can I ensure I am protected from liability once the hazardous waste leaves my facility?

Answer A CESQG is required to ensure that their hazardous waste is delivered to a permitted hazardous waste treatment, storage or disposal facility (TSD). Although CESQGs are not required to use a hazardous waste manifest to ship hazardous medication waste, this would be the best method to ensure that your hazardous medication waste was properly disposed. The TSD is required to send a signed copy of the manifest back to the generator. For hazardous medication waste that is sent to a reverse distributor, the Department accepts that a statement from the reverse distributor, on a contract or invoice, that they properly dispose of all hazardous medication wastes at a permitted TSD as proof that the generator has met the CESQG requirement. However, this is not a guarantee that you are immune from liability if the reverse distributor fails to properly dispose of the waste.

What about epinephrine salts or nitroglycerin? • Epinephrine salts are not P 042 hazardous waste in Colorado. • Medicinal nitroglycerin is generally not reactive (i. e. , not P 081 hazardous waste) but 5 mg/ml concentration may be ignitable (D 001).

QUESTIONS?
- Slides: 24